Abstract
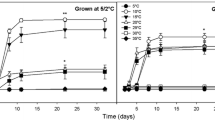
Seed responses to temperature are often essential to the study of germination ecology, but the ecological role of temperature in orchid seed germination remains uncertain. The response of orchid seeds to cold stratification have been studied, but the exact physiological role remains unclear. No studies exist that compare the effects of either cold stratification or temperature on germination among distant populations of the same species. In two separate experiments, the role of temperature (25, 22/11, 27/15, 29/19, 33/24°C) and chilling at 10°C on in vitro seed germination were investigated using distant populations of Calopogon tuberosus var. tuberosus. Cooler temperatures promoted germination of Michigan seeds; warmer temperatures promoted germination of South Carolina and north central Florida seeds. South Florida seed germination was highest under both warm and cool temperatures. More advanced seedling development generally occurred at higher temperatures with the exception of south Florida seedlings, in which the warmest temperature suppressed development. Fluctuating diurnal temperatures were more beneficial for germination compared to constant temperatures. Cold stratification had a positive effect on germination among all populations, but South Carolina seeds required the longest chilling treatments to obtain maximum germination. Results from the cold stratification experiment indicate that a physiological dormancy is present, but the degree of dormancy varies across the species range. The variable responses among populations may indicate ecotypic differentiation.




Similar content being viewed by others
References
Allen P. S.; Meyer S. E. Ecological aspects of seed dormancy loss. Seed Sci. Res. 8: 183–191; 1998.
Ballard W. W. Sterile propagation of Cypripedium reginae from seeds. Am. Orchid Soc. Bull. 56: 935–946; 1987.
Baskin C. C.; Baskin J. M. Seeds: ecology, biogeography, and evolution of dormancy and germination. Academic, San Diego; 2001.
Baskin C. C.; Baskin J. M. Determining dormancy-breaking and germination requirements from the fewest seeds. In: Guerrant E. O.; Havens K.; Maunder M. (eds) Ex situ plant conservation: supporting species survival in the wild. Island Press, Washington, pp 162–179; 2004.
Baskin C. C.; Thompson K.; Baskin J. M. Mistakes in germination ecology and how to avoid them. Seed Sci. Res. 16: 165–168; 2006.
Bewley J. D.; Black M. Seeds: physiology of development and germination. Plenum, New York; 1994.
Brundrett M. C.; Scade A.; Batty A. L.; Dixon K. W.; Sivasithamparam K. Development of in situ and ex situ seed baiting techniques to detect mycorrhizal fungi from terrestrial orchid habitats. Mycol. Res. 107: 1210–1220; 2003.
Chu C. C.; Mudge K. W. Effects of prechilling and liquid suspension culture on seed germination of the yellow lady's slipper orchid (Cypripedium calceolus var. pubescens). Lindleyana 9: 153–159; 1994.
De Pauw M. A.; Remphrey W. R. In vitro germination of three Cypripedium species in relation to time of seed collection, media, and cold treatment. Can. J. Bot. 71: 879–885; 1993.
Diez J. M. Hierarchical patterns of symbiotic orchid germination linked to adult proximity and environmental gradients. J. Ecol. 95: 159–170; 2007.
Donohue K. Seeds and seasons: interpreting germination timing in the field. Seed Sci. Res. 15: 175–187; 2005.
Fowler D. P.; Dwight T. W. Provenance differences in the stratification requirements of white pine. Can. J. Bot. 42: 669–675; 1964.
Goldman D. H.; Jansen R. K.; van den Berg C.; Leitch I. J.; Fay M. F.; Chase M. W. Molecular and cytological examination of Calopogon (Orchidaceae, Epidendroideae): circumscription, phylogeny, polyploidy, and possible hybrid speciation. Am. J. Bot. 91: 707–723; 2004a.
Goldman D. H.; van den Berg C.; Griffith M. P. Morphometric circumscription of species and infraspecific taxa in Calopogon R. Br. (Orchidaceae). Plant Syst. Evol. 247: 37–60; 2004b.
Harvais G. Growth requirements and development of Cypripedium reginae in axenic culture. Can. J. Bot. 51: 327–332; 1973.
Hidayati S. N.; Baskin C. C.; Baskin J. M. Dormancy-breaking and germination requirements of seeds of four Lonicera species (Caprifoliaceae) with underdeveloped spatule embryos. Seed Sci. Res. 10: 459–469; 2000.
Hufford K. M.; Mazer S. J. Plant ecotypes: genetic differentiation in the age of ecological restoration. Trends Ecol. Evol. 18: 147–155; 2003.
