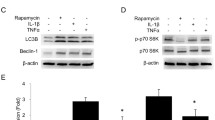
Summary
Endothelial cells assume divergent physiologic postures, from a ‘quiescent’ to an ‘activated’ state, and this ability is thought to critically regulate homeostasis of the vascular milieu. While establishing endothelial cell lines, we observed that most of the endothelial cells exhibited ‘spontaneous’ activation with morphological changes once the cells surpassed confluence. In the present study, we investigated whether accumulation and subsequent availability of certain factors in the cells might induce morphologic changes indicating activated phenotype in confluent endothelial cells. Cell lysate from nonactivated confluent endothelial cells activated autologous target cells, whereas culture supernatants did not. Stimulatory activity of the cell lysate was dependent on the concentration of cell lysate, i.e., nonactivated endothelial cell lysate at 25% concentration induced a substantial level of morphologic change. The ‘spontaneous’ as well as the cell lysate-induced change in morphology of endothelial cells was inhibited by neutralization of interleukin (IL)-1α with anti-IL-1α antibody. Correspondingly, cell lysate from confluent non-IL-1-expressing endothelial cells did not alter the morphology of autologous confluent cells even at a higher concentration. Cells that spontaneously changed their morphology correspondingly upregulated IL-1α and IL-8 transcripts. The results indicate that the accumulation and availability of endogenous IL-1α is responsible for the ‘spontaneous’ activation of endothelial cells in culture. Ultimately, through this property, endothelial cells may facilitate normal in vivo host responses, including the regulation of leukocyte trafficking.
Similar content being viewed by others
References
Albrightson, C. R.; Baenziger, N. L.; Needleman, P. Exaggerated human vascular cell prostaglandin biosynthesis mediated by monocytes: role of monokines and interleukin-1. J. Immunol. 135:1872–1877; 1985.
Anklesaria, P.; Teixido, M.; Laiho, M., et al. Cell-cell adhesion mediated by binding of membrane-anchored transforming growth factor-α to epidermal growth factor receptors promotes cell proliferation. Proc. Natl. Acad. Sci. USA 87:3289–3293; 1990.
Bakouche, O.; Moreau, J.-L.; Lachman, L. B. Acylation of cell-associated IL-1 by palmitic acid. J. Immunol. 147:2164–2169; 1991.
Bevilacqua, M. P.; Pober, J. S.; Wheeler, R. S., et al. Interleukin-1 acts on cultured human vascular endothelium to increase the adhesion of polymorphonuclear leukocytes, monocytes and related leukocyte cell lines. J. Clin. Invest. 76:2003–2011; 1985.
Cavender, D. E.; Haskard, D. O.; Joseph, B., et al. Interleukin-1 increases the binding of human B and T lymphocytes to endothelial cell monolayers. J. Immunol. 136:203–207; 1986.
Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162:156–159; 1987.
Cotran, R. S. New roles for the endothelium in inflammation and immunity. American Association of Pathologists president’s address. Am. J. Pathol. 129:407–413; 1987.
Cozzolino, F.; Torcia, M.; Aldinicci, D., et al. Interleukin-1 is an autocrine regulator of human endothelial cell growth. Proc. Natl. Acad. Sci. USA 87:6487–6491; 1990.
Dinarello, C. A. Interleukin-1 and its biologically related cytokines. Adv. Immunol. 44:153–205; 1989.
Dinarello, C. A. Interleukin-1 and Interleukin-1 antagonism. Blood 77:1627–1652; 1991.
Dinarello, C. A.; Ikejima, T.; Warner, S. J. C., et al. Interleukin-1 induces interleukin-1: induction of circulating interleukin-1 in rabbits in vivo and in human mononuclear cells in vitro. J. Immunol. 139:1902–1910; 1987.
Emeis, J. J.; Kooistra, T. Interleukin-1 and lipopolysaccharide induce an inhibitor of tissue-type plasminogen activator in vivo and in cultured endothelial cells. J. Exp. Med. 163:1260–1266; 1986.
