Abstract
Objective
To investigate whether Leech-Centipede (LC) Granules can improve erectile function in rats with diabetes mellitus-associated erectile dysfunction (DMED) through endothelial-to-mesenchymal transition (EndMT) inhibition.
Methods
Components of LC Granules were identified via ultra-high-performance liquid chromatography. Thirty male Sprague Dawley rats were injected with streptozotocin and fed continuously for 8 weeks to establish the DMED rat model. Rats with erectile dysfunction symptoms diagnosed using apomorphine were divided into DMED and low-, medium-, and high-doses LC groups (n=6 in each). The negative control (NC, n=6) and DMED groups were given 5 mL of deionized water via intragastric gavage, and the low-, medium- and the high-doses LC groups were administered LC at 1.6, 3.2, and 6.4 g/kg, respectively, via intragastric gavage for 4 weeks. The intracavernous pressure (ICP), mean arterial pressure (MAP), and nitric oxide (NO) levels in cavernous tissue were measured for each group. Quantitative reverse transcription-polymerase chain reaction and Western blot were used to detect mRNA and protein expressions of endothelial and mesenchymal markers. Immunofluorescence staining was used to observe α-SMA, and Masson’s trichrome staining was performed to determine the myofiber/collagen ratio.
Results
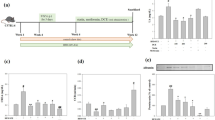
A total of 474 active components were identified. After treatment, the ICP/MAP value and NO level were significantly higher in the medium- and high-dose LC groups than in the DMED group (P<0.05). Compared with the DMED groups, the medium- and high-dose groups LC significantly increased and decreased endothelial and mesenchymal markers expression, respectively (P<0.05). Tumor growth factor (TGF)β R II, p-Smad2, and p-Smad3 levels were considerably higher following diabetes onset but reduced following LC intervention (P<0.05), except for TGF β 1 (P>0.05). α-SMA expression was significantly higher in the DMED group and was reduced in all LC intervention groups (P>0.05). The myofiber/collagen ratio in the LC groups was higher than that in the DMED group but lower than that in the NC group (all P<0.05).
Conclusions
LC Granules may improve the erectile function of DMED rats by suppressing TGF-β/Smad pathway to reverse EndMT.
Similar content being viewed by others
References
Malavige LS, Levy JC. Erectile dysfunction in diabetes mellitus. J Sex Med 2009;6:1232–1247.
Goldstein I, Lue TF, Padma-Nathan H, Rosen RC, Steers WD, et al. Oral sildenafil in the treatment of erectile dysfunction. New Eng J Med 1998;338:1397–1404.
Spivak L, Shultz T, Appel B, Verze P, Yagudaev D, Vinarov A. Low-intensity extracorporeal shockwave therapy for erectile dysfunction in diabetic patients. Sex Med Rev 2021;9:619–627.
El-Sakka AI. Efficacy of sildenafil citrate in treating erectile dysfunction: effect of type 2 diabetes. Eur Urol 2004;46:503–509.
Zhang H, Tong WT, Zhang CR, Li JL, Meng H, Yang HG, et al. Gross saponin of Tribulus Terrestris improves erectile dysfunction in type 2 diabetic rats by repairing the endothelial function of the penile corpus cavernosum. Diabetes Metab Synd Ob 2019;12:1705–1716.
Zheng X, Peng M, Li Y, Wang X, Lu W, Wang X, et al. Cathelicidin-related antimicrobial peptide protects against cardiac fibrosis in diabetic mice heart by regulating endothelial-mesenchymal transition. Int J Biol Sci 2019;15:2393–2407.
Thomas AA, Biswas S, Feng B, Chen S, Gonder J. lncRNA H19 prevents endothelial-mesenchymal transition in diabetic retinopathy. Diabetologia 2019;62:517–530.
Guan GY, Wei N, Song T, Zhao C, Sun Y, Pan RX, et al. miR-448-3p alleviates diabetic vascular dysfunction by inhibiting endothelial-mesenchymal transition through DPP-4 dysregulation. J Cell Physiol 2020;235:10024–10036.
Yu W, Liu Z, An S, Zhao J, Xiao L, Gou Y, et al. The endothelial-mesenchymal transition (EndMT) and tissue regeneration. Curr Stem Cell Res T 2014;9:196–204.
Wang JS, Li X, Chen ZL, Feng JL, Bao BH, Deng S, et al. Effect of leech-centipede medicine on improving erectile function in diabetes-induced erectile dysfunction rats via PDE5 signaling pathway-related molecules. Pharm Biol 2021;59:167–174.
Wang JS, Li X, Chen ZL, Feng JL, Bao BH, Deng S, et al. Effect of leech-centipede medicine on improving erectile function in DIED rats via PKC signaling pathway-related molecules. J Ethnopharmacol 2021;267:113463.