Kauth P. J.; Dutra D.; Johnson T. R.; Stewart S. L.; Kane M. E.; Vendrame W. A. Techniques and applications of in vitro orchid seed germination. In: Teixeira da Silva J. A. (ed) Floriculture, ornamental and plant biotechnology: advances and topical issues. Global Science Books, Isleworth, pp 375–391; 2008a.
Kauth P. J.; Kane M. E. In vitro ecology of Calopogon tuberosus var. tuberosus (Orchidaceae) seedlings from distant populations: implications assessing for ecotypic differentiation. J. Torrey Bot. Soc. 136: 433–444; 2009.
Kauth P. J.; Kane M. E.; Vendrame W. A.; Reinhardt-Adams C. Asymbiotic germination response to photoperiod and nutritional media in six populations of Calopogon tuberosus var. tuberosus (Orchidaceae): evidence for ecotypic differentiation. Ann. Bot. 102: 783–793; 2008b.
Kauth P. J.; Vendrame W. A.; Kane M. E. In vitro seed culture and seedlings development of Calopogon tuberosus. Plant Cell Tissue Organ Cult. 85: 91–102; 2006.
Lauzer D.; Renaut S.; St-Arnaud M.; Barabe D. In vitro asymbiotic germination, protocorm development, and plantlet acclimatization of Aplectrum hyemale (Muhl. ex Willd.) Torr. (Orchidaceae). J. Torrey Bot. Soc. 134: 344–348; 2007.
Lauzer D.; St-Arnaud M.; Barabe D. Tetrazolium staining and in vitro germination of mature seeds of Cypripedium acaule (Orchidaceae). Lindleyana 9: 197–204; 1994.
Luer C. A. The native orchids of Florida. The New York Botanical Garden, New York; 1972.
McKay J. K.; Christian C. E.; Harrison S.; Rice K. J. “How local is local?”—a review of practical and conceptual issues in the genetics of restoration. Restor. Ecol. 13: 432–440; 2005.
Meyer S. E. Habitat correlated variation in firecracker penstemon (Penstemon eatonii Gray: Scrophulariaceae) seed germination response. Bull. Torrey Bot. Club 119: 268–279; 1992.
Meyer S. E.; Kitchen S. G.; Carlson S. L. Seed germination timing patterns in intermountain Penstemon (Scrophulariaceae). Am. J. Bot. 82: 377–389; 1995.
Miyoshi K.; Mii M. Stimulatory effects of sodium and calcium hypochlorite, pre-chilling and cytokinins on the germination of Cypripedium macranthos seed in vitro. Physiol. Plant. 102: 481–486; 1998.
Mweetwa A. M.; Welbaum G. E.; Tay D. Effects of development, temperature, and calcium hypochlorite treatment on in vitro germinability of Phalaenopsis seeds. Sci. Hortic. 117: 257–262; 2008.
Nakamura S. I. Einige experimente zur samenkeimung einer chlorophyllfreien Erodorchidee Galeola septentrionalis Reichb. f. Mem. Coll. Agric., Kyoto Univ. 86: 1–48; 1964.
Øien D. I.; O'Neill J. P.; Whigham D. F.; McCormick M. K. Germination ecology of the boreal-alpine terrestrial orchid Dactylorhiza lapponica (Orchidaceae). Ann Bot Fenn 45: 161–172; 2008.
Otero J. T.; Ackerman J. D.; Bayman P. Differences in mycorrhizal preferences between two tropical orchids. Mol. Ecol. 13: 2393–2404; 2004.
Probert R. J.; Smith R. D.; Birch P. Germination responses to light and alternating temperatures in European populations of Dactylis glomerata L. I. variability in relation to origin. New Phytol. 99: 305–316; 1985.
Probert R. J.; Smith R. D.; Birch P. Germination responses to light and alternating temperatures in European populations of Dactylis glomerata L.5. The principle components of the alternating temperature requirement. New Phytol. 102: 133–142; 1986.
Rasmussen H. N. Seed dormancy patterns in Epipactis palustris (Orchidaceae): requirements for germination and establishment of mycorrhiza. Physiol. Plant. 86: 161–167; 1992.
Rasmussen H. N. Terrestrial orchids from seed to mycotropic plant. Cambridge University Press, Cambridge; 1995.
Rasmussen H. N.; Andersen T. F.; Johansen B. Temperature sensitivity of in vitro germination and seedling development of Dactylorhiza majalis (Orchidaceae) with and without a mycorrhizal fungus. Plant Cell Environ. 13: 171–177; 1990.
Rasmussen H. N.; Rasmussen F. N. Climactic and seasonal regulation of seed plant establishment in Dactylorhiza majalis inferred from symbiotic experiments in vitro. Lindleyana 5: 221–227; 1991.
Roberts E. H.; Benjamin S. K. The interaction of light, nitrate and alternating temperature on the germination of Chenopodium album, Capsella bursa-pastoris and Poa annua before and after chilling. Seed Sci. Technol. 7: 379–392; 1979.
