Abstract
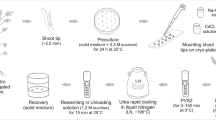
A simple, in vitro multiplication and mid-term conservation system was developed for Chlorophytum borivilianum Sant. et Fernand, which is documented as a critically endangered herb on the International Union for Conservation of Nature and Natural Resources red list. In vitro shoot multiplication was achieved by culture on Murashige and Skoog medium supplemented with 2 mg L−1 6-benzyladenine and 0.2 mg L−1 naphthalene acetic acid. Continuous shoot proliferation was observed up to the seventh subculture cycle on this medium. A production cycle of 1 yr was completed when rooting and acclimatization times were included in the subculture cycle. In vitro, slow-growth storage was efficiently used for mid-term conservation of elite clones when the sucrose concentration of the medium was 120 g L−1, which enabled 100% survival from cultures stored for 4 mo without any subculture or medium addition. After 4 mo of in vitro storage, cultures transferred to the standard medium exhibited 100% regeneration, and the shoots were morphologically identical. These shoots were rooted in a medium supplemented with 2 mg L−1 indole-3-butyric acid. Rooted plantlets were transferred to the field and showed 95% survival after acclimatization. This is the first report of in vitro conservation via slow-growth storage of this important medicinal plant.






Similar content being viewed by others
References
Acharya D, Mitaine-Offer A-C, Kaushik N, Miyamoto T, Paululat T, Mirjolet J-F, Lacaille-Dubois M-A (2008) Furostane-type steroidal saponins from the roots of Chlorophytum borivilianum. Helv Chim Acta 91:2262–2269
Acharya D, Mitaine-Offer A-C, Kaushik N, Miyamoto T, Paululat T, Mirjolet J-F, Duchamp O, Lacaille-Dubois M-A (2009) Cytotoxic spirostane-type saponins from the roots of Chlorophytum borivilianum. J Nat Prod 72:177–181
Bairu MW, Aremu AO, Staden JV (2011) Somaclonal variation in plants: causes and detection methods. Plant Growth Regul 63:147–173
Bordia PC, Joshi A, Simlot MM (1995) Safed Musli. In: Chadha KL, Gupta R (eds) Advances in horticulture, vol 11, Medicinal and aromatic plants. Malhotra Publishing House, New Delhi, pp 429–451
Capuana M, Lonardo SD (2013) In vitro conservation of chestnut (Castanea sativa) by slow growth. In Vitro Cell Dev Biol Plant 49:605–610
Dave A, Bilochi G, Purohit SD (2003) Scaling-up production and field performance of micropropagated medicinal herb ‘Safed Musli’ (Chlorophytum borivilianum). In Vitro Cell Dev Biol Plant 39:419–424
Dave A, Joshi N, Purohit SD (2004) In vitro propagation of Chlorophytum borivilianum using encapsulated shoot buds. Eur J Hortic Sci 69:37–42
Devi SP, Kumaria S, Rao SR, Tandon P (2014) Single primer amplification reaction (SPAR) methods reveal subsequent increase in genetic variations in micropropagated plants of Nepenthes khasiana Hook. f. maintained for three consecutive regenerations. Gene 538:23–29
Dodds JH, Roberts LW (1995) Experiments in plant tissue culture. Cambridge University Press, New York
George EF (1996) Micropropagation in practice. In: Plant propagation by tissue culture. Part 2, 2nd edition. Exegetics Ltd, England
Gopal J, Chauhan NS (2010) Slow growth in vitro conservation of potato. Potato Res 53:141–149
Hrahsel L, Basu A, Sahoo L, Thangjam R (2014) In vitro propagation and assessment of the genetic fidelity of Musa acuminata (AAA) cv. Vaibalhla derived from immature male flowers. Appl Biochem Biotechnol 172:1530–1539
IUCN: International Union for Conservation of Nature and Natural Resources (2015) The IUCN red list of threatened species. Version 2015–4. http://www.iucnredlist.org. Accessed on Jan 2015
Jat RD, Bordia PC (1990) Propagation studies in safed musli (Chlorophytum species). In: Chaudhary BL, Aery NC, Katewa SS (eds) Current status and emerging challenges. Proceedings of the National Symposium on Advances in Plant Sciences. ML Sukhadia University, Udaipur
Joshi N, Purohit SD (2012) Optimization of factors influencing shoot multiplication during micropropagation of Chlorophytum borivilianum Sant. et Fernand. Proc Natl Acad Sci India Sect B Biol Sci 82:421–429
