Abstract
The inadequate response of Indica rice (Oryza sativa L.) to anther culture hinders the utility of this technique for rice crop improvement. This study focused on intensifying the in vitro anther response of selected varieties from Sri Lankan germplasm by precisely targeting the most effective maturity stage of microspores for culture. Analysis of five varieties responsive to anther culture (“At 303”, “Bw 272-6B”, “Dahanala”, “Dikwee”, and “Herath Banda”), revealed that the microspore development stage at the time of culture was crucial for fine-tuning the callus induction response. The external guide used to determine microspore maturity which was the internode distance between the last two leaves of the panicle, could not be applied equally for all rice varieties. Anther response could be improved by, at least, four- to sixfold by precisely targeting anthers with late uninucleate microspores for culture. Close correlation was demonstrated between the stage of nuclear division in the microspores and the degree of starch accumulation. Therefore, the degree of starch deposition in the microspores can be used as an effective cytological marker, which can be applied more rapidly and reliably to assess microspore maturity, in place of the internode distance.
Similar content being viewed by others
Introduction
Anther culture is a useful technique in plant breeding, which allows the development of homozygous plant lines. Regeneration of haploid plants from microspores in the anthers, and the subsequent chromosome doubling of the haploids, results in fully homozygous diploids. Therefore, anther culture facilitates the immediate conversion of heterozygous breeding material to homozygous breeding material, which is a faster alternative to conventional inbred line development. In rice, haploid plant production by anther culture is accomplished in two steps. First, induction of a callus from the cultured anther followed by regeneration of plants from induced callus tissue. For the technique to be practical, these two in vitro events must occur efficiently and reliably in rice varieties.
Even though this anther culture approach has been integrated into varietal improvement programs in Japonica rice, it has not been very successful in Indica rice varieties (Silva 2010; Mishra and Rao 2016). The major constraints of using anther culture with Indica rice are the low rate of callus induction, poor shoot regeneration ability, and a high tendency to regenerate albino shoots (Sripichitt et al. 2000; He et al. 2006).
Embryogenic induction of microspores within the anthers is only possible at certain stages of microspore maturity (Dunwell 2010; Germana 2011). In rice, the best stage for embryogenic induction is the mid- to late-uninucleate stage of microspore division (Niroula and Bimb 2009; Silva and Ratnayake 2009; Mishra and Rao 2016), and it is important to collect anthers with microspores at this most responsive stage. Determining when rice is in this stage requires nuclear staining and cytological examination of microspores prior to culture. However, nuclear staining of microspores from each rice panicle, before dissecting anthers for culture, greatly impedes the anther culture process. Therefore, it is common to use an easily identifiable morphological indicator trait that correlates well with the maturity stage in microspores. The visual guide that has been used with rice anther culture is the measured distance between the nodes of the last two leaves (flag leaf and penultimate leaf) (Bishnoi et al. 2000; Silva and Ratnayake 2009), or, less frequently, the panicle length at the time of harvest (Cha-um et al. 2009; Rout et al. 2016).
The present study critically examined whether a specific fixed internode distance (or a distance range), could be used as a guide to accurately determine microspore maturity in different target varieties. Further, it is proposed that the use of a direct cytological marker such as the degree of starch accumulation in microspores, would be more accurate than internode distance, and more convenient than nuclear staining to rapidly determine microspore maturity.
Materials and methods
Plant material
From the local Indica rice germplasm, five varieties were selected for anther culture including three traditional varieties (“Dahanala”, “Dikwee”, and “Herath Banda”) and two new improved popular varieties (“At 303” and “Bw 272-6B”). These varieties are useful in introgressive breeding and as parents in crosses because they possess many features of agronomic interest. The mother plants were grown in rice fields using standard agronomic practices. Several batches of seedlings were successively raised from each variety in the nursery and stagger-planted in the field at approximately weekly intervals, so that panicles at the required stage of maturity were available for anther culture throughout the season.
