Abstract
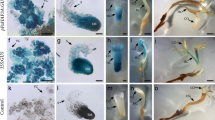
MicroRNA319 (miR319) plays an important role in leaf development. Although leaf discs are frequently used for plant transformation, little is known about the function of miR319 in shoot regeneration from leaf explants. To investigate the role of miR319 in de novo shoot organogenesis from leaf explants, the MIR319 gene was overexpressed in petunia (Petunia × hybrida), under the control of the 35S promoter. Petunia leaves overexpressing miR319 (miR319-OX) showed several morphological abnormalities, such as rounded, uneven, and curled leaf blade, larger leaf size, and smaller and rounded abaxial surface cells, which were differences compared to wild-type (WT) leaves. Additionally, miR319-OX leaf explants demonstrated enhanced shoot organogenesis on a growth medium containing 6-benzylaminopurine (BA) at shoot-inducing concentrations, and cytokinin-independent shoot regeneration on hormone-free medium compared with WT explants. Quantitative real-time PCR (qRT-PCR) analysis indicated that the expression of petunia NO APICAL MERISTEM (PhNAM) and SHOOT MERISTEMLESS (PhSTM) genes was markedly increased in miR319-OX explants compared with WT explants under shoot induction conditions, whereas the expression of petunia cytokinin-responsive type-A response regulator genes, PhRR2 and PhRR3, showed no differences in expression between WT and miR319-OX leaf explants. These results suggest that miR319 regulates the formation of shoot meristems from leaf explants in petunia during in vitro culture.



Similar content being viewed by others
References
Bombarely A, Moser M, Amrad A, Bao M, Bapaume L, Barry CS, Bliek M, Boersma MR, Borghi L, Bruggmann R, Bucher M, D’Agostino N, Davies K, Druege U, Dudareva N, Egea-Cortines M, Delledonne M, Fernandez-Pozo N, Franken P, Grandont L, Heslop-Harrison JS, Hintzsche J, Johns M, Koes R, Lv X, Lyons E, Malla D, Martinoia E, Mattson NS, Morel P, Mueller LA, Muhlemann J, Nouri E, Passeri V, Pezzotti M, Qi Q, Reinhardt D, Rich M, Richert-Poeggeler KR, Robbins TP, Schatz MC, Schranz ME, Schuurink RC, Schwarzacher T, Spelt K, Tang H, Urbanus SL, Vandenbussche M, Vijverberg K, Villarino GH, Warner RM, Weiss J, Yue Z, Zethof J, Quattrocchio F, Sims TL, Kuhlemeier C (2016) Insight into the evolution of the Solanaceae from the parental genomes of Petunia hybrida. Nat Plant 2:16074. https://doi.org/10.1038/nplants.2016.74
Bresso EG, Chorostecki U, Rodriguez RE, Palatnik JF, Schommer C (2018) Spatial control of gene expression by miR319-regulated TCP transcription factors in leaf development. Plant Physiol 176:1694–1708. https://doi.org/10.1104/pp.17.00823
Das Gupta M, Aggarwal P, Nath U (2014) CINCINNATA in Antirrhinum majus directly modulates genes involved in cytokinin and auxin signaling. New Phytol 204:901–912. https://doi.org/10.1111/nph.12963
Efroni I, Blum E, Goldshmidt A, Eshed Y (2008) A protracted and dynamic maturation schedule underlies Arabidopsis leaf development. Plant Cell 20:2293–2306. https://doi.org/10.1105/tpc.107.057521
Efroni I, Han S-K, Kim HJ, Wu M-F, Steiner E, Birnbaum KD, Hong JC, Eshed Y, Wagner D (2013) Regulation of leaf maturation by chromatin-mediated modulation of cytokinin responses. Dev Cell 24:438–445. https://doi.org/10.1016/j.devcel.2013.01.019
