Abstract
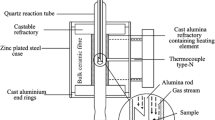
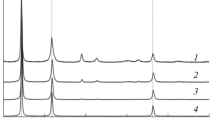
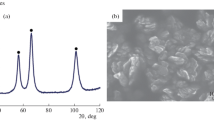
The product microstructures developed during the reduction of dense synthetic nickel oxide in H2-N2 and H2-H2O mixtures between 300 °C and 1000 °C are reported. A number of distinctly different types of nickel product structures have been identified, namely, coarse fibrous nickel with fissures, fine porous nickel-planar interface, large porous nickel-irregular interface, and dense nickel layer. The influences of gas composition and temperature on the conditions for formation of these microstructures, and their evolution with temperature and time, have been systematically investigated. The condition for formation of the various microstructure types was summarized on morphology maps as functions of reducing gas partial pressure, thermodynamic driving force, and reaction temperature.













Similar content being viewed by others
Notes
JEOL is a trademark of Japan Electron Optics Ltd., Tokyo.
References
W. Betteridge: Nickel and Its Alloys, John Wiley & Sons Inc., Brisbane, 1984, pp. 171–74.
S. Luidold and H. Antrekowitsch: JOM, 2007, vol. 59 (6), pp. 20–26.
L.L. Bergeson: Chem. Process. Mag., 2003, Oct., http://www.chemicalprocessing.com/articles/2003/143.html?page=print (accessed September 19, 2007).
A. Kivnick and N. Hixson: Chem. Eng. Prog., 1952, vol. 48 (8), pp. 394–400.
Y. Iida and K. Shimada: Bull. Chem. Soc. Jpn., 1960, vol. 33 (6), pp. 790–93.
J. Bandrowski, C.R. Bickling, K.H. Yang, and O.A. Hougen: Chem. Eng. Sci., 1962, vol. 17, pp. 379–90.
H. Mine, M. Tokuda, and M. Ohtani: Jpn. Inst. Met., 1970, vol. 34, pp. 814–20.
T. DebRoy and K.P. Abraham: Proc. Physical Chemistry of Process Metallurgy: The Richardson Conf., Institute of Mining and Metallurgy, 1973, pp. 85–93.
J.W. Evans, S. Song, and C.E. Leon-Sucre: Metall. Mater. Trans. B, 1976, vol. 7B, pp. 55–65.
A.H. Rashed and Y.K. Rao: Chem. Eng. Commun., 1996, vol. 156, pp. 1–30.
Y.K. Rao and A.H. Rashed: Trans. Inst. Min. Metall. (Sect. C: Mineral Process. Extr. Metall.), 2001, vol. 110, pp. 1–6.
J.T. Richardson, R. Scates, and M.V. Twigg: Appl. Catal. A-Gen., 2003, vol. 246, pp. 137–50.
T.A. Utigard, M. Wu, G. Plascencia, and T. Marin: Chem. Eng. Sci., 2005, vol. 60 (7), pp. 2061–68.
M.A. Rhamdhani, E. Jak, and P.C. Hayes: Metall. Mater. Trans. B, 2008, vol. 39B, pp. 218–33.
M.A. Rhamdhani, E. Jak, and P.C. Hayes: Metall. Mater. Trans. B, 2008, vol. 39B, pp. 234–45.
T. Hidayat, M.A. Rhamdhani, E. Jak, and P. Hayes: Miner. Eng., 2008, vol. 21 (2), pp. 157–66.
S.P. Matthew, D.H. St.John, J.V. Hardy, and P.C. Hayes: Metallography, 1985, vol. 17, pp. 367–79.
W. Pluschkell and B.V.S. Sarma: Arch. Eisenhuttenwes., 1974, vol. 45 (1), pp. 23–31.
J.R. Heffelfinger and C.B. Carter: Surf. Sci., 1997, vol. 389 (1–3), pp. 188–200.
W.D. Kingery, H.K. Bowen, and D.R. Uhlman: Introduction to Ceramics, 2nd ed., John Wiley & Sons, Sydney, 1976, pp. 474–75.
Acknowledgments
The authors thank the BHP Billiton Yabulu Refinery and Australian Research Council Linkage program for the financial support, and AusAid for providing a scholarship. The authors also acknowledge Mr. John Fittock and Dr. Joy Morgan (BHP Billiton Yabulu) for their valuable help and critical discussions. The authors also thank Messrs. Yanan Guo and Jiang Chen for carrying out TEM analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted August 24, 2008.
Rights and permissions
About this article
Cite this article
Hidayat, T., Rhamdhani, M., Jak, E. et al. Investigation of Nickel Product Structures Developed during the Gaseous Reduction of Solid Nickel Oxide. Metall Mater Trans B 40, 462–473 (2009). https://doi.org/10.1007/s11663-009-9247-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-009-9247-x