Abstract
Objective
To evaluate the efficacy of kushenin in treating patients with chronic hepatitis C after renal transplantation.
Methods
Fifty-five patients were randomly assigned by lottery to the treatment group (29 cases) and control group (26 cases). The same immunosuppression therapy was given to all patients in both groups. Patients in the treatment group were treated with kushenin 0.6 g once a day, while those in the control group were treated with conventional liver protective agents such as vitamins. The treatment duration of both groups was 3 months. The incidences of serious hepatitis and acute rejection reaction, serum biochemistry parameters including indicators of liver and kidney functions, hepatic fibrosis index, and serum HCV-RNA were compared between the two groups.
Results
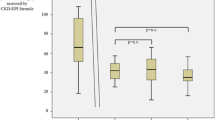
(1) The incidence of serious hepatitis in the treatment group and the control group was 3.45% (1/29 cases) and 11.54% (3/26 cases), respectively, which was insignificantly different between the two groups (P=0.335). (2) The incidence of acute rejection in the treatment group was 6.90% (2/29 cases) and that in the control group was 7.69% (2/26 cases), showing insignificant difference (P=0.335). (3) The differences in serum alanine aminotransferase (ALT), direct bilirubin (DBIL), hyaluronic acid (HA), propeptide collagen type III (PC III), laminin (LN), collagen type IV (Col IV) levels between the two groups were insignificant before transplantation (P>0.05), while the above-mentioned parameters in the treatment group were significantly lower than those in the control group after transplantation (P<0.05). The difference in serum creatinine (SCr) and endogenous creatinine clearance rate (CCr) between the two groups was insignificant before and after transplantation (P>0.05). (4) The negative conversion rate of HCV-RNA in the treatment group was 31.03% (9/29 cases), significantly higher than the value of 11.54% (3/26 cases) in the control group after transplantation (P<0.05). (5) The levels of serum ALT and DBIL in patients with HCV-RNA converted to negative were significantly lower than those with still-positive HCV-RNA (P<0.05).
Conclusions
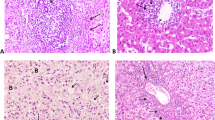
Kushenin has a certain effect on inhibiting the proliferation of HCV, protecting liver cells, and anti-liver fibrosis. On the other hand, it has no obvious influence on renal allograft function. Thus, the drug is clinically safe and effective for use in treating patients with chronic hepatitis C after renal transplantation.
Similar content being viewed by others
References
Li JQ, Li CQ, Zeng MD, et al. Preliminary stuty on therapeutic effect of oxymatrine in treating patients with chronic hepatitis C. Chin J Integr Tradit West Med (Chin) 1998;18(4):227–228.
Qi JY, Xie FD, Guo LS, et al. The status of hepatitis B and C virus infection in patients receiving maintenance hemodialysis after kidney transplantation. Chin J Nosocomiol (Chin) 2003;13(9):805–807.
Lin HH, Huang CC, Sheen IS, et al. Prevalence of antibodies to hepatitis C virus in the hemodialysis unit. Am J Nephrol 1991;11:192–194.
Fabrizi F, Martin P, Dixit V, et al. Hepatitis C virus antibody status and survival after renal transplantation: meta-analysis of observational studies. Am J Transplant 2005;5:1452–1461.
Chen YX, Li JQ, Zeng MD, et al. The inhibitory effect oxymatrine on hepatitis C virus in vitro. Chin J Hepatol (Chin) 2001;7(S1):12–14.
Wang JX, Wang GJ, Cai X, et al. Effect of oxymatrin and glycyrrhizin on hepatocyte apoptosis induced by cessation of phenobarbital treatment in mice. Acad J Sec Mil Med Univ (Chin) 1999;20(4):222–224.
Sun YN, Xu B, Huang ZQ, et al. Effect of matrine on levels of TGF-β1, TNF-α, HA and PcIII in sera of chronic hepatitis B patients. China J Modern Med (Chin) 2004;14(1):41–44.
Shang Z, Ding T, Wen FC, et al. Effect of matrine on immunological function in mice. J Changchun Univ Tradit Chin Med (Chin) 2007;23(2):21–22.
Society of Infection and Parasite, Society of Hepatopathy Chinese Medical Association. Prevention and cure project of virus hepatitis. Chin J Infect Dis (Chin) 2001;19(1):56–62.
Boletis J, Delladetsima J, Psimenou E, et al. Liver biopsy is essential in anti-HCV (+) renal transplant patients irrespective of liver function tests and serology for HCV. Transplant Proc 1995;27:945–947.
Wang XF, Li L, Han GZ. Progress of research on pharmacologic action of Matrine. Herald Med (Chin) 2005;6(6):483–484.
Sun HW. Clinical application of kushenin. Chin J Integr Tradit West Med (Chin) 1986;6(7):46–47.
Zhang Q, Zhao FS, Hao ZM, et al. Research on effect and mechanism of oxymatrine inhibiting mice abdominal labrocyte to release histamine. Chin J Microbiol Immunol (Chin) 1992;12(1):41–44.
Yu GQ, Zhou XH, Shi XH, et al. Preliminary study on therapeutic effect of oxymatrine in treating patients with chronic hepatitis C. Sichuan Med J (Chin) 2007;28(6):593–594.
Chen JY. Observation on protection effect of oxymatrine to experimental liver damage. Chin Pharm J (Chin) 1982;18(7):407–409.
Gan LW, Wang GJ, Li YL. Effects of oxymatrine on liver fibrosis in rats. J Sec Milit Med Univ (Chin) 1999;20(7):445–448.
Qing ZL, Zhuang HM, Liu Y, et al. Study on effect of oxymatrine on prolonging the survival time of cardiac tissue allograft in mice and its immunological mechanisms. Chin J Integr Tradit West Med (Chin) 1990;10(2):99–100.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supportted by National Natural Science Foundation of China (No. 30571799, 30471640)
Rights and permissions
About this article
Cite this article
Li, Y., Xue, Wj., Fan, P. et al. Application of kushenin on patients with chronic hepatitis C after renal transplantation. Chin. J. Integr. Med. 14, 167–172 (2008). https://doi.org/10.1007/s11655-008-0167-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-008-0167-4