Abstract
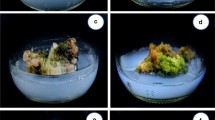
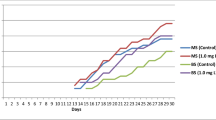
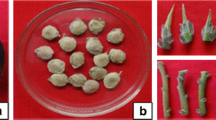
Catasetum integerrimum is an orchid used in Mexican traditional medicine, which is overexploited due to its ornamental and medicinal properties. In this scenario, conservation strategies are mandatory to protect wild populations from this valuable orchid. The aim of the present study was to establish a micropropagation protocol by direct organogenesis, as a tool for Catasetum integerrimum production and conservation. Seeds from a mature capsule were cultivated in a Murashige and Skoog basal medium. Then, 6-mo-old vitroplants were transplanted into treatments fortified with 6-bencilaminopurin (BAP) or indole-3-acetic acid (IAA) to induce plant development. Treatment fortified with 2.5 mg L−1 BAP in combination with 5.0 mg L−1 IAA developed 5.73 ± 0.45 shoots per explant and 5.84 ± 0.48 leaves per shoot. For the rooting process, the best treatment was 2.5 mg L−1 IAA, which developed 11.20 ± 0.28 roots with a 13.20 ± 0.28-cm root length. C. integerrimum vitroplants were used as a source of leaves, roots, and pseudobulbs for the induction of direct organogenesis. Leaf explants did not induce any morphogenic response. Pseudobulb explants had the best response when developing 1 shoot per explant with 5.50 ± 0.18 leaves per shoot and 4.37 ± 0.37 roots per explant with a 4.88 ± 0.20-cm root length. For the acclimatization stage, a survival rate of over 90% was achieved. The results obtained prove the efficacy of the micropropagation method, which contributes significantly to the conservation of this orchid, preserving its wild populations from indiscriminate collection.




Similar content being viewed by others
References
Alayón-Gamboa J (2011) Orquídeas en huertos familiares de Calakmul, Campeche, México. Comisión Nacional de Áreas Naturales Protegidas, Mexico.
Alcorn JB (1984) Huastec Mayan ethnobotany. University of Texas Press, USA
Altinkaynak H, Ozkoc I (2020) Isolation and molecular characterization of plant growth promoting bacteria from the rhizosphere of orchids in Turkey. Rhizosphere 16:100280. https://doi.org/10.1016/j.rhisph.2020.100280
Arda A (2020) Chitosan versus plant growth regulators: a comparative analysis of their effects on in vitro development of Serapias vomeracea (Burm. f.) Briq. Plant Cell Tissue Organ Cult 141:327–338. https://doi.org/10.1007/s11240-020-01789-3
Baker A, Kaviani B, Nematzadeh G, Negahdar N (2014) Micropropagation of Orchis Catasetum - a rare and endangered orchid. Acta Sci Pol 13:197–205
Bhowmik TK, Rahman MM (2020a) In vitro study of medicinally important orchid Aerides multiflora Roxb. from nodal and leaf explants. J Pharmacogn Phytochem 9:179–184
Bhowmik TK, Rahman MM (2020b) Micropropagation of commercially important orchid Dendrobium palpebrae Lindl. through in vitro developed pseudobulb culture. J Adv Biotechnol Exp Ther 3:225–232. https://doi.org/10.5455/jabet.2020.d128
Bielach A, Hrtyan M, Tognetti VB (2017) Plants under stress: involvement of auxin and cytokinin. Int J Mol Sci 18:1427. https://doi.org/10.3390/ijms18071427
Carranza-Álvarez C, Trinidad-García KL, Reyes-Hernández H, Castillo-Pérez LJ, Fortanelli-Martínez J (2020) Effect of natural extracts on the micropropagation of Vanilla planifolia Jacks. ex Andrews (Orchidaceae). Biotecnia 23:5–12. https://doi.org/10.18633/biotecnia.v23i1.805