Anther culture using the external visual guide to determine microspore maturity
Anther culture was performed using anthers extracted from panicles that were collected at the booting stage (Yoshida 1981). Panicle boots were harvested from mother plants between 9:00 am and 10:00 am. Internode distance between the last two leaves, an external indicator previously used with rice anther culture, was used to estimate microspore maturity at the time of panicle harvest (Fig. 1 a). Panicle boots were collected in all the rice varieties when the internode length was 8–11 cm. After the leaf blades of the flag leaf and the penultimate leaf were removed, leaf sheath-enclosed panicle boots were wiped with 70% (v/v) ethanol and wrapped in aluminum foil. These panicles were placed (five panicles per bag) in labeled, sealed bags (5 cm × 15 cm) made out of polypropylene material for cold pre-treatment at 10 °C for 7–10 d (Fig. 1 b).
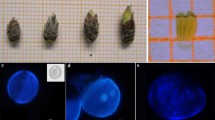
Booting stage rice panicles for anther culture. (a) A panicle boot measured for the internode distance: (a1) leaf sheath-enclosed panicle, (a2) node of the penultimate leaf, (a3) node of the flag leaf, (a4) internode distance. Scale bar = 3 cm. (b) Panicle boots (7–12 cm internode distance) wrapped in aluminum foil prepared for cold pre-treatment. Scale bar = 2 cm.
Cold pre-treated panicles were disinfected, and anther dissection and culture were carried out under sterile conditions using a laminar air-flow cabinet (Biobase BBS-H1300-D, Biobase Biodustry Co., Ltd., Shandong, China) as described by Silva and Ratnayake (2009). Excised anthers were cultured on N6 medium (Chu 1978), modified by replacing sucrose with maltose (60.0 g L−1) and solidified with bacteriological grade agar (8.0 g L−1). The pH of the medium was adjusted to 5.8 with 1 M NaOH, and the medium was sterilized for 20 min at 120 °C using an autoclave (Tomy ES-215, Tomy Digital Biology Co., Ltd., Tokyo, Japan). The growth regulators 2,4-dichlorophenoxyacetic acid (2,4-D; 0.5 mg L−1), naphthaleneacetic acid (NAA; 2.5 mg L−1), and kinetin (0.5 mg L−1) were filter-sterilized and added to the autoclaved callus induction medium. For each rice variety, three replicate Petri dishes (9 cm), each containing 100–200 anthers, were cultured. Petri dishes with cultured anthers were wrapped in aluminum foil and incubated at 27 ± 2 °C in a dark room. For each variety, the number of anthers forming callus tissue was recorded up to 12 wk after the initial culture. Percentage data on callus induction were arcsine transformed and analyzed using one-way analysis of variance (ANOVA) and Statistical Analyses System (SAS®) version 9.0 (SAS® Institute, Inc., Cary, NC).
Calluses that grew to a size of 1–5 mm in diameter were separated from parent anther tissue and transferred to Petri dishes containing Murashige and Skoog (MS) medium (Murashige and Skoog 1962) for regeneration. Sucrose (30.0 g L−1) was used as the carbohydrate source, and the medium was solidified with bacteriological grade agar (8.0 g L−1). The pH of the medium was adjusted to 5.8 with 1 M NaOH before autoclaving (Tomy ES-215) for 20 min at 120 °C. The growth regulators 1.0 mg L−1 NAA, 0.5 mg L−1 kinetin, and 1.0 mg L−1 6-benzylaminopurine (BAP) were filter-sterilized and added to the autoclaved MS medium. The cultures were maintained at 27 ± 2 °C provided with continuous light using cool white florescent tubes (36 W) with a light intensity of 60 μmol m−2 s−1 until shoot regeneration occurred. Regenerated shoots with roots were transferred to test tubes containing half-strength MS medium prepared with half the concentration of the salts used previously, and without growth regulators. Then, the pH of the medium was adjusted to 5.8 with 1 M NaOH and bacteriological grade agar (8.0 g L−1) was used as the solidifying agent. Young plantlets were removed from the agar, transferred to pots (10 cm in diameter and 12 cm in height) with washed and cleaned river sand, and subjected to gradual acclimatization before being planted in paddy soil (collected from 10 cm depth of the plow layer of the standard agricultural field) and grown to maturity. Chemicals including agar used in the experiment were from HiMedia® Laboratories Pvt. Ltd., Mumbai, India, and growth regulators were from Sigma-Aldrich®, St. Louis, MO.