Faria DV, de Freitas Correia LN, de Matos EM, de Souza MVC, Batista DS, Costa MGC, de Paiva Neto VB, Xavier A, Rogalski M, Otoni WC (2019) Wounding and medium formulation affect de novo shoot organogenic responses in hypocotyl-derived explants of annatto (Bixa orellana L.). Vitro Cell Dev Biol - Plant 55:277–289. https://doi.org/10.1007/s11627-019-09978-2
Gordon-Kamm B, Sardesai N, Arling M, Lowe K, Hoerster G, Betts S, Jones AT (2019) Using morphogenic genes to improve recovery and regeneration of transgenic plants. Plants (Basel) 8:38. https://doi.org/10.3390/plants8020038
Guo Y, Han Y, Ma J, Wang H, Sang X, Li M (2014) Undesired small RNAs originate from an artificial microRNA precursor in transgenic petunia (Petunia hybrida). PLoS One 9:e98783. https://doi.org/10.1371/journal.pone.0098783
Hwang I, Sheen J, Muller B (2012) Cytokinin signaling networks. Annu Rev Plant Biol 63:353–380. https://doi.org/10.1146/annurev-arplant-042811-105503
Ikeuchi M, Iwase A, Rymen B, Lambolez A, Kojima M, Takebayashi Y, Heyman J, Watanabe S, Seo M, de Veylder L, Sakakibara H, Sugimoto K (2017) Wounding triggers callus formation via dynamic hormonal and transcriptional changes. Plant Physiol 175:1158–1174. https://doi.org/10.1104/pp.17.01035
Koyama T, Furutani M, Tasaka M, Ohme-Takagi M (2007) TCP transcription factors control the morphology of shoot lateral organs via negative regulation of the expression of boundary-specific genes in Arabidopsis. Plant Cell 19:473–484. https://doi.org/10.1105/tpc.106.044792
Koyama T, Mitsuda N, Seki M, Shinozaki K, Ohme-Takagi M (2010) TCP transcription factors regulate the activities of ASYMMETRIC LEAVES1 and miR164, as well as the auxin response, during differentiation of LEAVES in Arabidopsis. Plant Cell 22:3574–3588. https://doi.org/10.1105/tpc.110.075598
Koyama T, Sato F, Ohme-Takagi M (2017) Roles of miR319 and TCP transcription factors in leaf development. Plant Physiol 175:874–885. https://doi.org/10.1104/pp.17.00732
Li Z, Li B, Shen WH, Huang H, Dong A (2012) TCP transcription factors interact with AS2 in the repression of class-I KNOX genes in Arabidopsis thaliana. Plant J 71:99–107. https://doi.org/10.1111/j.1365-313X.2012.04973.x
Mallona I, Lischewski S, Weiss J, Hause B, Egea-Cortines M (2010) Validation of reference genes for quantitative real-time PCR during leaf and flower development in Petunia hybrida. BMC Plant Biol 10:4. https://doi.org/10.1186/1471-2229-10-4
Mao YF, Wu FJ, Yu X, Bai JJ, Zhong WL, He YK (2014) microRNA319a-targeted Brassica rapa ssp pekinensis TCP genes modulate head shape in chinese cabbage by differential cell division arrest in leaf regions. Plant Physiol 164:710–720. https://doi.org/10.1104/pp.113.228007
Motte H, Vereecke D, Geelen D, Werbrouck S (2014) The molecular path to in vitro shoot regeneration. Biotechnol Adv 32:107–121. https://doi.org/10.1016/j.biotechadv.2013.12.002
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nath U, Crawford BC, Carpenter R, Coen E (2003) Genetic control of surface curvature. Science 299:1404–1407
Nicolas M, Cubas P (2016) TCP factors: new kids on the signaling block. Curr Opin Plant Biol 33:33–41. https://doi.org/10.1016/j.pbi.2016.05.006
Nishijima T, Niki T, Niki T (2011) The large-flowered petunia (Petunia hybrida Vilm.) genotype promotes expressions of type-a response regulator and cytokinin receptor genes like cytokinin response. J Jpn Soc Hortic Sci 80:343–350. https://doi.org/10.2503/jjshs1.80.343
