Abstract
Objective
To determine whether topical applications of thiosulfinate-enriched Allium sativum extract (TASE) can accelerate acute cutaneous wound healing (WH) in a murine model.
Methods
Keratinocyte viability and in vitro wound closure were assessed in keratinocyte cultures. Effects of topical TASE (0.5 μg/mL of allicin in 97% ethanol) on acute cutaneous WH were determined in a murine model of acute cutaneous wound. Twelve mice were alternately assigned to the vehicle- and TASE-treated groups (n=6 per group). Expression levels of mRNA for keratinocyte differentiation marker-related proteins (filaggrin, loricrin and involucrin) and lipid synthetic enzymes (elongation of very long chain fatty acids protein 4 (ELOVL4), fatty acid synthase (FA2H), 3-hydroxy- 3-methyl-glutaryl-coenzyme A reductase (HMGCoA), and serine palmitoyltransferase (SPT)) were assessed using real-time quantitative polymerase chain reaction on day 3 and 8 after wounding, while transepidermal water loss (TEWL) rates were measured in wounded areas.
Results
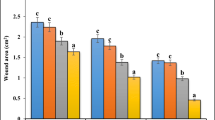
TASE accelerated WH both in vivo (40% vs. 22% reduction in wound area, P<0.01) and in vitro (90% vs. 65% reduction in wound area, P<0.01). Moreover, topical applications of TASE upregulated the expression levels of epidermal mRNA for ELOVL4, HMGCoA, SPT, filaggrin, loricrin and involucrin (P<0.05 vs. vehicle-treated controls) on day 3 after wounding. Likewise, TASE significantly lowered TEWL rates in comparison with vehicle alone on day 8 (33.06±2.09 g/(m2·h) vs. 24.60±2.04 g/(m2·h), P<0.01).
Conclusions
Topical applications of TASE stimulated keratinocyte proliferation and formation of epidermal permeability barrier function, leading to acceleration of acute cutaneous WH. Topical products containing TASE could be used to manage acute cutaneous WH.
Similar content being viewed by others
References
Guo S Dipietro LA. Factors affecting wound healing. J Dent Res 2010;89:219–229.
Jhamb S Vangaveti VN Malabu UH. Genetic and molecular basis of diabetic foot ulcers: clinical review. J Tissue Viability 2016;25:229–236.
Wilson JA Clark JJ. Obesity: impediment to postsurgical wound healing. Adv Skin Wound Care 2004;17:426–435.
Delmore B Cohen JM O’Neill D Chu A Pham V Chiu E. Reducing postsurgical wound complications: a critical review. Adv Skin Wound Care 2017;30:272–286.
Han G Ceilley R. Chronic wound healing: a review of current management and treatments. Adv Ther 2017;34:599–610.
Vermeulen H Ubbink D Goossens A de Vos R Legemate D. Dressings and topical agents for surgical wounds healing by secondary intention. Cochrane Database Syst Rev 2004:CD003554.
Sivamani RK Ma BR Wehrli LN Maverakis E. Phytochemicals and Naturally derived substances for wound healing. Adv Wound Care 2012;1:213–217.
Hu F Yan Y Wang CW Liu Y Wang JJ Zhou F, et al. Effect and mechanism of Ganoderma lucidum polysaccharides on human fibroblasts and skin wound healing in mice. Chin J Integr Med 2019;25:203–209.
Kawanami H Kawahata H Mori HM Aoki M. Moxibustion promotes formation of granulation in wound healing process through induction of transforming growth factor-ß in rats. Chin J Integr Med 2020;26:26–32.
Tattelman E. Health effects of garlic. Am Fam Phys 2005;72:103–106.
Pasteur L. Memoire sur la fermentation appelee lactique. Mem Soc Imp Sci Agric Lille 1858;5:13–26.
Harris JC Cottrell SL Plummer S Lloyd D. Antimicrobial properties of Allium sativum (garlic). Appl Microbiol Biotechnol 2001;57:282–286.
Li T Shi HY Hua YX Gao C Xia Q Yang G, et al. Effects of allicin on the proliferation and cell cycle of chondrocytes. Int J Clin Exp Pathol 2015;8:12525–12532.
Fujisawa H Suma K Origuchi K Kumagai H Seki T Ariga T. Biological and chemical stability of garlic-derived allicin. J Agric Food Chem 2008;56:4229–35.
Livak KJ Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001;25:402–408.
Man G Mauro TM Zhai Y Kim PL Cheung C Hupe M, et al. Topical hesperidin enhances epidermal function in an aged murine model. J Invest Dermatol 2015;135:1184–1187.
Sorg H Tilkorn DJ Hager S Hauser J Mirastschijski U. Skin wound healing: an update on the current knowledge and concepts. Eur Surg Res 2017;58:81–94.
