Abstract
Objective
To evaluate the safety and efficacy of composite sophora colon-soluble capsule (CSCC) in treating ulcerative colitis (UC) of the damp-heat accumulation syndrome pattern (DHAS) and to prepare a basis for a phase III clinical trial.
Methods
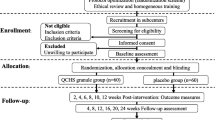
A multi-center, randomized, single-blind, and positive drug parallel-controlled design was adopted. There were 126 patients of UC-DHAS stratified and assigned equally to three groups. Patients in two CSCC treated groups, Groups T1 and T2, were treated orally with high (six capsules, thrice a day) and low (four capsules, thrice a day) doses CSCC, and patients in the control group were treated orally with Mesalazine Enteric-coated Tablets (four tablets, thrice a day), respectively, all for eight weeks. The clinical efficacy and safety of treatments were evaluated through clinical symptom observations and colonoscopic examinations.
Results
(1) Full analysis set (FAS) and per-protocol set (PPS) analyses showed the comprehensive curative effect in Groups T1, T2, and the control group, obtaining the values of 85.7%, 92.9%, and 71.4% (P=0.330), and 89.5%, 92.7%, and 73.2% (P=0.552), respectively, demonstrating no statistical significance among the three groups. (2) FAS and PPS analysis showed the efficacy on membranous lesions in Groups T1, T2, and the control group, obtaining the values of 83.3%, 92.9%, and 73.8% (P=0.063), and 86.8%, 92.7%, and 75.6% (P=0.070), respectively, showing statistical insignificance among the three groups. (3) FAS analysis showed an efficacy tendency on improving tenesmus (P=0.056). No changes were found in improving the other symptoms, and statistical significance was not shown among the three groups (P>0.05). PPS analysis showed the efficacy on single item symptom in Groups T1, T2, and the control group was not statistically significant among the three groups (P=0.082).
Conclusions
The comprehensive effect of CSCC in treating UC is basically equivalent to that of Mesalazine enteric-coated tablet; however, the tendency was shown to improve symptoms. Its efficacy could not be raised by increasing the dosage used. Therefore, the recommended dosage of CSCC is four capsules, three times a day.
Similar content being viewed by others
References
Zheng ZT, ed. Gastroenterology. 3 rd ed. Beijng: People’s Medical Publishing House; 2000:775–789.
Tong ZQ, Wu ZJ, Zhao ML, Yang B, Chen BY. Clinical study on Sophora colonsoluble capsules for the treatment of ulcerative colitis. Chin J Inf Tradit Chin Med (Chin) 2009;16(9):67–68.
Chinese Society of Digestive Diseases, Chinese Medical Association. Suggestion on the norm for diagnosis and treatment of inflammatory bowel diseases. Chin J Dig (Chin) 2001;21:236–239.
Zheng XY, ed. Guiding principle of clinical research on new drugs of traditional Chinese medicine. Beijng: China Medical Science and Technology Press;2002:129–134.
Zhou LL, Fang TH, Yang SM, Liang T, Xu L, Xu L, et al. Protective effect of Composite Sophora colon-soluble capsules on inflammatory colitis in rats. Shaanxi J Tradit Chin Med (Chin) 2004;25:89–91.
Zhou LL, Fang TH, Yang SM, Si JF, Liang T, Xu L. Protecting effect of Compound Radix Sophorae Flavescentis capsule on ulcerative colitis of guinea pigs caused by chlorodinitrobenzene. J Nanjing Univ Tradit Chin Med (Chin) 2004;20:107–109.
Quan W, Lee HJ, Kim CY, Noh CW, Um BH, Oak MH, et al. Anti-allergic prenylated flavonoids from the roots of Sophora flavescens. Planta Med 2008;74:168–170.
Hong MH, Lee JY, Jung H, Jin DH, Go HY, Kim JH, et al. Sophora flavescens Aiton inhibits the production of pro-inflammatory cytokines through inhibition of the NF kappaB/IkappaB signal pathway in human mast cell line (HMC-1). Toxicol Vitro 2009,23:251–258.
Liu F, Liu J, Chen X, Wand QJ, Li K. Analgesic action of oxymatrine and its mechanism. J Jilin Univ (Med Edition, Chin) 2005;31:883–885.
Ren HZ, He YM, Yang L. Research progress in the chemical constituents and pharmacological activities of Rhizoma Bletillae. Asia-Pacific Tradit Med (Chin) 2009;5:134–140.
Wang HY. Experimental study on Baiji manna polysaccharide’s anti-gastric ulcer anti-inflammation and abirritation. J Zhejiang Coll Tradit Chin Med (Chin) 2009;33:119–121.
Luo Y, Wang H, Yuan Z. Triterpenoid saponins of Sanguisorba officinalis and their anti-inflammatory activity. Chin J Med Chem (Chin) 2008;18:138–141.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by Items of Creative Research and Development of Drugs, Beijing Municipal Committee of Sciences (No. D08080200290805)
Rights and permissions
About this article
Cite this article
Tong, Zq., Yang, B., Chen, By. et al. A multi-center, randomized, single-blind, controlled clinical study on the efficacy of composite sophora colon-soluble capsules in treating ulcerative colitis. Chin. J. Integr. Med. 16, 486–492 (2010). https://doi.org/10.1007/s11655-010-0562-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-010-0562-5