Determination of optimum microspore maturity stage for in vitro anther response Anther culture from panicles harvested at specific stages of maturity
Booting-stage panicles corresponding to different internode lengths (7, 8, 9, 10, 11, and 12 cm) were collected from each of the five rice varieties. Harvested panicles were cold pre-treated, and anthers from spikelets in the middle portion of each panicle were cultured in Petri plates containing N6 callus induction medium. Anthers collected from panicles pertaining to different internode lengths were cultured separately in individual Petri dishes. Three replicate Petri dishes were maintained for each internode length and each variety, with 100–200 anthers in each dish. Data on callus induction in anthers were recorded for each internode length, and the internode length that produced the optimum anther response was determined for each individual rice variety.
Cytological observations on microspores from panicles harvested at specific stages of maturity
In a parallel study, a sample of microspores extracted from panicles with internode lengths of 7–12 cm from the five rice varieties were subjected to cytological examination. Anthers from the middle segment of the panicle were fixed in a 1:3 solution of glacial acetic acid and absolute ethanol for 24 h. Anthers were stained with iron alum-hematoxylin following the procedure of Gupta and Borthakur (1987). Microspores were then extracted from stained anthers and examined for nuclear division stage. Another sample of microspores was stained with I2/KI to detect the deposition of starch. From each variety and development stage, 300 microspores were examined. Micrographs were taken using a light microscope (Carl Zeiss, Oberkochen, Germany) with a Zeiss Axiocam digital camera (Carl Zeiss) attached and images were processed with Zen 2012 (blue edition) imaging software (Carl Zeiss) to record the stages of nuclear division (early uninucleate, late uninucleate, or binucleate) and the degree of starch deposition in cells. Cytological data were combined with anther response data to determine the stage of microspore development that produced the highest anther response. All the chemicals used in the cytological study were from HiMedia® Laboratories Pvt. Ltd.
Results
Anther culture using an external visual guide to determine microspore maturity
In the initial experiment in which anthers were cultured from panicles with an 8–11 cm internode length, the mean callus induction frequency varied significantly (p ≤ 0.05) and ranged from 1.51 to 20.84% among the five rice varieties (Table 1). Mean callus induction was highest in the “Dahanala” variety and lowest in the “Bw 272-6B” variety. However, callus induction frequencies varied widely among replicate Petri dishes of any given variety. For example, in “Dahanala,” the callusing percentage varied from 11.9 to 27.1, and in “At 303,” anthers in two of the replicates showed zero response compared to 12.4% in the third. The variation in anther response was likely due to differences in the maturity stages of microspores in anthers used for culture because anthers collected from panicles of different maturities (8–11 cm internode length) were cultured together. Therefore, the effect of the microspore maturity stage on anther culture efficiency was further investigated in a subsequent experiment by culturing anthers from panicles at different maturity levels individually and evaluating the response.
In the regeneration medium, only the rice variety “At 303” produced complete green shoots (Fig. 2 a), and these were raised to complete plants through gradual acclimatization. In three other varieties (“Dahanala”, “Dikwee”, and “Herath Banda”), some calluses became chlorophyllous and showed signs of shoot bud initiation (Fig. 2 b) but did not progress further in development. Shoots produced in the rice variety “Bw 272-6B” were all albinos (Fig. 2 c; Table 2).
