Abstract
Objective
To investigate the mechanistic basis for the attenuation of bone degeneration by edible bird’s nest (EBN) in ovariectomized rats.
Methods
Forty-two female Sprage-Dawley rats were randomized into 7 groups (6 in each group). The ovariectomized (OVX) and OVX + 6%, 3%, and 1.5% EBN and OVX +estrogen groups were given standard rat chow alone, standard rat chow +6%, 3%, and 1.5% EBN, or standard rat chow +estrogen therapy (0.2mg/kg per day), respectively. The sham-operation group was surgically opened without removing the ovaries. The control group did not have any surgical intervention. After 12 weeks of intervention, blood samples were taken for serum estrogen, osteocalcin, and osteoprotegerin, as well as the measurement of magnesium, calcium abd zinc concentrations. While femurs were removed from the surrounding muscles to measure bone mass density using the X-ray edge detection technique, then collected for histology and estrogen receptor (ER) immunohistochemistry.
Results
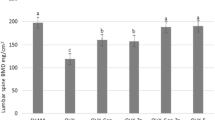
Ovariectomy altered serum estrogen levels resulting in increased food intake and weight gain, while estrogen and EBN supplementation attenuated these changes. Ovariectomy also reduced bone ER expression and density, and the production of osteopcalcin and osteorotegerin, which are important pro-osteoplastic hormones that promote bone mineraliztion and density. Conversely, estrogen and EBN increased serum estrogen levels leading to increased bone ER expression, pro-osteoplastic hormone production and bone density (all P<0.05).
Conclusion
EBN could be used as a safe alternative to hormone replacement therapys for managing menopausal complications like bone degeneration.
Similar content being viewed by others
References
Buckler H. The menopause transition: endocrine changes and clinical symptoms. J Br Menopause Soc 2005;11:61–65.
Wise PM. Estrogens and neuroprotection. Trend Endocrinol Metab 2002;13:229–230.
Pavel OR, Popescu M, Novac L, Mogoantă L, Pavel LP, Vicaş RM, et al. Postmenopausal osteoporosis-clinical, biological and histopathological aspects. Rom J Morphol Embryol 2016;57:121–130.
Gameiro CM, Romão F, Castelo-Branco C. Menopause and aging: changes in the immune system—a review. Maturitas 2010;67:316–320.
Martín-Millán M, Castañeda S. Estrogens, osteoarthritis and inflammation. Joint Bone Spine 2013;80:368–373.
Johnston BD, Ward WE. The ovariectomized rat as a model for studying alveolar bone loss in postmenopausal women. Bio Med Res Int 2015;2015:635023.
Hickey M, Elliott J, Davison SL. Hormone replacement therapy. BMJ 2012;344:e763.
Marcone MF. Characterization of the edible bird’s nest the “Caviar of the East”. Food Res Int 2005;38:1125–1134.
Yida Z, Imam MU, Ismail M, Hou Z, Abdullah MA, Ideris A, et al. Edible bird’s nest attenuates high fat diet-induced oxidative stress and inflammation via regulation of hepatic antioxidant and inflammatory genes. BMC Complement Alternat Med 2015;15:310. doi: 10.1186/s12906-015-0843-9.
Hu Q, Li G, Yao H, He S, Li H, Liu S, et al. Edible bird’s nest enhances antioxidant capacity and increases lifespan in Drosophila Melanogaster. Cell Mol Biol (Noisy-le-grand) 2016;62:116–122.
Hou ZP, Imam MU, Ismail M, Ismail N, Yida Z, Ideris A, et al. Effects of edible bird’s nest on hippocampal and cortical neurodegeneration in ovariectomized rats. Food Funct 2015;6:1701–1711.
Matsukawa N, Matsumoto M, Bukawa W, Chiji H, Nakayama K, Hara H, et al. Improvement of bone strength and dermal thickness due to dietary edible bird’s nest extract in ovariectomized rats. Biosci Biotechnol Biochem 2011;75:590–592.
Imam MU, Ishaka A, Der Juin O, Zamri ND M, Sarega N, Ismail M, et al. Germinated brown rice regulates hepatic cholesterol metabolism and cardiovascular disease risk in hypercholesterolemic rats. J Funct Foods 2014;8:193–203.
Muhammad SI, Ismail M, Mahmud RB, Salisu AM, Zakaria ZA. Germinated brown rice and its bioactives modulate the activity of uterine cells in oophorectomised rats as evidenced by gross cytohistological and immunohistochemical changes. BMC Complement Alternat Med 2013;13:198.
