Abstract
Objective
To evaluate the effect and safety of low-dose of apatinib and S-1 combined with Jianpi Bushen Jiedu Decoction (JBJD) in patients with metastatic colorectal cancer (mCRC) who have failed second or above lines treatment, in order to provide more treatment option for mCRC patients by integrated medicine.
Methods
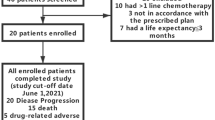
Thirteen patients were selected from a single-arm, open-label clinical study from April 2019 to September 2020. The patients were treated with low-dose apatinib (250 mg, once a day) and S-1 (20 mg, twice a day) combined with JBJD for at least one cycle and were followed up to August 2021. The primary endpoint was disease progression-free survival (PFS). Disease control rate (DCR), objective response rate (ORR), and overall survival (OS) of patients were observed as the secondary endpoints. Adverse events were recorded as well.
Results
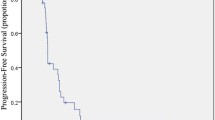
The average age of the 13 patients was 56.5 ±13.0 years and 76.9% were male. The median PFS and median OS were 4.6 and 8.3 months, respectively. The ORR was 7.7% (1/13) while the DCR was 61.5% (8/13). The common adverse events were hypertension, proteinuria, elevated transaminase, and thrombocytopenia. One patient experienced thrombocytopenia of grade 3.
Conclusions
Patients with mCRC after failure of the second or above lines of treatment may potentially benefit from the treatment of low-dose apatinib and S-1 combined with JBJD because of its similar effect as the standard dose of target therapy and relatively better safety. (Registration No. ChiCTR1900022673)
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dekker E, Tanis PJ, Vleugels J, Kasi P, Wallace M. Colorectal cancer. Lancet 2019;394:1467–1480.
Zheng R, Sun K, Zhang S, Zeng H, Zou X, Chen R, et al. Report of cancer epidemiology in China, 2015. China J Oncol (Chin) 2019;41:19–28.
Feng R, Zong Y, Cao S, Xu R. Current cancer situation in China: good or bad news from the 2018 Global Cancer Statistics? Cancer Commun 2019;39:12–22.
Chen H, Zheng R, Wang L, Lv Z, Du L, Wei W, et al. Progress in cancer epidemiology research in China in 2019. Chin J Dis Control Prev (Chin) 2020;24:373–379.
Chen WQ, Li N, Lan P, Chen HD, Du LB, Sun F, et al. Expert consensus on national early diagnosis and early treatment of colorectal cancer. Nat Med J China (Chin) 2020;100:1691–1698.
Brouwer NPM, van der Kruijssen DEW, Hugen N, de Hingh IHJT, Nagtegaal ID, Verhoeven RHA, et al. The impact of primary tumor location in synchronous metastatic colorectal cancer: differences in metastatic sites and survival. Ann Surg Oncol 2020;27:1580–1588.
Cheng L, Eng C, Nieman LZ, Kapadia AS, Du XL. Trends in colorectal cancer incidence by anatomic site and disease stage in the United States from 1976 to 2005. Am J Clin Oncol 2011;34:573–580.
Shao C, Zuo Q, Lin J, Yu RJ, Fu Y, Xiao M, et al. Effect of Chinese herbal medicine on the survival of colorectal cancer patients with liver-limited metastases: a retrospective cohort study, 2008 to 2017. Integr Cancer Ther 2019;18:1871102119.
Li NN, Zhou JF, Zhao L, Ying HY, Jia N. Efficacy and safety of apatinib in treating advanced colorectal cancer. Acta Acad Med Sin (Chin) 2019;41:170–174.
Bao JM, Gao YM, Qiu HL, Zhou XM. Clinical trial of apatinib mesylate tablets combined with capecitabine tablets and oxaliplatin injection in the treatment of advanced colorectal cancer. Chin J Clin Pharm (Chin) 2019;35:1565–1567.
Gu JY, Ye JH, Zhao Q, Zhang ZW. Apatinib combined with chemotherapy for advanced colon cancer: one case report and literature review. J Modern Oncol (Chin) 2020;28:3596–3599.
Li N, Deng W, Zhang G, Du Y, Guo Y, Ma Y, et al. Low-dose apatinib combined with S-1 in refractory metastatic colorectal cancer: a phase 2, multicenter, single-arm, prospective study. Front Oncol 2021;11:728854.
Dai YH, Sun L, Huang TT, Qiu H. Clinical analysis of third-line combination therapy with S-1 plus apatinib for advanced colorectal cancer. Chin J Clin Oncol (Chin) 2019;46:945–948.
Zhang T, Yang YF, He B, Yi DH, Hao J, Zhang D. Efficacy and safety of Quxie Capsule in metastatic colorectal cancer: a double-blind randomized placebo controlled trial. Chin J Integr Med 2018;24:171–177.
Zhang T, He WT, Zi MJ, Song G, Yi DH, Yang YF. Cohort study on prognosis of patients with metastatic colorectal cancer treated with integrated Chinese and Western medicine. Chin J Integr Med 2018;24:573–578.
Miao WR, Liu NN, Zhu HR, Li Q. Traditional Chinese medicine combined with chemotherapy in treatment of advanced colorectal cancer: a systematic review. Chin Arch Tradit Chin Med (Chin) 2018;36:1119–1124.
Jiang HJ, He B, Zhang T, Yang YF. Yang Yufei’s experience in the treatment of advanced colorectal cancer. Guid J Tradit Chin Med Pharm (Chin) 2020;26:187–189.