SAS Institute. SAS for Windows, version 9.1. Cary, NC, USA; 2003.
Schütz W.; Milberg P. Seed dormancy in Carex canescens: regional differences and ecological consequences. Oikos 78: 420–428; 1997.
Seneca E. D. Germination and seedling response of Atlantic and Gulf coast populations of Uniola paniculata. Am. J. Bot. 59: 290–296; 1972.
Seneca E. D. Germination and seedling response of Atlantic and Gulf coasts populations of Spartina alterniflora. Am. J. Bot. 61: 947–956; 1974.
Shimura H.; Koda Y. Micropropagation of Cypripedium macranthos var. rebunense through protocorm-like bodies derived from mature seeds. Plant Cell Tissue Organ Cult. 78: 273–276; 2004.
Shimura H.; Koda Y. Enhanced symbiotic seed germination of Cypripedium macranthos var. rebunense following inoculation after cold treatment. Physiol. Plant. 123: 281–287; 2005.
Singh K. P. Effect of temperature and light on seed germination of two ecotypes of Portulaca oleracea L. New Phytol. 72: 289–295; 1973.
Stoutamire W. P. Terrestrial orchid seedlings. In: Withner C. L. (ed) The orchids: scientific studies. Wiley, New York, pp 101–128; 1974.
Thompson K.; Grime J. P. A comparative study of germination responses to diurnally fluctuating temperatures. J. Appl. Ecol. 20: 141–156; 1983.
Thompson K.; Grime J. P.; Mason G. Seed germination in response to diurnal fluctuations of temperature. Nature 267: 147–149; 1977.
Tomita M.; Tomita M. Effects of culture media and cold treatment on germination in asymbiotic culture of Cypripedium macranthos and Cypripedium japonica. Lindleyana 12: 208–213; 1997.
Toole E. H.; Toole V. K.; Borthwick H. A.; Hendricks S. B. Interaction of temperature and light in germination of seeds. Plant Physiol. 30: 473–478; 1955.
Trapnell D. W.; Hamrick J. L.; Giannasi D. E. Genetic variation and species boundaries in Calopogon (Orchidaceae). Syst. Bot. 29: 308–315; 2004.
van Assche J. A.; Debucquoy K. L. A.; Rommens W. A. F. Seasonal cycles in the germination capacity of buried seeds of some Leguminosae (Fabaceae). New Phytol. 158: 315–323; 2003.
van Assche J. A.; Vanlerberghe K. A. The role of temperature on the dormancy cycle of seeds of Rumex obtusifolius L. Funct. Ecol. 3: 107–115; 1989.
van Waes J. M.; Debergh P. C. In vitro germination of some Western European orchids. Physiol. Plant. 67: 253–261; 1986.
Vujanovic V.; St-Arnaud M.; Barabe D.; Thibeault G. Viability testing of orchid seed and the promotion of colouration and germination. Ann. Bot. 86: 79–86; 2000.
Zettler L. W.; Piskin K. A.; Stewart S. L.; Hartsock J. J.; Bowles M. L.; Bell T. J. Protocorm mycobionts of the federally threatened eastern prairie fringed orchid, Platanthera leucophaea (Nutt.) Lindley, and a technique to prompt leaf elongation in seedlings. Stud. Mycol. 53: 163–171; 2005.
Zettler L. W.; Stewart S. L.; Bowles M. L.; Jacobs K. A. Mycorrhizal fungi and cold-assisted symbiotic germination of the federally threatened eastern prairie fringed orchid, Platanthera leucophaea (Nuttall) Lindley. Am. Midl. Nat. 145: 168–175; 2001.
Acknowledgments
We thank the following for collecting seed: Larry Richardson (Florida Panther National Wildlife Refuge, Naples, FL), Jim Fowler (South Carolina populations), and Kip Knudson (Michigan population). We also thank Mary Bunch (South Carolina Heritage Preserve Program) for issuing collection permits. We thank Dr. Hector Perez (University of Florida, Gainesville, FL) for use of growth chambers. We also thank Timothy Johnson for providing insightful comments regarding the manuscript. Brand names are provided as references; the authors do not solely recommend or endorse these products. US Fish and Wildlife and the Florida Panther National Wildlife Refuge provided partial funding.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: P. Lakshmanan
Rights and permissions
About this article
Cite this article
Kauth, P.J., Kane, M.E. & Vendrame, W.A. Comparative in vitro germination ecology of Calopogon tuberosus var. tuberosus (Orchidaceae) across its geographic range. In Vitro Cell.Dev.Biol.-Plant 47, 148–156 (2011). https://doi.org/10.1007/s11627-010-9316-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-010-9316-5