Abstract
Objective
To assess the inhibitory modulation of blood pressure by stimulation of the deep peroneal nerve (DPN) and to determine the involvement of nociceptive fibers in the modulation.
Methods
All the animals were divided into six groups (A-F). The rats in groups A and B received no pretreatment. The rats in groups C and D received subcutaneous injection of capsaicin or control vehicle, respectively, near the DPN for 2 days. Those in groups E and F had the DPN exposed to capsaicin or control vehicle, respectively, for 20 min. Subsequently, pressor responses were induced by stimulation of paraventricular nucleus (PVN) either electrically (groups A and C C-F) or chemically via injection of glutamate (group B). After two stable pressor responses (baseline), all groups were subject to 5-min DPN stimulation followed by PVN stimulation for 10 s. Arterial blood pressure, heart rate, and electrocardiogram were recorded. The pressor response was calculated as the difference in the mean arterial pressure (MAP) before and after PVN stimulation, and changes from baseline in pressor response after DPN stimulation were compared between the groups.
Results
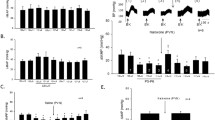
Increases of MAP of 22.88±2.18 mm Hg and 20.32±5.25 mm Hg were induced by electrical (group A) or chemical (group B) stimulation of the PVN, respectively. These pressor responses were inhibited by stimulation of the DPN, and the MAP was reduced to 12.00±2.10 mm Hg in group A (n=6, P<0.01) and 7.00±2.85 mm Hg in group B (n=6, P<0.01). Subcutaneous injection of capsaicin (125 mg/kg) near the DPN in group C (n=7) had no effect on the inhibitory effect of DPN stimulation compared with the group D (n=9), and neither did blockade of nociceptive fibers with capsaicin in group E (n=6) compared with group F (n=8).
Conclusion
Stimulation of the DPN mimicking acupuncture has an inhibitory effect on the pressor response, and the effect is mediated by capsaicin-insensitive afferent fibers in the DPN.
Similar content being viewed by others
References
Lee HJ, Lee B, Choi SH, Hahm DH, Kim MR, Roh PU, et al. Electroacupuncture reduces stress-induced expression of c-fos in the brain of the rat. Am J Chin Med 2004;32:795–806.
Tjen ALSC, Li P, Longhurst JC. Medullary substrate and differential cardiovascular responses during stimulation of specific acupoints. Am J Physiol Regul Integr Comp Physiol 2004;287:R852–R862.
Li P, Rowshan K, Crisostomo M, Tjen ALSC, Longhurst JC. Effect of electroacupuncture on pressor reflex during gastric distension. Am J Physiol Regul Integr Comp Physiol 2002;283:R1335–R1345.
Wu HC, Lin JG, Chu CH, Chang YH, Chang CG, Hsieh CL, et al. The effects of acupuncture on cardiac muscle cells and blood pressure in spontaneous hypertensive rats. Acupunct Electrother Res 2004;29:83–95.
Kurono Y, Egawa M, Yano T, Shimoo K. The effect of acupuncture on the coronary arteries as evaluated by coronary angiography: a preliminary report. Am J Chin Med 2002;30:387–396.
Zhu WJ, Zhang RB. Spinal mechanism of the inhibitory effect of somatic input on the cardiac ischemia induced by hypothalamus stimulation. Acta Physiol Sin 1991;43:141–148.
Zhang LJ, Zhang RB. The role of dorsomedial hypothalamic nucleus in the cardiovascular responses induced by habenular nucleus stimulation and inhibitory effect of deep peroneal nerve. Chin J Appl Physiol 2002;18:230–233.
Zhou SY, Zhu YP, Gao F. The regulation of deep peroneal nerve inputs on cardiovascular activity caused by area postrema excitation. Chin J Appl Physiol 2003;19:7–11.
Zhou S, Zhang R, Luo J. The regulation of deep peroneal nerve inputs on depressor response evoked by caudal ventrolateral medulla excitation in rabbits. Acupunct Res 1995;20:12–17.
Swanson LW, Sawchenko PE. Paraventricular nucleus: a site for the integration of neuroendocrine and autonomic mechanisms. Neuroendocrinology 1980;31:410–417.
Schramm LP, Strack AM, Platt KB, Loewy AD. Peripheral and central pathways regulating the kidney: a study using pseudorabies virus. Brain Res 1993;616:251–262.
Hubscher CH, Kaddumi EG, Johnson RD. Brain stem convergence of pelvic viscerosomatic inputs via spinal and vagal afferents. Neuroreport 2004;15:1299–1302.
Uchida Y, Nishigori A, Takeda D, Ohshiro M, Ueda Y, Ohshima M, et al. Electroacupuncture induces the expression of Fos in rat dorsal horn via capsaicininsensitive afferents. Brain Res 2003;978:136–140.
Tjen ALSC, Fu LW, Zhou W, Syuu Z, Longhurst JC. Role of unmyelinated fibers in electroacupuncture cardiovascular responses. Auton Neurosci 2005;118:43–50.
Fitzgerald M. Capsaicin and sensory neurones—a review. Pain 1983;15:109–130.