Olsen AN, Ernst HA, Leggio LL, Skriver K (2005) NAC transcription factors: structurally distinct, functionally diverse. Trends Plant Sci 10:79–87. https://doi.org/10.1016/j.tplants.2004.12.010
Ori N, Cohen AR, Etzioni A, Brand A, Yanai O, Shleizer S, Menda N, Amsellem Z, Efroni I, Pekker I, Alvarez JP, Blum E, Zamir D, Eshed Y (2007) Regulation of LANCEOLATE by miR319 is required for compound-leaf development in tomato. Nat Genet 39:787–791. https://doi.org/10.1038/ng2036
Palatnik JF, Allen E, Wu X, Schommer C, Schwab R, Carrington JC, Weigel D (2003) Control of leaf morphogenesis by microRNAs. Nature 425:257–263
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108. https://doi.org/10.1038/nprot.2008.73
Schommer C, Palatnik JF, Aggarwal P, Chetelat A, Cubas P, Farmer EE, Nath U, Weigel D (2008) Control of jasmonate biosynthesis and senescence by miR319 targets. PLoS Biol 6:1991–2001. https://doi.org/10.1371/journal.pbio.0060230
Sharma VK, Fletcher JC (2002) Maintenance of shoot and floral meristem cell proliferation and fate. Plant Physiol 129:31–39. https://doi.org/10.1104/pp.010987
Stuurman J, Jaggi F, Kuhlemeier C (2002) Shoot meristem maintenance is controlled by a GRAS-gene mediated signal from differentiating cells. Genes Dev 16:2213–2218. https://doi.org/10.1101/gad.230702
Uberti-Manassero NG, Coscueta ER, Gonzalez DH (2016) Expression of a repressor form of the Arabidopsis thaliana transcription factor TCP16 induces the formation of ectopic meristems. Plant Physiol Biochem 108:57–62. https://doi.org/10.1016/j.plaphy.2016.06.031
Yang C, Li D, Mao D, Liu X, Ji C, Li X, Zhao X, Cheng Z, Chen C, Zhu L (2013) Overexpression of microRNA319 impacts leaf morphogenesis and leads to enhanced cold tolerance in rice (Oryza sativa L.). Plant Cell Environ 36:2207–2218. https://doi.org/10.1111/pce.12130
Zhang B, Yang X, Yang CP, Li MY, Guo YL (2016) Exploiting the CRISPR/Cas9 system for targeted genome mutagenesis in petunia. Sci Rep 6:20315. https://doi.org/10.1038/Srep20315
Zhang G, Zhao F, Chen L, Pan Y, Sun L, Bao N, Zhang T, Cui CX, Qiu Z, Zhang Y, Yang L, Xu L (2019) Jasmonate-mediated wound signalling promotes plant regeneration. Nat Plant 5:491–497. https://doi.org/10.1038/s41477-019-0408-x
Zhou M, Li D, Li Z, Hu Q, Yang C, Zhu L, Luo H (2013) Constitutive expression of a miR319 gene alters plant development and enhances salt and drought tolerance in transgenic creeping bentgrass. Plant Physiol 161:1375–1391. https://doi.org/10.1104/pp.112.208702
Zhou WK, Lozano-Torres JL, Blilou I, Zhang XY, Zhai QZ, Smant G, Li CY, Scheres B (2019) A jasmonate signaling network activates root stem cells and promotes regeneration. Cell 177:942–956. https://doi.org/10.1016/j.cell.2019.03.006
Funding
This work was supported by the National Natural Science Foundation of China (31272199) and Fundamental Research Funds for the Central Universities (XDJK2020D038).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Editor: Todd Jones
Rights and permissions
About this article
Cite this article
Guo, Y., Qin, X., Zhang, B. et al. Overexpression of miR319 in petunia (Petunia × hybrida) promotes de novo shoot organogenesis from leaf explants. In Vitro Cell.Dev.Biol.-Plant 57, 72–79 (2021). https://doi.org/10.1007/s11627-020-10063-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-020-10063-2