Abstract
Objective
To evaluate the effect of Jianpi Huoxue decoction (健脾活血方, JHD)-containing serum on tumor necrosis factor-α (TNF-α) secretion and endotoxin receptor gene expression in RAW264.7 cells induced by lipopolysaccharide (LPS).
Methods
The cytotoxicity of blank-control serum and JHD-containing serum at different concentrations were evaluated through the lactate dehydrogenase (LDH) assay in RAW264.7 cells. RAW264.7 cells were divided into six groups: 5% blank-control serum group (C1, n=3), 5% blank-control serum plus LPS group (L1, n=4), 5% JHD-containing serum plus LPS group (J1, n=4), 10% blank-control serum group (C2, n=3), 10% blank-control serum plus LPS group (L2, n=4), and 10% JHD-containing serum plus LPS group (J2, n=4). After cultured with the corresponding serum for 1 h, cells in L1, L2, J1 and J2 were treated with LPS (0.1 μ g/mL) for 12 h without rinse. The supernate, cells, protein and RNA were collected for assay. TNF-α in the culture supernate was assayed by the enzyme linked immunosorbent assay (ELISA). Protein expression of TNF-α in RAW cells was detected by Western-blot. TNF-α, Toll-like receptor 2 (TLR2), TLR4 and CD14 mRNA expression in RAW cells were detected by real-time RT-PCR.
Results
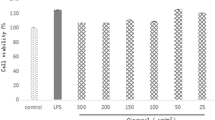
The LDH assay supported that cultured for 24 h or less with the JHD-containing serum at the concentration of 10% or lower, RAW264.7 cells showed no cytotoxicity. After stimulation with LPS for 2 h, TNF-α in the culture supernate of the 5% blank-control serum plus LPS group (L1, P=0.03), 10% blank-control serum plus LPS group (L2, P=0.002) and in the cell layer (P=0.01) of these groups increased remarkably. After stimulation with LPS for 1 h, the mRNA expression of TNF-α (P=0.004), TLR (P=0.03), CD14 (P=0.004) was up-regulated obviously. In the 10% JHD-containing serum plus LPS group (J2), the protein expression of TNF-α in both supernate (P=0.04) and cell layer (P=0.04), gene expression of TNF-α (P=0.03), TLR4 (P=0.001), CD14 (P=0.001) were all inhibited. On the other hand, the TLR2 mRNA expression was not up-regulated after LPS stimulation in the 10% blank-control serum plus LPS group (L2).
Conclusion
JHD-containing serum inhibited the LPS-induced cytokines expression in RAW264.7 which was probably associated with its inhibitory effect on the mRNA expression of LPS receptors TLR and CD14.
Similar content being viewed by others
References
Nanji AA. Role of Kupffer cells in alcoholic hepatitis. Alcohol 2002;27:13–15.
Lorenz E. TLR2 and TLR expression during bacterial infections. Curr Pharm Des 2006; 12: 4185–4193.
Kirschning CJ, Wesche H, Merrill Ayres T, Rothe M. Human toll-like receptor 2 confers responsiveness to bacterial lipopolysaccharide. J Exp Med 1998; 188:2091–2097.
Heine H, Kirschning CJ, Lien E, Monks BG, Rothe M, Golenbock DT. Cutting edge: cells that carry A null allele for toll-like receptor 2 are capable of responding to endotoxin. J Immunol 1999; 162: 6971–6975.
Dziarski R, Wang Q, Miyake K, Kirschning CJ, Gupta D. MD-2 enables Toll-like receptor 2 (TLR2)-mediated responses to lipopolysaccharide and enhances TLR2-mediated responses to Gram-positive and Gram-negative bacteria and their cell wall components. J Immunol 2001;166:1938–1944.
Liu S, Salyapongse AN, Geller DA, Vodovotz Y, Billiar TR. Hepatocyte toll-like receptor 2 expression in vivo and in vitro: role of cytokines in induction of rat TLR2 gene expression by lipopolysaccharide. Shock 2000;20:915–921.
Matsumura T, Ito A, Takii T, Hayashi H, Onozaki K. Endotoxin and cytokine regulation of toll-like receptor (TLR) 2 and TLR gene expression in murine liver and hepatocytes. J Interferon Cytokine Res 2000;20:915–921.
Frost RA, Nystrom G, Burrows PV, Lang CH. Temporal differences in the ability of ethanol to modulate endotoxin-induced increases in inflammatory cytokines in muscle under in vivo conditions. Alcohol Clin Exp Res 2005;29:1247–1256.