Kaur R, Arora S, Thukral AK (2009) Enhancing seed germination of Chlorophytum borivilianum Sant. et Fernand. with PGRs, steroidal hormones and zinc. Res J Seed Sci 2:32–39
Kaushik N (2005) Saponins of Chlorophytum species. Phytochem Rev 4:191–196
Khuri S, Moorby J (1995) Investigations into the role of sucrose in potato cv. Estima microtubers production in vitro. Ann Bot 75:295–303
Lattoo SK, Bamotra S, Dhar RS, Khan S, Dhar AK (2006) Rapid plant regeneration and analysis of genetic fidelity of in vitro derived plants of Chlorophytum arundinaceum Baker an endangered medicinal herb. Plant Cell Rep 25:499–506
Lipavska H, Vreugdenhil D (1996) Uptake of mannitol from the media by in vitro grown plants. Plant Cell Tissue Organ Cult 45:103–107
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nayar MP, Sastry ARK (1988) Chlorophytum borivilianum. In: Nayar MP, Sastry ARK (eds) Red data book of Indian plants, vol 2. Botanical Survey of India, Calcutta, p 412
NMPB: National Medicinal Plant Board, New Delhi, Govt of India (2002) Medicinal plant database. http://www.impla.in/. Accessed on June 2014
Panis B, Totte NV, Nimmen K, Withers LA, Swennen R (1996) Cryopreservation of banana (Musa spp.) meristem cultures after preculture on sucrose. Plant Sci 121:95–106
Peng X, Ji Q, Wu H, Li Y (2015) Slow-growth conservation and clonal fidelity of Tetrastigma hemsleyanum microplants. In Vitro Cell Dev Biol Plant 51:463–470
Purohit SD, Dave A, Kukda G (1994) Micropropagation of safed musli (Chlorophytum borivilianum), a rare Indian medicinal herb. Plant Cell Tissue Organ Cult 39:93–96
Quraishi A, Koche V, Sharma P, Mishra SK (2004) In vitro clonal propagation of neem (Azadirachta indica). Plant Cell Tissue Organ Cult 78:281–284
Reed BM (1992) Cold storage of strawberries in vitro: a comparison of three storage system. Fruit Var J 46:98–102
Remya M, Bai VN, Mutharaian VN (2013) In vitro regeneration of Aristolochia tagala and production of artificial seeds. Biol Plant 57:210–218
Rizvi MZ, Kukreja AK, Khanuja SPS (2007) In vitro culture of Chlorophytum borivilianum Sant. et Fernand. in liquid culture medium as cost-effective measure. Curr Sci India 92:87–90
Sharma U, Mohan JSS (2006) In vitro clonal propagation of Chlorophytum borivilianum Sant. et Fernand., a rare medicinal herb from immature floral buds along with inflorescence axis. Indian J Exp Biol 44:77–82
Siddique I, Bukhari NAW, Perveen K, Siddiqui I (2015) Influence of plant growth regulators on in vitro shoot multiplication and plantlet formation in Cassia angustifolia Vahl. Braz Arch Biol Technol 58:686–691
SPSS, Inc. (1999) SPSS 10.0 syntax reference guide. Chicago, IL
Thakur S, Tiwari KL, Jadhav SK (2015) In vitro approaches for conservation of Asparagus racemosus Willd. In Vitro Cell Dev Biol Plant 51:619–625
Trejgell A, Kaminska M, Tretyn A (2015) In vitro slow growth storage of Senecio macrophyllus shoots. Acta Physiol Plant 37:234
Withers LA (1986) In vitro approaches to the conservation of plant genetic resources. In: Withers LA, Alderson PG (eds) Plant tissue culture and its agriculture applications. Butterworths, London, pp 261–276
Withers LA, Engelmann F (1997) In vitro conservation of plant genetic resources. In: Altman A (ed) Biotechnology in agriculture. Marcel Dekker, New York, pp 57–88
Acknowledgments
The authors gratefully acknowledge financial assistance provided by University Grants Commission, New Delhi in the form of Rajiv Gandhi National Fellowship (F1-17.1/2013-14/RGNF-2013-14-SC-CHH-36789). The authors also acknowledge DST, New Delhi (DST-FIST Sl. No. 270 for tenure of 2013–18) and CCOST, Raipur (Endt. No. 1089/CCOST/MRP/2014) for providing financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Wenhao Dai
Rights and permissions
About this article
Cite this article
Chauhan, R., Keshavkant, S., Jadhav, S.K. et al. In vitro slow-growth storage of Chlorophytum borivilianum Sant. et Fernand: a critically endangered herb. In Vitro Cell.Dev.Biol.-Plant 52, 315–321 (2016). https://doi.org/10.1007/s11627-016-9756-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-016-9756-7