Abstract
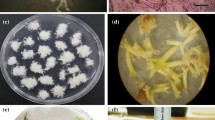
Somatic embryogenesis was developed as a method of mass propagation for Lepidosperma drummondii (Cyperaceae), a difficult to propagate but important species for post-mining restoration in a region of high plant biodiversity, in the southwest of Western Australia. Cultures were initiated from excised zygotic embryos, shoot cultures to rhizomes. Only zygotic embryos of L. drummondii developed somatic embryos, with half strength Murashige and Skoog basal medium (BM) and 1 μM 2,4-dichlorophenoxyacetic acid (2,4-D) being the most effective combination. The first culture cycle yielded a mean of 30 somatic embryos per excised zygotic embryo forming an embryo cluster. After a further 6 wk in culture (on fresh BM with 1 μM 2,4-D), approximately 350 somatic embryos per starting embryo cluster were recorded. Following regular sub-culturing of primary somatic embryo clusters onto fresh media (every 4 wk), more than 74,000 secondary somatic embryos were estimated to have been produced after eight subculture periods. This translates to between 1,000 and 2,000 somatic embryos produced from an estimated 45 mg of starting tissue per culture plate or potentially 22,0000–44,000 somatic embryos per gram of tissue. This is a significant improvement over all previous methods used to propagate L. drummondii, in which typical in vitro shoot multiplication rates are as low as 1.43 per 8 wk. This also compared favourably with published data and concurrent experiments undertaken in this study (as an extra control measure) on somatic embryo production for a related species Baloskion tetraphyllum (using the same BM with 1 μM 2,4-D and coleoptile segments as explants). Various media combinations were investigated for efficacy in converting somatic embryos into plants with best results ranging from 86% to 100% conversion for B. tetraphyllum on BM without plant growth regulators. Development of L. drummondii somatic embryos into plants was not observed on BM without plant growth regulators. However, a best result of 39% conversion to plants was observed on BM with 1 μM thidiazuron. This is the first report of successful development of somatic embryogenesis and conversion of somatic embryos into plants using thidiazuron for the Australian cyperale L. drummondii.




Similar content being viewed by others
References
Bremer K. Gondwanan evolution of the grass alliance of families (Poales). Evolution 56: 1374–1387; 2002.
Conacher C. A.; Poiner I. R.; Butler J.; Pun S.; Tree D. J. Germination, storage and viability testing of seeds of Zostera capricorni Aschers. from a tropical bay in Australia. Aquatic Botany 49: 47–58; 1994.
Custers J. B. M.; Cordewener J. H. G.; Nollen Y.; Dons H. J. M.; Van Lookeren Campagne M. M. Temperature controls both gametophytic and sporophytic development in microspore cultures of Brassica napus. Plant Cell Rep 13: 267–271; 1994.
George E. F. Plant propagation by tissue culture (part II: in practice). Exegetics, Edington; 1996.
Halperin W.; Wetherell D. F. Adventive embryogeny in tissue cultures of the wild carrot, Daucus carota. Am J Bot 51: 274–283; 1964.
Keller W. A. Stimulation of embryogenesis and haploid production in Brassica campestris anther cultures by elevated temperature treatments. Theo App Gen 55(2): 65–67; 1979.
Maximova S. N.; Alemanno L.; Young A.; Ferriere N.; Traore A.; Guiltinan M. J. Efficiency, genotypic variability and cellular origin of primary and secondary somatic embryogenesis of Theobroma cacao L. In Vitro Cell Dev Biol-Plant 38: 252–259; 2002.
Meney K. A.; Dixon K. W. In vitro propagation of Western Australian rushes (Restionaceae and related families) by embryo culture. Part 1. In vitro embryo growth. Plant Cell Tiss Org Cult 41: 107–113; 1995a.
Meney K. A.; Dixon K. W. In vitro propagation of Western Australian rushes (Restionaceae and related families) by embryo culture. Part 2. Micropropagation. Plant Cell Tiss Org Cult 41: 115–124; 1995b.
Meney K. A.; Pate J. S. (eds). Australian rushes. Biology, identification and conservation of Restionaceae and allied families. University of Western Australia Press, Perth; 1999.
Murashige T.; Skoog F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15: 473–497; 1962.
Panaia M. Somatic embryogenesis as an efficient method for clonal propagation of Australian native southern rushes (Restionaceae) and sedges (Cyperaceae) for rehabilitation of disturbed habitats and horticultural utilisation, Ph.D. thesis. University of Western Australia, Crawley, WA, Australia; 2002.
Panaia M.; Bunn E.; McComb J. Incubation temperature critical to successful stimulation of in vitro zygotic embryo growth in four Australian native Cyperaceae species. Plant Cell Tiss Org Cult 97: 197–202; 2009.
Panaia M.; Senaratna T.; Dixon K. W.; Sivasithamparam K. High-frequency somatic embryogenesis of Koala Fern (Baloskion tetraphyllum, Restionaceae). In Vitro Cell Dev Biol-Plant 40: 303–310; 2004a.
Panaia M.; Senaratna T.; Dixon K. W.; Sivasithamparam K. The role of cytokinins and thidiazuron in the stimulation of somatic embryogenesis in key members of the Restionaceae. Aust J Bot 52: 1–9; 2004b.
Rossetto M.; Dixon K. D.; Meney K. A.; Bunn E. In vitro propagation of Chinese puzzle (Caustis dioica Cyperaceae)—a commercial sedge species from Western Australia. Plant Cell Tiss Org Cult 30: 65–67; 1992.
Ward S. C.; Koch J. M.; Ainsworth G. L. The effect of timing of rehabilitation procedures on the establishment of a jarrah forest after bauxite mining. Res Ecol 4: 19–24; 1996.
Willyams D. Tissue culture of geophytic rush and sedge species for revegetation of bauxite mine sites in the northern jarrah forest of Western Australia. Conference proceedings: Contributing to a sustainable future. In: Bennett I. J.; Bunn E.; Clarke H.; McComb J. A. (eds) Proceedings of the Australian Branch of the IAPTC&B, Perth, Western Australia, School of Plant Biology. University of Western Australia, Perth, pp 21–24; 2005.
Zimmermann J. L. Somatic embryogenesis: A model for early development in higher plants. The Plant Cell 5: 1411–1423; 1993.
Acknowledgements
The authors thank the Australian Research Council and Worsley Alumina Pty Ltd for providing funds for this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Anna Pretov
Rights and permissions
About this article
Cite this article
Panaia, M., Bunn, E. & McComb, J. Primary and repetitive secondary somatic embryogenesis of Lepidosperma drummondii (Cyperaceae) and Baloskion tetraphyllum (Restionaceae) for land restoration and horticulture. In Vitro Cell.Dev.Biol.-Plant 47, 379–386 (2011). https://doi.org/10.1007/s11627-010-9335-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-010-9335-2