Castillo-Pérez LJ, Alonso-Castro AJ, Fortanelli-Martínez J, Carranza-Álvarez C (2021a) Biotechnological approaches for conservation of medicinal plants. In: Bhat RA, Hakeem KR, Dervash MA (eds) Phytomedicine: a treasure of pharmacologically active products from plants. Academic Press, London, pp 35–58. https://doi.org/10.1016/B978-0-12-824109-7.00002-9
Castillo-Pérez LJ, Martínez-Soto D, Fortanelli-Martínez J, Carranza-Álvarez C (2021b) Asymbiotic seed germination, in vitro seedling development, and symbiotic acclimatization of the Mexican threatened orchid Stanhopea tigrina. Plant Cell Tissue Organ Cult 146:249–257. https://doi.org/10.1007/s11240-021-02064-9
Castillo-Pérez LJ, Martínez-Soto D, Maldonado-Miranda JJ, Alonso-Castro AJ, Carranza-Álvarez C (2019) The endemic orchids of Mexico: a review. Biologia 74:1–13. https://doi.org/10.2478/s11756-018-0147-x
Chase MW, Cameron KM, Freudenstein JV, Pridgeon AM, Salazar G, Van den Berg C, Schuiteman A (2015) An updated classification of Orchidaceae. Bot J Linn Soc 177:151–174. https://doi.org/10.1111/boj.12234
Cheruvathur MK, Abraham J, Mani B, Thomas TD (2010) Adventitious shoot induction from cultured internodal explants of Malaxis acuminata D. Don, a valuable terrestrial medicinal orchid. Plant Cell Tissue Organ Cult 101:163–170. https://doi.org/10.1007/s11240-010-9673-0
Chookoh N, Chiu YT, Chang C, Hu WH, Dai TE (2019) Micropropagation of Tolumnia orchids through induction of protocorm-like bodies from leaf segments. Hortscience, 54:1230–1236. https://doi.org/10.21273/HORTSCI13747-18
De Melo-Ferreira WD, Pereira-de Oliveira S, Mamoru-Suzuki R, Ferreira-Silva KL, Pereira-Soares WP (2018) Germination, growth and morpho-anatomical development of Catasetum macrocarpum (Orchidaceae) in vitro. Rodriguésia 69:2137–2151. https://doi.org/10.1590/2175-7860201869442
Deb CR, Arenmongla T (2014) Development of cost effective in vitro regeneration protocol of Malaxis acuminata D. Don a therapeutically important orchid using pseudobulbs as explant source. J Plant Stud 3:13.
Deepthi AS, Ray JG (2019) Applications of endophytic-fungal-isolates from velamen root of wild orchids in floriculture. Braz J Biol Sci 6:577–589. https://doi.org/10.21472/bjbs.061409
Faria DV, Simão MJ, Cipriano R, Werner ET, Soares TCB, Aoyama EM, Lima-Gontijo ABP (2018) In vitro morphogenesis and micropropagation of Aechmea ramosa var. ramosa Mart. ex Schult. f. (Bromeliaceae) from leaf explants. In Vitro Cell Dev Biol Plant 54:530–536. https://doi.org/10.1007/s11627-018-9907-0
Gantait S, Sinniah UR (2012) Rapid micropropagation of monopodial orchid hybrid (Aranda Wan Chark Kuan ‘Blue’× Vanda coerulea Grifft. ex. Lindl.) through direct induction of protocorm-like bodies from leaf segments. Plant Growth Regul 68:129–140. https://doi.org/10.1007/s10725-012-9698-y
Ghimire BK, Yu CY, Chung IM (2012) Direct shoot organogenesis and assessment of genetic stability in regenerants of Solanum aculeatissimum Jacq. Plant Cell Tissue Organ Cult 108:455–464. https://doi.org/10.1007/s11240-011-0057-x
Hnatuszko-Konka K, Kowalczyk T, Gerszberg A, Glińska S, Grzegorczyk-Karolak I (2019) Regeneration of Phaseolus vulgaris from epicotyls and hypocotyls via direct organogenesis. Sci Rep 9:1–12. https://doi.org/10.1038/s41598-019-42723-8