Muhammad SI, Maznah I, Mahmud RB, Esmaile MF, Zuki ABZ. Bone mass density estimation: Archimede’s principle versus automatic X-ray histogram and edge detection technique in ovariectomized rats treated with germinated brown rice bioactives. Clin Interv Aging 2013;8:1421–1431.
Jaji AZ, Zuki ZAB, Mahmud R, Loqman MY, Hezmee MNM, Abba Y, et al. Safety assessments of subcutaneous doses of aragonite calcium carbonate nanocrystals in rats. J Nanopart Res 2017;19:175. doi: 10.1007/s11051-017-3849-z. Epub 2017 May 11.
Files JA, Ko MG, Pruthi S. Bioidentical hormone therapy. Mayo Clin Proc 2011;86:673–680.
Baker ME. What are the physiological estrogens? Steroids 2013;78:337–340.
Miller KK, Al-Rayyan N, Ivanova MM, Mattingly KA, Ripp SL, Klinge CM, et al. DHEA metabolites activate estrogen receptors alpha and beta. Steroids 2013;78:15–25.
Bhavnani BR, Nisker JA, Martin J, Aletebi F, Watson L, Milne JK. Comparison of pharmacokinetics of a conjugated equine estrogen preparation (premarin) and a synthetic mixture of estrogens (C.E.S.) in postmenopausal women. J Soc Gynecol Invest 2000;7:175–183.
Davis SR, Castelo-Branco C, Chedraui P, Lumsden MA, Nappi RE, Shah D, et al. Writing Group of the International Menopause Society for World Menopause Day 2012. Understanding weight gain at menopause. Climacteric 2012;15:419–429.
Teede HJ, Lombard C, Deeks AA. Obesity, metabolic complications and the menopause: an opportunity for prevention. Climacteric 2010;13:203–209.
Lee NK, Sowa H, Hinoi E, Ferron M, Ahn JD, Confavreux C, et al. Endocrine regulation of energy metabolism by the skeleton. Cell 2007;130:456–469.
Bharadwaj S, Naidu AG, Betageri GV, Prasadarao NV, Naidu AS. Milk ribonuclease-enriched lactoferrin induces positive effects on bone turnover markers in postmenopausal women. Osteoporos Int 2009;20:1603–1611.
Bateman TA, Countryman S. Osteoprotegerin and bone loss associated with spaceflight. Drug Discov Today 2002;7:456–457.
Pelekanou V, Kampa M, Kiagiadaki F, Deli A, Theodoropoulos P, Agrogiannis G, et al. Estrogen anti-inflammatory activity on human monocytes is mediated through cross-talk between estrogen receptor ERa36 and GPR30/GPER1. J Leukocyte Biol 2016;99:333–347.
Bord S, Ireland DC, Beavan SR, Compston JE. The effects of estrogen on osteoprotegerin, RANKL, and estrogen receptor expression in human osteoblasts. Bone 2003;32:136–141.
Author information
Authors and Affiliations
Contributions
Hou ZP conceived the idea for this study and carried out the experiment, and together with He PY and Ismail M drafted the mamuscript. Ji HR and Du MN helped cultured rats. Tang SY and Li YH participated in its design and coordination. Dong XL and He WJ helped to calculate bone density. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
The authors declare that they have no competing interests.
Additional information
Supported by Grants from Ministry of Science, Technology and Innovation, E-science Fund (No. 5450666), Malaysia; the Health and Family Planning Commission of Hebei (No. 20160315); the Administration of Traditional Chinese Medicine of Hebei (No. 2017196); the Key Discipline Construction Project of Hebei Provincial Universities [No. JiJiaoGao-2013-(4)-2012-37]; and Start up Fund for High Level Talents of Chengde Medical University (No. 201704)
Supplementary material
11655_2019_3209_MOESM1_ESM.pdf
Edible Bird’s Nest Attenuates Menopause-Related Bone Degeneration in Rats via Increasing BoneEstrogen-Receptor Expression
Rights and permissions
About this article
Cite this article
Hou, Zp., Tang, Sy., Ji, Hr. et al. Edible Bird’s Nest Attenuates Menopause-Related Bone Degeneration in Rats via Increaing Bone Estrogen-Receptor Expression. Chin. J. Integr. Med. 27, 280–285 (2021). https://doi.org/10.1007/s11655-019-3209-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-019-3209-1