Determination of microspore maturity stage for optimal in vitro anther response Callus induction from anthers in panicles at different maturity levels
In each of the five rice varieties (“At 303”, “Bw 272-6B”, “Dahanala”, “Dikwee”, and “Herath Banda”), the optimal anther response occurred at a specific stage of panicle maturity, as denoted by the internode length. In the three traditional varieties, “Dahanala” (17.22% callus induction frequency), “Herath Banda” (9.40%), and “Dikwee” (7.69%), optimal anther response was observed from anthers of panicle boots taken at an internode distance of 11 cm. In the variety “At 303,” the optimal callus induction frequency (6.57%) coincided with an internode length of 10 cm. In the variety “Bw 272-6B,” the optimal callus induction frequency (1.91%) was obtained from panicles with an internode length of 9 cm (Table 3). Based on these results, it was clear that in all varieties except “At 303”, the anther response was limited to one or two panicle maturity levels (Table 3).
Cytological observation of microspore development stage
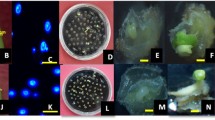
Nuclear staining with iron alum-hematoxylin was used to distinguish the different developmental stages (early uninucleate, late uninucleate, or binucleate) of the microspore. Uninucleate microspores had a single large nucleus that appeared darkly stained. These microspores were in either the early or late uninucleate stage, as determined by the presence or absence, respectively, of a vacuole. In early uninucleate microspores, a large vacuole occupied most of the cell space, and the nucleus was close to the periphery of the cell. The more mature cells, which were in the late uninucleate stage, were densely cytoplasmic and the vacuole had been reabsorbed (Fig. 3 a, b). Binucleate microspores could be clearly identified by the presence of two separate nuclei (Fig. 3 c). Unfortunately, not all of the cells in an anther were stained equally with iron alum-hematoxylin, and in some microspores, the nuclei could not be clearly observed. Therefore, numerous microspores within an anther had to be screened to find cells with well-stained nuclei to reliably establish the frequency of cells at a particular stage of division. In all five varieties, the optimal anther response was observed when the majority of microspores (59–84%) were in the late uninucleate stage, and some (16–41%) were in the binucleate stage (Table 4). Microspores in the early uninucleate stage were unresponsive.
Staining for starch with I2/KI showed that the stage of nuclear division was related to the degree of starch accumulation within the cells (Fig. 5). For example, early and late uninucleate microspores had no starch or very little starch deposition within the cytoplasm, and therefore remained yellow in color after I2/KI staining (Fig. 4 a). The more mature binucleate cells were moderately or densely filled with starch grains and were stained a deep blue/black with I2/KI (Fig. 4 b, c). Almost all cells in an anther were clearly distinguishable based on the degree of starch deposition when the cells were stained with I2/KI. Therefore, the presence of starch may be used as a quick and reliable guide to establish the appropriate stage of microspore development for in vitro culture of anthers. In general, anthers should be used for culture when a sample of microspores shows that approximately 25% of cells have starch deposition (Fig. 5), which would correspond with approximately 75% of the microspores being in the late uninucleate stage, which is the most appropriate stage of maturity for culture.
Microspores stained with I2/KI. (a) A uninucleate cell with no starch remained yellow in color. (b) A binucleate cell with slight starch deposition exhibited light staining in deep blue/black color. (c) A binucleate cell with dense starch deposition exhibited heavy staining in deep blue/black color.