Hoang NH, Kane ME, Radcliffe EN, Zettler LW, Richardson LW (2017) Comparative seed germination and seedling development of the ghost orchid, Dendrophylax lindenii (Orchidaceae), and molecular identification of its mycorrhizal fungus from South Florida. Ann Bot 119:379–393. https://doi.org/10.1093/aob/mcw220
Huang Z, Li H (2020) Control of oxidative stress by a combination of PBU, BAP and DMTU enhances adventitious shoot formation in Eucalyptus urophylla. Plant Cell Tissue Organ Cult 141:533–541. https://doi.org/10.1007/s11240-020-01812-7
Islam SS, Islam T, Bhattacharjee B, Mondal TK, Subramaniam S (2015) In vitro pseudobulb based micropropagation for mass development of Cymbidium finlaysonianum Lindl. Emir J Food Agric 27:469–474. https://doi.org/10.9755/ejfa.2015.04.017
Jiang H, Chen MC, Lee YI (2017) In vitro germination and low-temperature seed storage of Cypripedium lentiginosum PJ Cribb & SC Chen, a rare and endangered lady’s slipper orchid. Sci Hort 225:471–479. https://doi.org/10.1016/j.scienta.2017.07.040
Jung WS, Chung IM, Kim SH, Chi HY, Yu CY, Ghimire BK (2021) Direct shoot organogenesis from Lycium chinense Miller leaf explants and assessment of genetic stability using ISSR markers. Agronomy 11:503. https://doi.org/10.3390/agronomy11030503
Kumar SS, Giridhar P (2021) In vitro micropropagation of Basella rubra L. through proliferation of axillary shoots. Plant Cell Tissue Organ Cult 144:477–483. https://doi.org/10.1007/s11240-020-01960-w
Kumar N, Reddy MP (2011) In vitro plant propagation: a review. J for Environ Sci 27:61–72
Mata-Rosas M, Baltazar-García RJ, Moon P, Hietz P, Luna-Monterrojo VE (2010) In vitro regeneration of Lycaste aromatica (Graham ex Hook) Lindl. (Orchidaceae) from pseudobulb sections. Plant Biotechnol Rep 4:157–163. https://doi.org/10.1007/s11816-010-0132-5
Moraes MC, Camolesi MR, Palmieri DA, Bertão MR (2020) Commercial fertilizers and organic additives in orchid micropropagation. Plant Cell Culture & Micropropagation 16:16-e162. https://doi.org/10.46526/pccm.2020.v16.162
Mose W, Indrianto A, Purwantoro A, Semiarti E (2017) The influence of thidiazuron on direct somatic embryo formation from various types of explant in Phalaenopsis amabilis (L.) blume orchid. Hayati J Biosci 24:201–205. https://doi.org/10.1016/j.hjb.2017.11.005
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Pant B, Thapa D (2012) In vitro mass propagation of an epiphytic orchid, Dendrobium primulinum Lindl. through shoot tip culture. Afr J Biotechnol 11:9970–9974. https://doi.org/10.5897/AJB11.3106
Paul P, Joshi M, Gurjar D, Shailajan S, Kumaria S (2017) In vitro organogenesis and estimation of β-sitosterol in Dendrobium fimbriatum Hook.: an orchid of biopharmaceutical importance. S Afr J Bot 113:248–252. https://doi.org/10.1016/j.sajb.2017.08.019
Peres LEP, Kerbauy GB (1999) High cytokinin accumulation following root tip excision changes the endogenous auxin-to-cytokinin ratio during root-to-shoot conversion in Catasetum fimbriatum Lindl (Orchidaceae). Plant Cell Rep 18:1002–1006. https://doi.org/10.1007/s002990050698
Safarpour M, Sinniah UR, Subramaniam S, Swamy MK (2017) A novel technique for Musa acuminata Colla ‘Grand Naine’(AAA) micropropagation through transverse sectioning of the shoot apex. In Vitro Cell Dev Biol Plant 53:226–238. https://doi.org/10.1007/s11627-017-9809-6
Salazar GA, Soto-Arenas MA, Hágsater E, Jiménez R, Aguirre-O I, Greenwood EW (1990) Icones Orchidacearum. Fascicle I. Orchids of Mexico. Herbario AMO, Mexico.