Ma JX, Wang B, Ding CF, Li HS, Jiang XJ, Wang CY, et al. Couplet medicines of leech and centipede granules improve erectile dysfunction via inactivation of the CaSR/PLC/PKC signaling in streptozotocin-induced diabetic rats. Bioscience Rep 2020;40;1–14.
Ma JX, Wang B, Li HS, Yu J, Hu HM, Ding CF, et al. Uncovering the mechanisms of leech and centipede granules in treating diabetes mellitus-induced erectile dysfunction utilizing network pharmacology. J Ethnopharmacol 2021;265:113358.
Saeedi P, Petersohn I, Salpea P, Malanda B, Karuranga S, Unwin N, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas. Diabetes Res Clin Pr 2019;157:107843.
Giuliano FA, Leriche A, Jaudinot EO, de Gendre AS. Prevalence of erectile dysfunction among 7689 patients with diabetes, hypertension, or both. Urology 2004;64:1196–1201.
Xu Y, Zhang Y, Yang Y, Liu L, Chen Y, Liu X. Prevalence and correlates of erectile dysfunction in type 2 diabetic men: a population-based cross-sectional study in Chinese men. Int J Impot Res 2019;31:9–14.
Lue TF. Erectile dysfunction. New Engl J Med 2000;342:1802–1813.
Piera-Velazquez S, Li Z, Jimenez SA. Role of endothelial-mesenchymal transition (EndoMT) in the pathogenesis of fibrotic disorders. Am J Pathol 2011;179:1074–1080.
Gressner OA, Rizk MS, Kovalenko E, Weiskirchen R, Gressner AM. Changing the pathogenetic roadmap of liver fibrosis? Where did it start; where will it go? J Gastroen Hepatol 2008;23:1024–1035.
Bi WR, Yang CQ, Shi Q. Transforming growth factor-β 1 induced epithelial-mesenchymal transition in hepatic fibrosis. Hepato-gastroenterology 2012;59:1960–1963.
Shi K, Wang F, Xia J, Zuo B, Wang Z, Cao X. Pirfenidone inhibits epidural scar fibroblast proliferation and differentiation by regulating TGF-β1-induced Smad-dependent and-independent pathways. Am J Translat Rec 2009;11:1593–1604.
Shi Y, Massagué J. Mechanisms of TGF-β signaling from cell membrane to the nucleus. Cell 2003;113:685–700.
Degryse AL, Tanjore H, Xu XC, Polosukhin VV, Jones BR, Boomershine CS, et al. TGFβ signaling in lung epithelium regulates bleomycin-induced alveolar injury and fibroblast recruitment. Am J Physiol Lung C 2011;300:L887–L897.
Liu X, Zhang H, Gao L, Yin Y, Pan X, Li Z, et al. Negative interplay of retinoic acid and TGF-β signaling mediated by TG-interacting factor to modulate mouse embryonic palate mesenchymal-cell proliferation. Birth Defects Res B 2014;101:403–409.
Baglieri J, Brenner DA, Kisseleva T. The role of fibrosis and liver-associated fibroblasts in the pathogenesis of hepatocellular carcinoma. Int J Mol Sci 2019;20:1723.
Tang PM, Zhang YY, Lan HY. LncRNAs in TGF-β-driven tissue fibrosis. Noncoding RNA 2018;4:26.
Jiao K, Zhen J, Wu M, Teng M, Yang K, Zhou Q, et al. 27-Hydroxycholesterol-induced EndMT acts via STAT3 signaling to promote breast cancer cell migration by altering the tumor microenvironment. Cancer Biol Med 2020;17:88.
Yin Q, He M, Huang L, Zhang X, Zhan J, Hu J. lncRNA ZFAS1 promotes ox-LDL induced EndMT through miR-150-5p/Notch3 signaling axis. Microvasc Res 2021;134:104118:1–7.
Author information
Authors and Affiliations
Contributions
Zhang H and Feng CH contributed equally to this work. Liu H conceived and designed the experiments; Zhang H, Feng CH, He S, Deng MX and Meng H performed the experiments; Feng CH and Zhang H contributed to the statistical data analysis; Zhang H and Feng CH wrote the manuscript. All authors have read and approved the manuscript for publication.
Corresponding author
Ethics declarations
The authors declare that they have no competing interests.
Additional information
Supported by the Scientific Research Project of the Traditional Chinese Medicine Bureau of Guangdong Province, China (Nos. 20211136 and 20221086)
Supplementary material
11655_2022_3728_MOESM1_ESM.pdf
Leech-centipede Granules Suppress EndMT to Improve Erectile Dysfunction in Rats with Diabetes Mellitus via TGF-β/Smad Pathway
Rights and permissions
About this article
Cite this article
Zhang, H., Feng, Ch., He, S. et al. Leech-Centipede Granules Suppress EndMT to Improve Erectile Dysfunction in Rats with Diabetes Mellitus via TGF-β/Smad Pathway. Chin. J. Integr. Med. 29, 28–36 (2023). https://doi.org/10.1007/s11655-022-3728-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-022-3728-z