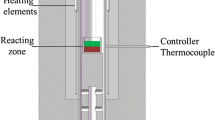
Abstract
Energy storage and recovery using the redox reaction of tungsten/tungsten-oxide is proposed. The system will store energy as tungsten metal by reducing the tungsten oxide with hydrogen. Thereafter, steam will be used to reoxidize the metal and recover the hydrogen. The volumetric energy density of W for storing hydrogen by this process is 21 kWh/L based on the lower heating value (LHV) of hydrogen. The main objective of this investigation was to study the kinetics of the reduction process of tungsten oxide (WO3) and determine the optimum parameters for rapid and complete reduction. Theoretical treatment of isothermal kinetics has been extended in the current work to the reduction of tungsten oxide in powder beds. Experiments were carried out using a thermogravimetric technique under isothermal conditions at different temperatures. The reaction at 1073 K (800 °C) was found to take place in the following sequence: WO3 → WO2.9 → WO2.72 → WO2 → W. Expressions for the last three reaction rate constants and activation energies have been calculated based on the fact that the intermediate reactions proceed as a front moving at a certain velocity while the first reaction occurs in the entire bulk of the oxide. The gas–solid reaction kinetics were modeled mathematically in terms of the process parameters. This model of the reduction has been found to be accurate for bed heights above 1.5 mm and hydrogen partial pressures greater than 3 pct, which is ideal for implementing the energy storage concept.



















Similar content being viewed by others
References
HSC Thermodynamic Software: Outokumpo Res. Oy, 5th ed., Pori, Finland, 2003.
J.A. Bustnes, D.U. Sichen, and S. Seetharaman: Metall. Trans. B, 1993, vol. 24B, pp. 475–80.
D.U. Sichen and S. Seetharaman: Metall. Trans. B, 1992, vol. 23B, pp. 317–24.
D.S. Venables and M.E. Brown: Thermochim. Acta, 1996, vol. 285, no. 2, pp. 361–82.
X.-W. Wu: Trans. Nonferr. Met. Soc. China, 2009, vol. 19, pp. 785–89.
W.D. Schubert: Int. J. Refract. Met. H., 1990, vol. 9, pp. 178–91.
M.S. Whittingham and P.G. Dickens: 7 th Int. Symp. on the Reactivity of Solids, Chapman & Hall, London, U.K., 1972, pp. 640–50.
Yuntao Cui, J. Wang, W. Liu, and X. Wang: J. Rare Earth, 2010, vol. 28, special issue, pp. 202–05.
N.E. Fouad: J. Anal. Appl. Pyrol., 1997, vol. 1, no. 44, pp. 13–28.
A. Lackner: Int. J. Refract. Met. Hard Mater., 1996, vol. 14 (5, 6), pp. 383–91.
J.M. Smith: Chemical Engineering Kinetics, 3rd ed., McGraw-Hill Book Company, New York, NY, 1981, pp. 643–47.
Acknowledgments
The financial support of the Boston University (Ignition Award) is gratefully acknowledged. The assistance of Mr. Jarrod Milshtein and Dr. Stephen Topping is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted September 12, 2011.
Rights and permissions
About this article
Cite this article
Haboury, R., Pal, U.B., Zink, P.A. et al. Study of an Energy Storage and Recovery Concept Based on the W/WO3 Redox Reaction: Part I. Kinetic Study and Modeling of the WO3 Reduction Process for Energy Storage. Metall Mater Trans B 43, 1001–1010 (2012). https://doi.org/10.1007/s11663-012-9656-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-012-9656-0