Sarkar J, Banerjee N (2020) Influence of different cytokinins on micropropagation of an important medicinal plant, Solanum erianthum Don, and assessment of the genetic fidelity of the regenerants. In Vitro Cell Dev Biol Plant 56:480–490. https://doi.org/10.1007/s11627-020-10054-3
Seon KM, Kim DH, Kang KW, Sivanesan I (2018) Highly competent in vitro propagation of Thrixspermum japonicum (Miq.) Rchb. f., a rare epiphytic orchid. In Vitro Cell Dev Biol Plant 54:302–308. https://doi.org/10.1007/s11627-018-9890-5
Sharma A, Zheng B (2019) Molecular responses during plant grafting and its regulation by auxins, cytokinins, and gibberellins. Biomolecules 9:397. https://doi.org/10.3390/biom9090397
Sharma V (2017) Regeneration competence of pseudobulb explants of endangered orchid genera: a study in vitro. Int J Recent Sci Res 8:21722–21724. https://doi.org/10.24327/ijrsr.2017.0811.1126
Sharma V (2021) Micropropagation of therapeutically important swarna Jibanti (Coelogyne cristata Lindl.) through pseudobulb segments-a study in vitro. Ecol Environ Conserv 27:S229–S234
Sherif NA, Benjamin JF, Kumar TS, Rao MV (2018) Somatic embryogenesis, acclimatization and genetic homogeneity assessment of regenerated plantlets of Anoectochilus elatus Lindl., an endangered terrestrial jewel orchid. Plant Cell Tissue Organ Cult 132:303–316. https://doi.org/10.1007/s11240-017-1330-4
Sidky R. (2017) Optimized direct organogenesis from shoot tip explants of date palm. In: Al-Khayri J, Jain S, Johnson D (eds) Date palm biotechnology protocols Volume I. Methods in molecular biology, vol 1637. Humana Press, New York, pp 37–45. https://doi.org/10.1007/978-1-4939-7156-5_4
Silva-Maia JA, Pedroso-de Moraes C (2017) Influence of different concentrations of jasmonic acid on in vitro development of Catasetum fimbriatum Lindl. (Orchidaceae). Mod Phytomorphol 11:99–104. https://doi.org/10.5281/zenodo.1039717
Teoh ES (2019) Medicinal orchids of Central America. In: Teoh ES (ed) Orchids as aphrodisiac, medicine or food. Springer, Cham, pp 139–158. https://doi.org/10.1007/978-3-030-18255-7_9
Vasudevan R, Van Staden J (2011) Cytokinin and explant types influence in vitro plant regeneration of Leopard Orchid (Ansellia africana Lindl.). Plant Cell Tissue Organ Cult 107:123–129. https://doi.org/10.1007/s11240-011-9964-0
Vudala SM, Padial AA, Ribas LLF (2019) Micropropagation of Hadrolaelia grandis through transverse and longitudinal thin cell layer culture. S Afr J Bot 121:76–82. https://doi.org/10.1016/j.sajb.2018.07.017
Whiteley SE, Bunn E, Menon A, Mancera RL, Turner SR (2016) Ex situ conservation of the endangered species Androcalva perlaria (Malvaceae) by micropropagation and cryopreservation. Plant Cell Tissue Organ Cult 125:341–352. https://doi.org/10.1007/s11240-016-0955-z
Yeung EC (2017) A perspective on orchid seed and protocorm development. Bot Stud 58:1–14. https://doi.org/10.1186/s40529-017-0188-4
Zahid NA, Jaafar HZ, Hakiman M (2021) Micropropagation of ginger (Zingiber officinale Roscoe) ‘Bentong’ and evaluation of its secondary metabolites and antioxidant activities compared with the conventionally propagated plant. Plants 10:630. https://doi.org/10.3390/plants10040630
Zakizadeh S, Kaviani B, Hashemabadi D (2019) Micropropagation of two near threatened orchid. Part 1: Catasetum pileatum cv. Alba. Adv Hort Sci 33:475–483. https://doi.org/10.13128/ahsc8112
Acknowledgements
We thank M. Sc. Claudia Moctezuma-Granados for her invaluable technical assistance. This work was partially supported by Consejo Nacional de Ciencia y Tecnología (CONACYT, Mexico), grant number 269491/2016 provided to CCA. LJCP is a student of doctors in science supported by CONACYT fellowship (773045).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Castillo-Pérez, L.J., Alonso-Castro, A.J., Fortanelli-Martínez, J. et al. Micropropagation of Catasetum integerrimum Hook (Orchidaceae) through seed germination and direct shoot regeneration from pseudobulbs and roots. In Vitro Cell.Dev.Biol.-Plant 58, 279–289 (2022). https://doi.org/10.1007/s11627-021-10248-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-021-10248-3