Abstract
Summary
The association between osteoporosis and polycyclic aromatic hydrocarbons was not clear. However, recent studies showed that both osteoporosis and polycyclic aromatic hydrocarbons were associated with reactive oxygen species. So we presumed that polycyclic aromatic hydrocarbons were associated with increased odds of osteoporosis. We found 3-hydroxyfluorene was associated with decreased odds of osteoporosis, while 2-hydroxyfluorene was associated with increased odds of osteoporosis.
Purpose
Previous studies showed that polycyclic aromatic hydrocarbons were involved in the production of reactive oxygen species that played an important role in illness. Other studies demonstrated that the reactive oxygen species might play a role in occurrence of osteoporosis. However, the association between polycyclic aromatic hydrocarbons and osteoporosis was not clear. Therefore, we conducted a study to investigate the relationship between polycyclic aromatic hydrocarbons and osteoporosis.
Methods
A total of 3053 individuals aged 50 years or older were drawn from National Health and Nutritional Examination Survey, 2005–2014. Eight polycyclic aromatic hydrocarbons metabolites (1-hydroxynaphthalene, 2-hydroxynaphthalene, 2-hydroxyfluorene, 3-hydroxyfluorene, 1-hydroxyphenanthrene, 2-hydroxyphenanthrene, and 3-hydroxyphenanthrene and 1-hydroxypyrene) were examined to represent exposure of polycyclic aromatic hydrocarbons in the body. Weighted percentage and weighted mean were used to describe classified variable and continuous variable, respectively. We used weighted logistic regression to estimate the association of polycyclic aromatic hydrocarbons and osteoporosis.
Results
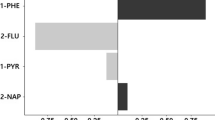
Five hundred seventy-seven (18.9%) osteoporosis patients were included. Among the eight polycyclic aromatic hydrocarbons metabolites, 3-hydroxyfluorene was associated with decreased odds of osteoporosis after controlling socio-demographic status, body mass index, alcohol, previous fracture, parental fracture, glucocorticoid use, dietary calcium intake, smoking, and other polycyclic aromatic hydrocarbons metabolites (OR = 0.63, 95%CI 0.41–0.98). 2-hydroxyfluorene (OR = 1.87, 95%CI 1.07–3.29) was associated with increased odds of osteoporosis. In the nonsmoking group, we found the association of osteoporosis with 2-hydroxyfluorene (OR = 2.56, 95%CI 1.10–5.96) and 3-hydroxyfluorene (OR = 0.41, 95%CI 0.22–0.77) still exists.
Conclusion
Our study found that 3-hydroxyfluorene was associated with decreased odd of osteoporosis, while 2-hydroxyfluorene was associated with increased odds of osteoporosis. Our study was a cross-sectional study, therefore cohort studies or experimental studies about association of PAH and osteoporosis is needed.
Similar content being viewed by others
References
listed Na (2001) NIH consensus development panel on osteoporosis prevention, diagnosis, and therapy, march 7-29, 2000: highlights of the conference. South Med J 94(6):569–573
International Osteoporosis Foundation (2013) Osteoporosis - Incidence and burden. [cited 2018 March]; Available from: http://www.iofbonehealth.org/facts-statistics
Ioannidis G, Papaioannou A, Hopman WM, Akhtar-Danesh N, Anastassiades T, Pickard L, Kennedy CC, Prior JC, Olszynski WP, Davison KS, Goltzman D, Thabane L, Gafni A, Papadimitropoulos EA, Brown JP, Josse RG, Hanley DA, Adachi JD (2009) Relation between fractures and mortality: results from the Canadian multicentre osteoporosis study. Cmaj 181(5):265–271
Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A (2007) Incidence and economic burden of osteoporosis-related fractures in the United States, 2005-2025. J Bone Miner Res 22(3):465–475
National Osteoporosis Foundation What is osteoporosis and what causes it? [cited 2014 22 July ]; Available from: http://nof.org/articles/7
Kanis, J.A., Assessment of osteoporosis at the primary health care level. 2007
Pike C, Birnbaum HG, Schiller M, Sharma H, Burge R, Edgell ET (2010) Direct and indirect costs of non-vertebral fracture patients with osteoporosis in the US. Pharmacoeconomics 28(5):395–409
Department of Health and Human Services, U.S. Bone Health and Osteoporosis: A Report of the Surgeon General. Rockville, MD: U.S. Department of Health and Human Services, Office of the Surgeon General, 2004. [cited 2017 5 April]; Available from: https://www.ncbi.nlm.nih.gov/books/NBK45513/
Freeman DJ, Cattell FCR (1990) Woodburning as a source of atmospheric polycyclic aromatic hydrocarbons. Environ Sci Technol 65(3–4):235–242
Galarneau E (2008) Source specificity and atmospheric processing of airborne PAHs: implications for source apportionment. Atmos Environ 42(35):8139–8149
