Abstract
Objective
To investigate the effects of sodium copper chlorophyllin (SCC) on the proliferation, differentiation and immunomodulatory function of mesenchymal stem cells (MSCs) from mice with aplastic anemia.
Methods
A mouse model of aplastic anemia was established by exposure of BALB/c mice to sublethal doses of 5.0 Gy Co60 γ radiation, followed by transplantation of 2×106 lymph node cells from DBA/2 donor mice within 4 h after radiation. Aplastic anemic BALB/c mice were randomly divided into six groups: the treated groups, which received 25, 50, or 100 mg/kg/day SCC, respectively; a positive control group treated with cyclosporine A (CsA); and an untreated model control group (model group); while, the non-irradiated mice as the normal control group. SCC or CsA were administered by gastrogavage for 20 days, starting on day 4 after irradiation. Peripheral blood cells were counted and colony-forming fibroblasts (CFU-F) in the bone marrow were assayed. The ability of MSCs to form calcium nodes after culture in osteoinductive medium was also observed. The immunosuppressive effect of MSCs on T lymphocytes was analyzed by enzyme-linked immunosorbent assay and flow cytometry, to evaluate the efficacy of SCC in mice with aplastic anemia.
Results
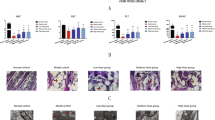
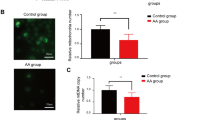
Peripheral blood white cell and platelet counts were increased by medium and high SCC doses, compared with the untreated control. CFU-Fs were also increased compared with the untreated control, and the numbers of calcium nodes in MSCs in osteoinductive medium were elevated in response to SCC treatment. The percentage of Forkhead box protein 3 (FOXP3+) T cells was increased in T cell-MSC cocultures, and the cytokine transforming growth factor β1 was up-regulated in SCC-treated groups.
Conclusion
The results of this study suggest that SCC not only promotes the proliferation and differentiation of MSCs, but also improves their immunoregulatory capacity in mice with aplastic anemia.
Similar content being viewed by others
References
Sarraf CE, Otto WR, Eastwood M. In vitro mesenchymal stem cell differentiation after mechanical stimulation. Cell Prolif 2011;44:99–108.
Bunnell BA, Betancourt AM, Sullivan DE. New concepts on the immune modulation mediated by mesenchymal stem cells. Stem Cell Res Ther 2010;1:34–39.
Li JP, Zheng CL, Han ZC. Abnormal immunity and stem/ progenitor cells in acquired aplastic anemia. Crit Rev Onco Hematol 2010;75:79–93.
Shipounova IN, Petrova TV, Svinareva DA, Momotuk KS, Mikhailova EA, Drize NI. Alterations in hematopoietic microenvironment in patients with aplastic anemia. Clin Transi Sci 2009;2:67–74.
Chao YH, Pent CT, Harn HJ, Chan CK, Wu KH. Poor potential of proliferation and differentiation in bone marrow mesenchymal stem cells derived from children with severe aplastic anemia. ANN Hematol 2010;89:715–723.
Gao RL, Yin LM, Qian XD, Lin XJ, Chen XH, Wang Xiao. The potential alteration of their osteogenesis and adipocyte differentiation in bone marrow mesenchymal stem cells from mice with immune-mediated aplastic anemia. J Zhejiang Univ Tradit Chin Med (Chin) 2009:652–654.
De Toni F, Poglio S, Youcef AB, Cousin B, Plfumio F, Bourin P, et al. Human adipose-derived stromal cells efficiently support hematopoiesis in vitro and in vivo: a key step for therapeutic studies. Stem Cells Dev 2011;20:2127–2138.
Park BW, Kang EJ, Byun JH, Son MG, Kim HJ, Hah YS, et al. In vitro and in vivo osteogenesis of human mesenchymal stem cells derived from skin, bone marrow and dental follicle tissues. Differentiation 2012;83:249–259.
Svobodova E, Krulova M, Zajicova A, Pokorna K, Prochazkova J, Trosan P, et al. The role of mouse mesenchymal stem cells in differentiation of naive T-cells into anti-inflammatory regulatory T-cell or proinflammatory helper T-cell 17 population. Stem Cells Dev 2012;21:901–910.
Walenda T, Bokermannn G, Ventura Ferreira MS, Piroth DM, Hieronymus T, Neuss S, et al. Synergistic effects of growth factors and mesenchymal stromal cells for expansion of hematopoietic stem and progenitor cells. Exp Hematol 2011;39:617–628.
Li N, Feuqier P, Serrurrier B, Latqer-Cannard V, Lesesve JF, Stoltz JF, et al. Human mesenchymal stem cells improve ex vivo expansion of adult human CD34+ peripheral blood progenitor cells and decrease their allostimulatory capacity. Exp Hematol 2007;35:507–515
Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005;105:1815–1822.
Ryan JM, Barry F, Murphy JM, Mahon BP. Interferon-gamma does not break, but promotes the immunosuppressive capacity of adult human mesenchymal stem cells. Clin Exp Immunol 2007;149:353–363.
Gorelik L, Flavell RA. Transforming growth factor beta in T-cell biology. Nat Rev Immunol 2002;2:46–53.
Ghannam S, Pene J, Torcy-Moquet G, Jorqensen C, Yssel H. Mesenchymal stem cells inhibit human Th17 cell differentiation and function and induce a T regulatory cell phenotype. J Immunol 2010;185:302–312.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by Zhejiang Provincial Natural Science Foundation of China (No. Y207728 and No. Y2080036) and Traditional Chinese Medicine Administration Bureau of Zhejiang Province, China (No. 2008YA005)
Rights and permissions
About this article
Cite this article
Yin, Lm., Jiang, Hf., Wang, X. et al. Effects of sodium copper chlorophyllin on mesenchymal stem cell function in aplastic anemia mice. Chin. J. Integr. Med. 19, 360–366 (2013). https://doi.org/10.1007/s11655-012-1210-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-012-1210-z