Cubero FJ, Nieto N. Kupffer cells and alcoholic liver disease. Rev Esp Enferm Dig 2006;98:460–472.
Rao RK, Seth A, Sheth P. Recent advances in alcoholic liver disease I. Role of intestinal permeability and endotoxemia in alcoholic liver disease. Am J Physiol Gastrointest Liver Physiol 2004;286:G881–G884.
Hanck C, Rossol S, Bocker U, Tokus M, Singer MV. Presence of plasma endotoxin is correlated with tumour necrosis factor receptor levels and disease activity in alcoholic cirrhosis. Alcohol Alcohol 1998;33:606–608.
Su GL. Lipopolysaccharides in liver injury: molecular mechanisms of Kupffer cell activation. Am J Physiol Gastrointest Liver Physiol 2002;283:G256–G265.
Hu YY, Fang ZH, Cui JW, et al. Relationship between alcoholic liver injury and gut permeability and the alleviating effect of Chinese herbs. Sci Technol Engineering 2005;5:1770–1771,1776.
Fang ZH, Hu YY, Cui JW, et al. Relationship between alcoholic liver injury and endotoxin leakage from gut and intervention effect of Jianpi Liqi Huoxue decoction. Chin J Integr Tradit West Med (Chin) 2006;26(9):813–817.
Peng JH, Hu YY, Cheng Y, et al. Effect of Jianpi Huoxue decoction on inflammatory cytokine secretion pathway in rat liver with lipopolysaccharide challenge. World J Gastroenterol 2008;14(12):1851–1857.
Cheng Y, Ping J, Xu LM. Effects of curcumin on peroxisome proliferator-activated receptor gamma expression and nuclear translocation/redistribution in culture-activated rat hepatic stellate cells. Chin Med J 2007;120:794–801.
Hansen J, Cherwitz DL, Allen JI. The role of tumor necrosis factor-alpha in acute endotoxin-induced hepatotoxicity in ethanol-fed rats. Hepatology 1994;20:461–474.
Thurman RG. II. Alcoholic liver injury involves activation of Kupffer cells by endotoxin. Am J Physiol 1998;275(4 Part 1):G605–G611.
Adachi Y, Moore LE, Bradford BU, Gao W, Thurman RG. Antibiotics prevent liver injury in rats following long-term exposure to ethanol. Gastroenterology 1995;108:218–224.
Uesugi T, Froh M, Arteel GE, Bradford BU, Thurman RG. Toll like receptor is involved in the mechanism of early alcohol-induced liver injury in mice. Hepatology 2001;34:101–108.
Triantafilou K, Triantafilou M, Dedrick RL. Interactions of bacterial lipopolysaccharide and peptidoglycan with a 70 kDa and an 80 kDa protein on the cell surface of CD14+ and CD14-cells. Hum Immunol 2001;62:50–63.
Akira S, Takeda K, Kaisho T. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat Immunol 2001;2:675–680.
Takeuchi O, Hoshino K, Kawai T, Sanjo H, Takada H, Ogawa T, et al. Differential roles of TLR2 and TLR in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity 1999;11:443–451.
Hirschfeld M, Ma Y, Weis JH, Vogel SN, Weis JJ. Cutting edge: repurification of lipopolysaccharide eliminates signaling through both human and murine toll-like receptor 2. J Immunol 2000;165:618–622.
Liu Y, Wang Y, Yamakuchi M, Isowaki S, Nagata E, Kanmura Y, et al. Upregulation of toll-like receptor 2 gene expression in macrophage response to peptidoglycan and high concentration of lipopolysaccharide is involved in NF-κ B activation. Infect Immun 2001;69:2788–2796.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by National Natural Science Foundation of China (No. 30371818); Shanghai Leading Academic Discipline Project (Y0302); Shanghai Educational Development Foundation, (No. 2007CG56); and Shanghai Rising-Star Program (No. 07QA14052)
Rights and permissions
About this article
Cite this article
Peng, Jh., Hu, Yy., Feng, Q. et al. Effect of Jianpi Huoxue decoction (健脾活血方)-containing serum on tumor necrosis factor-α secretion and gene Expression of endotoxin receptors in RAW264.7 cells induced by lipopolysaccharide. Chin. J. Integr. Med. 15, 198–203 (2009). https://doi.org/10.1007/s11655-009-9001-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-009-9001-x