Nagy JI, van der Kooy D. Effects of neonatal capsaicin treatment on nociceptive thresholds in the rat. J Neurosci 1983;3:1145–1150.
Jancso G, Kiraly E, Jancso-Gabor A. Pharmacologically induced selective degeneration of chemosensitive primary sensory neurones. Nature 1977;270:741–743.
Paxinos G, Watson, C. The rat brain in stereotaxic coordinates. 4th Ed. San Diego. Sydney Academic Press; 1998: Figure 24–26.
Kannan H, Hayashida Y, Yamashita H. Increase in sympathetic outflow by paraventricular nucleus stimulation in awake rats. Am J Physiol 1989;256:R1325–R1330.
Li YF, Mayhan WG, Patel KP. NMDA-mediated increase in renal sympathetic nerve discharge within the PVN: role of nitric oxide. Am J Physiol Heart Circ Physiol 2001;281:H2328–H2336.
Chen QH, Haywood JR, Toney GM. Sympathoexcitation by PVN-injected bicuculline requires activation of excitatory amino acid receptors. Hypertension 2003;42:725–731.
Martin DS, Egland MC, Barnes LU, Vogel EM. Adrenergic nerves mediate the venoconstrictor response to PVN stimulation. Brain Res 2006;1076:93–100.
Schlenker E, Barnes L, Hansen S, Martin D. Cardiorespiratory and metabolic responses to injection of bicuculline into the hypothalamic paraventricular nucleus (PVN) of conscious rats. Brain Res 2001;895:33–40.
Zhang K, Patel KP. Effect of nitric oxide within the paraventricular nucleus on renal sympathetic nerve discharge: role of GABA. Am J Physiol 1998;275:R728–R734.
Backer M, Hammes MG, Valet M, Deppe M, Conrad B, Tolle TR, et al. Different modes of manual acupuncture stimulation differentially modulate cerebral blood flow velocity, arterial blood pressure and heart rate in human subjects. Neurosci Lett 2002;333:203–206.
Friedemann T, Li W, Wang Z. Inhibitory regulation of blood pressure by manual acupuncture in the anesthetized rat. Auton Neurosci 2009;151:178–182.
Ohsawa H, Okada K, Nishijo K, Sato Y. Neural mechanism of depressor responses of arterial pressure elicited by acupuncture-like stimulation to a hindlimb in anesthetized rats. J Auton Nerv Syst 1995;51:27–35.
Reed WR, Chadha HK, Hubscher CH. Effects of 17beta-estradiol on responses of viscerosomatic convergent thalamic neurons in the ovariectomized female rat. J Neurophysiol 2009;102:1062–1074.
Strigo IA, Duncan GH, Boivin M, Bushnell MC. Differentiation of visceral and cutaneous pain in the human brain. J Neurophysiol 2003;89:3294–3303.
Cianchetti C. Capsaicin jelly against migraine pain. Int J Clin Pract 2010;64:457–459.
Barkin RL, Barkin SJ, Irving GA, Gordon A. Management of chronic noncancer pain in depressed patients. Postgrad Med 2011;123:143–154.
Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 1997;389:816–824.
Geppetti P, Nassini R, Materazzi S, Benemei S. The concept of neurogenic inflammation. BJU Int 2008;101(Suppl) 3:2–6.
Kissin I. Vanilloid-induced conduction analgesia: selective, dose-dependent, long-lasting, with a low level of potential neurotoxicity. Anesth Analg 2008;107:271–281.
Furuta A, Kita M, Suzuki Y, Egawa S, Chancellor MB, de Groat WC, et al. Association of overactive bladder and stress urinary incontinence in rats with pudendal nerve ligation injury. Am J Physiol Regul Integr Comp Physiol 2008;294:R1510–R1516.
Györfi A, Fazekas A, Fehér E, Ender F, Rosivall L. Effects of streptozotocin-induced diabetes on neurogenic inflammation of gingivomucosal tissue in rat. J Periodontal Res 1996;31:249–255.
McDougall JJ. Involvement of sympathetic efferents but not capsaicin-sensitive afferents in nociceptin-mediated dual control of rat synovial blood flow. Am J Physiol Regul Integr Comp Physiol 2003;284:R1477–R1485.
Xiao YF, Li P. Effect of deep peroneal nerve stimulation on renal nerve discharge under different levels of blood pressure in rabbits. Acta Physiol Sin 1983;35:432–439.
Lu GW. Characteristics of afferent fiber innervation on acupuncture points zusanli. Am J Physiol 1983;245:R606–R612.
Maggi CA, Conte B. Effect of urethane anesthesia on the micturition reflex in capsaicin-treated rats. J Auton Nerv Syst 1990;30:247–251.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No.30870834), the Zhejiang Medical and Health Science Research Foundation (No. 2008A042), and the Zhejiang Provincial Natural Science Foundation of China (No. Y2110057 and No. Y2090820)
Rights and permissions
About this article
Cite this article
Sun, X., Lan, Qq., Cai, Y. et al. Electrical stimulation of deep peroneal nerve mimicking acupuncture inhibits the pressor response via capsaicin-insensitive afferents in anesthetized rats. Chin. J. Integr. Med. 18, 130–136 (2012). https://doi.org/10.1007/s11655-012-0991-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-012-0991-4