DeMarini DM (2004) Genotoxicity of tobacco smoke and tobacco smoke condensate: a review. Mutat Res 567(2–3):447–474
Eom SY, Yim DH, Moon SI, Youn JW, Kwon HJ, Oh HC, Yang JJ, Park SK, Yoo KY, Kim HS, Lee KS, Chang SH, Kim YD, Kang JW, Kim H (2013) Polycyclic aromatic hydrocarbon-induced oxidative stress, antioxidant capacity, and the risk of lung cancer: a pilot nested case-control study. Anticancer Res 33(8):3089–3097
Osgood RS, Upham BL, Hill T 3rd, Helms KL, Velmurugan K, Babica P, Bauer AK (2014) Polycyclic aromatic hydrocarbon-induced signaling events relevant to inflammation and tumorigenesis in lung cells are dependent on molecular structure. PLoS One 8(6):e65150
Fu PP, Xia Q, Sun X, Yu H (2012) Phototoxicity and environmental transformation of polycyclic aromatic hydrocarbons (PAHs)-light-induced reactive oxygen species, lipid peroxidation, and DNA damage. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev 30(1):1–41
Henkler F, Stolpmann K, Luch A (2012) Exposure to polycyclic aromatic hydrocarbons: bulky DNA adducts and cellular responses. Exs 101:107–131
Xia Q, Yin JJ, Zhao Y, Wu YS, Wang YQ, Ma L, Chen S, Sun X, Fu P, Yu H (2013) UVA photoirradiation of nitro-polycyclic aromatic hydrocarbons-induction of reactive oxygen species and formation of lipid peroxides. Int J Environ Res Public Health 10(3):1062–1084
About the National Health and Nutrition Examination Survey. Available from: https://www.cdc.gov/nchs/nhanes/about_nhanes.htm
Siris ES, Adler R, Bilezikian J, Bolognese M, Dawson-Hughes B, Favus MJ, Harris ST, Jan de Beur SM, Khosla S, Lane NE, Lindsay R, Nana AD, Orwoll ES, Saag K, Silverman S, Watts NB (2014) The clinical diagnosis of osteoporosis: a position statement from the National Bone Health Alliance Working Group. Osteoporos Int 25(5):1439–1443
Wright NC, Saag KG, Dawson-Hughes B, Khosla S, Siris ES (2017) The impact of the new National Bone Health Alliance (NBHA) diagnostic criteria on the prevalence of osteoporosis in the USA. Osteoporos Int 28(4):1225–1232
National Center for Health Statistics. National Health and Nutrition Examination Survey: Plan and Operations, 1999–2010. Available from: https://wwwn.cdc.gov/nchs/data/series/sr01_056.pdf
Looker AC, Orwoll ES, Johnston CC Jr, Lindsay RL, Wahner HW, Dunn WL, Calvo MS, Harris TB, Heyse SP (1997) Prevalence of low femoral bone density in older U.S. adults from NHANES III. J Bone Miner Res 12(11):1761–1768
Jung KJ, Chung CY, Park MS, Kwon SS, Moon SY, Lee IH, Kim KH, Lee KM (2016) Different reference BMDs affect the prevalence of osteoporosis. J Bone Miner Metab 34(3):347–353
Dawson-Hughes B, Looker AC, Tosteson ANA, Johansson H, Kanis JA, Melton LJ (2012) The potential impact of the National Osteoporosis Foundation guidance on treatment eligibility in the USA: an update in NHANES 2005-2008. Osteoporos Int 23(3):811–820
Cosman F, de Beur SJ, LeBoff M, Lewiecki EM, Tanner B, Randall S, Lindsay R, National Osteoporosis Foundation (2014) Clinician's guide to prevention and treatment of osteoporosis. Osteoporos Int 25(10):2359–2381
The Centers for Disease Control and Prevention (2006) Laboratory Procedure Manual. [cited 2006 10/03]; Available from: https://www.cdc.gov/nchs/data/nhanes/nhanes_03_04/l31pah_c_met.pdf
The Centers for Disease Control and Prevention Laboratory Procedure Manual. Available from: https://www.cdc.gov/Nchs/Data/Nhanes/Nhanes_13_14/PAH_H_MET_Aromatic_Hydrocarbons.pdf
Charles J.. Rothwell, J.H.M., Kathryn S.Porter. (2013) National Health and Nutrition Examination Survey: Estimation Procedures, 2007–2010. [cited 2013 August]; Available from: https://wwwn.cdc.gov/nchs/data/series/sr02_159.pdf
Hu H, Kan H, Kearney GD, Xu X (2015) Associations between exposure to polycyclic aromatic hydrocarbons and glucose homeostasis as well as metabolic syndrome in nondiabetic adults. Sci Total Environ 505:56–64
Xu X, Hu H, Kearney GD, Kan H, Sheps DS (2013) Studying the effects of polycyclic aromatic hydrocarbons on peripheral arterial disease in the United States. Sci Total Environ 461-462:341–347
Acknowledgments
My deepest gratitude goes first and foremost to Professor Jia Chongqi for his constant encouragement and guidance. Second, we thank the National Center for Health Statistics (NCHS) for providing the datasets of National Health and Nutrition Examination Survey (NHANES). Third, I would like to express my heartfelt gratitude to my fellow classmates, who give me help in modifying the paper. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Duan, W., Meng, X., Sun, Y. et al. Association between polycyclic aromatic hydrocarbons and osteoporosis: data from NHANES, 2005–2014. Arch Osteoporos 13, 112 (2018). https://doi.org/10.1007/s11657-018-0527-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-018-0527-4