Kasuya A Tokura Y. Attempts to accelerate wound healing. J Dermatol Sci 2014;76:169–172.
Kunii T Hirao T Kikuchi K Tagami H. Stratum corneum lipid profile and maturation pattern of corneocytes in the outermost layer of fresh scars: the presence of immature corneocytes plays a much more important role in the barrier dysfunction than do changes in intercellular lipids. Br J Dermatol 2003;149:749–756.
Suetake T Sasai S Zhen YX Ohi T Tagami H. Functional analyses of the stratum corneum in scars. Sequential studies after injury and comparison among keloids, hypertrophic scars, and atrophic scars. Arch Dermatol 1996;132:1453–1458.
Feingold KR Elias PM. Role of lipids in the formation and maintenance of the cutaneous permeability barrier. Biochim Biophys Acta 2014;1841:280–294.
Schmuth M Jiang YJ Dubrac S Elias PM Feingold KR. Thematic review series: skin lipids. Peroxisome proliferatoractivated receptors and liver X receptors in epidermal biology. J Lipid Res 2008;49:499–509.
Salazar H Llorente I Jara-Oseguera A García-Villegas R Munari M Gordon SE. A single N-terminal cysteine in TRPV1 determines activation by pungent compounds from onion and garlic. Nat Neurosci 2008;11:255–261.
Himi N Hamaguchi A Hashimoto K Koga T Narita K Miyamoto O. Calcium influx through the TRPV1 channel of endothelial cells (ECs) correlates with a stronger adhesion between monocytes and ECs. Adv Med Sci 2012;57:224–229.
Wood LC Elias PM Sequeira-Martin SM Grunfeld C Feingold KR. Occlusion lowers cytokine mRNA levels in essential fatty acid-deficient and normal mouse epidermis, but not after acute barrier disruption. J Invest Dermatol 1994;103:834–838.
Harris IR Farrell AM Grunfeld C Holleran WM Elias PM Feingold KR. Permeability barrier disruption coordinately regulates mRNA levels for key enzymes of cholesterol, fatty acid, and ceramide synthesis in the epidermis. J Invest Dermatol 1997;109:783–787.
Acknowledgment
Authors are grateful to Dr. Lucia Gonzalez-Lopez from the Pathology Service (Hospital General Universitario de Ciudad Real, Ciudad Real, Spain) for her expert advice and kind support. We also thank David Gallego-Molinero, Amelia Gonzalez-Lopez and Clara Villar-Rodriguez for their technical support (Translational Research Unit, Hospital General Universitario de Ciudad Real, Ciudad Real, Spain).
Author information
Authors and Affiliations
Contributions
JLS, EMG-M, MQM and JMP-O designed the experiments, conducted data analyses, and wrote the manuscript; PME critically reviewed the manuscript; JLS, FJR-C and JMP-O performed animal experiments; EMG-M did the in vitro studies; JRM-R and MAC-M performed molecular experiments and analyzed data; IG-F prepared and provided the lyophilized Allium sativum extract for both in vivo and in vitro studies.
Corresponding authors
Ethics declarations
All authors, except IG-F, have no conflicts of interest. IG-F is part of the patent authors [WO 2008/102036 A1. Method for obtaining a freeze-dried, stable extract from plants of the Allium genus] from which the lyophilized Allium sativum extract was obtained in a standardized way. This content is solely the responsibility of the authors and does not necessarily represent the official views of either the funding agencies or the US Department of Veterans Affairs.
Additional information
Supported by the European Commission/FSE Funds to EMG-M, the European Regional Development Fund (JRM-R and JMP-O, Castilla-La Mancha FEDER 2014-20 PO), the Government of Castilla-La Mancha (MAC-M, Ref. II-2016-06), and the National Institute of Arthritis, Musculoskeletal and Skin Diseases of the National Institutes of Health (PEM & MQM, AR061106), with additional resources provided by The Veterans Affairs Medical Center, San Francisco, CA, USA
Electronic supplementary material
11655_2020_3086_MOESM1_ESM.pdf
Appendix 1. Composition of Organic and Inorganic Compounds of Lyophilized Allium sativum Extract from Las Pedroñeras (Ciudad Real, Spain) under Optimized Conditions (WO 2008/102036 A1)
11655_2020_3086_MOESM5_ESM.pdf
Appendix 5. Expression Levels of mRNAs Following 8-Day Treatments with Thiosulfinate-Enriched Allium sativum Extract or Vehicle
Rights and permissions
About this article
Cite this article
Santiago, J.L., Galan-Moya, E.M., Muñoz-Rodriguez, J.R. et al. Topical Applications of Thiosulfinate-Enriched Allium sativum Extract Accelerates Acute Cutaneous Wound Healing in Murine Model. Chin. J. Integr. Med. 26, 812–818 (2020). https://doi.org/10.1007/s11655-020-3086-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-020-3086-7