Hospital Administration, National Health Commision of the People’s Republic of China; Oncology Branch of Chinese Medical Association. Chinese protocol of diagnosis and ctreatment of colorectal cancer (2017 ed). Chin J Pract Surg (Chin) 2018;38:1089–1103.
Benson AB, Venook AP, Al-Hawary MM, Arain MA, Chen YJ, Ciombor KK, et al. Colon cancer, Version 2.2021, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw 2021;19:329–359.
Dong CX, Ding YW, Weng SS, Li CC, Huang YQ, Hu HG, et al. Update in version 2021 of CSCO guidelines for colorectal cancer from version 2020. Chin J Cancer Res (Chin) 2021;33:302–307.
Li J, Qin S, Xu R, Yau TC, Ma B, Pan H, et al. Regorafenib plus best supportive care versus placebo plus best supportive care in Asian patients with previously treated metastatic colorectal cancer (CONCUR): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 2015;16:619–629.
Li J, Qin S, Xu RH, Shen L, Xu J, Bai Y, et al. Effect of fruquintinib vs placebo on overall survival in patients with previously treated metastatic colorectal cancer: the FRESCO randomized clinical trial. JAMA 2018;319:2486–2496.
Chi Y, Shu Y, Ba Y, Bai Y, Qin B, Wang X, et al. Anlotinib monotherapy for refractory metastatic colorectal cancer: a double-blinded, placebo-controlled, randomized phase III trial (ALTER0703). Oncologist 2021;26:e1693–e1703.
Cheng Y, Du FC, Fang FQ, Duan ZJ, Lei W, Shi KG. Third-line treatment for metastatic colorectal cancer: anlotinib is superior to chemotherapy and similar to fruquintinib or regorafenib. Neoplasma 2020;67:1384–1390.
Wang F, Yuan X, Jia J, Bi X, Zhou Z, Zhou Q, et al. Apatinib monotherapy for chemotherapy-refractory metastatic colorectal cancer: a multi-centre, single-arm, prospective study. Sci Rep 2020;10:6058.
Chen X, Qiu T, Zhu Y, Sun J, Li P, Wang B, et al. A single-arm, phase II study of apatinib in refractory metastatic colorectal cancer. Oncologist 2019;24:407–883.
Dai LL, Chen DM, Zhou SM, Zhao N, Si WT, Cao Y, et al. Two-stage three-step method” assists in the prevention and treatment of adverse reactions of colorectal cancer chemotherapy. J Tradit Chin Med (Chin) 2019;60:982–985.
Xu L, Huang SP. Effect of the ginsenoside Rb1 on the spontaneous contraction of intestinal smooth muscle in mice. World J Gastroenterol 2012;18:5462–5469.
Li X, Lu X, Fan D, Li L, Lu C, Tan Y, et al. Synergistic effects of Erzhi Pill combined with methotrexate on osteoblasts mediated via the Wnt1/LRP5/β-catenin signaling pathway in collagen-induced arthritis rats. Front Pharmacol 2020;11:228.
Gao J, Wang T, Wang C, Wang S, Wang W, Ma D, et al. Effects of Tianshu Capsule on spontaneously hypertensive rats as revealed by1H-NMR-based metabolic profiling. Front Pharmacol 2019;10:989.
Tian Z, Zhang S, Wang H, Wang S, Wang W, Ma D, et al. Intervention of uncaria and its components on liver lipid metabolism in spontaneously hypertensive rats. Front Pharmacol 2020;11:910.
Pei Y, Wang R, Zhou H. Network pharmacology mechanism in treating hypertension with apocynum venetum. Central South Pharm (Chin) 2019;17:1997–2001.
Pu F, Chen F, Lin S, Chen S, Zhang Z, Wang B, et al. The synergistic anticancer effect of cisplatin combined with Oldenlandia diffusa in osteosarcoma MG-63 cell line in vitro. OncoTargets Ther 2016;9:255–263.
Wu Y, Xu J, Liu Y, Zeng Y, Wu G. A review on anti-tumor mechanisms of coumarins. Front Oncol 2020;10:592853.
Author information
Authors and Affiliations
Contributions
Chen Y wrote the manuscript and was involved in the extraction of data, the statistical analysis of data and the follow-up of the patients. This project was initiated and developed by Yang YF, Xu YY and Jiang HJ. Yang YF was involved in the design of the study and manuscript revision and review. Xu YY and Jiang HJ were involved in the design of the study, literature research and the follow-up of the patients. Wang L was responsible for the follow-up of the patients. Zhai JW was responsible for the follow-up of the patients and data checking. Zhang T was involved in manuscript revision and review. All authors read and approved the final manuscripts.
Corresponding author
Additional information
Conflict of Interest
The authors declare that there are no conflicts of interest regarding the publication of this paper.
Supported by National Administration of Traditional Chinese Medicine: Qihuang Scholar (No. 02045004) and Collaborative Pilot Project of Clinical Traditional Chinese and Western Medicine for Major and Difficult Diseases in 2019 (No. 070030003)
Supplementary material
11655_2022_3676_MOESM1_ESM.pdf
Patients with Metastatic Colorectal Cancer after Failure of Second-Line Treatment May Benefit from Low-Dose Apatinib and S-1 Combined with Jianpi Bushen Jiedu Decoction
Rights and permissions
About this article
Cite this article
Chen, Y., Xu, Yy., Jiang, Hj. et al. Patients with Metastatic Colorectal Cancer after Failure of Second-Line Treatment May Benefit from Low-Dose Apatinib and S-1 Combined with Jianpi Bushen Jiedu Decoction. Chin. J. Integr. Med. 28, 924–929 (2022). https://doi.org/10.1007/s11655-022-3676-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-022-3676-7