Percentage of microspores in different stages of nuclear division (early uninucleate, late uninucleate, binucleate) at the optimum anther responsive stage of the five rice varieties, correlated with the percentage of microspores with different levels of starch deposition (devoid of starch, filled with starch)
Discussion
Developing homozygous plants through anther culture in rice is considered a stepwise process, in which the initial stage is the development of callus on an appropriate callus induction medium. The second step is the regeneration of shoots from induced callus tissue, which usually occurs after transfer of the calluses onto separate regeneration medium. In many rice varieties, there was a wide disparity in the callusing frequency among the replicates. It was initially assumed that all cultured anthers were at a similar stage of maturity, as estimated by the distance between the flag leaf and penultimate leaf nodes at panicle harvest (8–11 cm). However, the present data suggest that there may have been distinct differences in the anthers collected from different panicles within this range, particularly in relation to the developmental stage of microspores at the cellular level, resulting in a wide variation in the callus induction frequency among the replicates. This led to an additional question of whether the external morphological guide used was stringent enough to distinguish developmental differences in anthers and microspores within a variety. If the external morphological guide was not stringent enough, then it may not accurately distinguish the most appropriate microspore development stage for culture among different varieties. In each rice variety, the overall callus induction frequency, determined by the total number of anthers cultured and their response at all maturity levels, was much lower than the response observed for anthers cultured at the optimal level of maturity. For example, in the variety “Dikwee”, a sixfold increase in callus induction frequency could be obtained by restricting panicle harvest to an internode distance of 11 cm (Table 3). Similarly, anthers at the most appropriate stage of maturity produced markedly enhanced anther response in all other rice varieties. In initial experiments, anthers used for culture were collected from panicles with common internode distances of 8–11 cm, irrespective of the variety. Clearly, this may have led to the widely variable results obtained for replicates within a variety. These subsequent results suggest that the stage of panicle maturity had a remarkable impact on the success of rice anther culture, and the challenge was to precisely identify the most suitable stage for culture for each individual variety.
Thus, although the distance between the flag leaf and penultimate leaf nodes had been used as a visual guide to microspore maturity previously (Bishnoi et al. 2000; Bagheri et al. 2009), the applicability of using a common distance in different varieties that vary in other morphological traits, such as plant height and age to maturity, is questionable. Panicle maturity, determined by the internode length that produces the optimal anther response, varied according to the rice variety and was observed to be somewhat related to individual plant height (Table 3). The average plant height of the different varieties tested in this study varied from 68.4 to 86.7 cm, and all of the traditional varieties (“Dahanala”, “Dikwee”, and “Herath Banda”) used in this experiment were significantly taller than the new improved varieties (p ≤ 0.05). The optimal anther response in these traditional varieties was obtained from panicles with larger internode distances than those of the two improved varieties (“At 303” and “Bw 272-6B”). These results demonstrate that the usual practice of using an arbitrary internode distance (or a range in distance) as a guide to panicle maturity is unlikely to be equally appropriate for all rice varieties and can lead to inferior performances with respect to callus induction.
The microspore development stage at the time of culture is a crucial factor to determine the success in callus induction (Datta 2005; Silva 2010; Germana 2011). In rice anther culture, the optimal stages of microspore development to induce an in vitro callus response are the late uninucleate to early binucleate stages (Afza et al. 2000). The present study similarly found that late uninucleate microspores produced the best anther response. In routine anther culture, an external morphological indicator is used as a guide to identify the appropriate stage of microspore division. In rice, 7–11 cm between the flag leaf and penultimate leaf nodes is generally considered an accurate guide for harvesting appropriately mature microspores. However, the present study demonstrated that this marker distance varies with the individual rice variety, and applying a range was not effective. The study also showed that by precisely targeting the correct microspore development stage for culture, callus induction frequency could be improved greater than sixfold in the rice varieties studied.
Staining microspores for nuclei and for starch were the two cytological procedures analyzed to assess the practical utility of these methods to precisely target the optimal microspore maturity stage. Visualizing nuclei in some microspores was problematic and required extensive examination of numerous cells after staining. Therefore, the degree of starch accumulation, which is more straightforward, may be a more efficient guide to establish the appropriate stage of microspore development prior to in vitro culture of anthers. This study provides a method to reduce the gap between the potential and actual level of success in Indica rice anther culture by culturing microspores at the most amenable stage.
References
Afza R, Shen M, Arias FJZ, Xie J, Fundi HK, Lee K, Bobadilla-Mucino E, Kodym A (2000) Effect of spikelet position on rice anther culture efficiency. Plant Sci 153:155–159
Bagheri N, Jelodar NB, Ghanbari A (2009) Evaluation of effective factors in anther culture of Iranian rice (Oryza sativa L.) cultivars, Biharean. Biologist 3:119–124
Bishnoi U, Jain RK, Rohilla JS, Chowdhury VK, Gupta KR, Chowdhury JB (2000) Anther culture of recalcitrant indica x Basmati rice hybrids. Euphytica 114:93–101
Cha-um S, Srianan B, Pichakum A, Kirdmanee C (2009) An efficient procedure for embryogenic callus induction and double haploid plant regeneration through anther culture of Thai aromatic rice (Oryza sativa L. subsp. indica). In Vitro Cell Dev Biol Plant 45:171–179
Chu C (1978) The N6 medium and its applications to anther culture of cereal crops. Proceedings of the Symposium on Plant Tissue Culture. Science Press, Beijing, pp 43–50
Datta SK (2005) Androgenic haploids: factors controlling development and its application in crop improvement. Curr Sci 89:1870–1878
Dunwell JM (2010) Haploids in flowering plants: origins and exploitation. Plant Biotechnol J 8:377–424
Germana MA (2011) Anther culture for haploid and doubled haploid production. Plant Cell Tissue Organ Cult 104:283–300
Gupta HS, Borthakur DN (1987) Improved rate of callus induction from rice anther culture following microscopic staging of microspores in iron alum-haematoxylin. Theor Appl Genet 74:95–99
He T, Yang Y, Tu SB, Yu MQ, Li XF (2006) Selection of interspecific hybrids for anther culture of indica rice. Plant Cell Tissue Organ Cult 86:271–277
Mishra R, Rao GJN (2016) In vitro androgenesis in rice: advantages, constraints and future prospects. Rice Sci 23:57–68
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Niroula RK, Bimb HP (2009) Effect of genotype and callus induction medium on green plant regeneration from anther of Nepalese rice cultivars. Asian J Plant Sci 8:368–374
Rout P, Naik N, Ngangkham U, Verma RL, Katara JL, Singh ON, Samantaray S (2016) Doubled haploids generated through anther culture from an elite long duration rice hybrid, CRHR32: method optimization and molecular characterization. Plant Biotechnol 33:177–186
Silva TD (2010) Indica rice anther culture - can the impasse be surpassed? Plant Cell Tissue Organ Cult 100:1–11
Silva TD, Ratnayake WJ (2009) Anther culture potential of indica rice varieties, kurulu thuda and BG 250. Trop Agric Res Ext 12:53–56
Sripichitt P, Ozawa T, Otani M, Shimada T (2000) Improved method for anther culture of an indica rice cultivar of Thailand. Plant Prod Sci 3:254–256
Yoshida S (1981) Fundamentals of rice crop science. International Rice Research Institute, Los Banos, pp 54–55
Acknowledgements
This research was graciously funded by the National Science Foundation (NSF) of Sri Lanka under the research grant RG/2011/BT/10.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
D.M.R.G. Mayakaduwa reports a grant from the National Science Foundation (NSF) of Sri Lanka (grant number RG/2011/BT/10), and Prof. T.D. Silva reports a grant from the National Science Foundation (NSF) of Sri Lanka (grant number RG/2011/BT/10).
Additional information
Editor: Jianxin Chen
Rights and permissions
About this article
Cite this article
Mayakaduwa, D.M.R.G., Silva, T.D. A cytological indicator allows rapid assessment of microspore maturity, leading to improved in vitro anther response in Indica rice (Oryza sativa L.). In Vitro Cell.Dev.Biol.-Plant 53, 591–597 (2017). https://doi.org/10.1007/s11627-017-9855-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-017-9855-0