Foreman, K. E.; Vaporciyan, A. A.; Bonish, B. A., et al. C5a-induced expression of P-selectin in endothelial cells. J. Clin. Invest. 94:1147–1155; 1994.
Gamble, J. R.; Harlan, J. M.; Klebanoff, S. J., et al. Stimulation of the adherence of neutrophils to umbilical vein endothelium by human recombinant tumor necrosis factor. Proc. Natl. Acad. Sci. USA 82:8667–8671; 1985.
Gimbrone, M. A., Jr. Vascular endothelium in hemostasis and thrombosis. Gimbrone, M. A., Jr., ed. Edinburgh: Churchill Livingstone; 1986:1–13.
Jaffe, E. A.; Nachman, R. L.; Becker, C. G., et al. Culture of human endothelial cells derived from umbilical veins: identification by morphologic and immunologic criteria. J. Clin. Invest. 52:2745–2756; 1973.
Kaplanski, G.; Farnarier, C.; Kaplanski, S., et al. Interleukin-1 induces interleukin-8 secretion from endothelial cells by a juxtacrine mechanism. Blood 84:4242–4248; 1994.
Kilgore, K. S.; Shen, J. P.; Miller, B. F., et al. Enhancement of the complement membrane attack complex of tumor necrosis factor-α-induced endothelial cell expression of E-selectin and ICAM-1. J. Immunol. 155:1434–1441; 1995.
Kurt-Jones, E. A.; Fiers, W.; Pober, J. S. Membrane interleukin-1 induction on human endothelial cells and dermal fibroblasts. J. Immunol. 139:2317–2324; 1987.
Loppnow, H.; Libby, P. Functional significance of human vascular smooth muscle cell-derived interleukin-1 in paracrine and autocrine regulation pathways. Exp. Cell Res. 198:283–290; 1992.
Lorant, D. E.; Patel, K. D. A.; McIntyre, T. M., et al. Coexpression of GMP-140 and platelet-activating factor by endothelium stimulated by histamine or thrombin: a juxtacrine system for adhesion and activation of neutrophils. J. Cell Biol. 115:223–234; 1991.
Maier, J. A.; Voulalas, P.; Roeder, D. Extension of the life-span of human endothelial cells by an interleukin-1 alpha antisense oligomer. Science (Wash DC) 249:1570–1574; 1990.
Massagué, J. Transforming growth factor-α: a model for membrane-anchored growth factor. J. Biol. Chem. 265:21393–21396; 1990.
Miossec, P.; Cavender, D.; Ziff, M. Production of interleukin-1 by human endothelial cells. J. Immunol. 136:2486–2491; 1986.
Nawroth, P. P.; Handley, D. A.; Esmon, C. T., et al. Interleukin-1 induces endothelial cell procoagulant while suppressing cell-surface anticoagulant activity. Proc. Natl. Acad. Sci. USA 83:3460–3464; 1986.
Pober, J. S. Cytokine-mediated activation of vascular endothelium: physiology and pathology. Am. J. Pathol. 133:426–433; 1988.
Pober, J. S.; Bevilacqua, M. P.; Mendrick, D. L., et al. Two distinct monokines, interleukin-1 and tumor necrosis factor, each independently induce biosynthesis and transient expression of the same antigen on the surface of cultured human vascular endothelial cells. J. Immunol. 136:1680–1687; 1986.
Pober, J. S.; Cotran, R. S. The role of endothelial cells in inflammation. Transplantation (Baltimore) 50:537–544; 1990.
Pober, J. S.; Lapierre, L. A.; Stolpen, A. H., et al. Activation of cultured human endothelial cells by recombinant lymphotoxin: comparison with tumor necrosis factor and interleukin-1 species. J. Immunol. 138:3319–3324; 1987.
Qi, J.; Kreutzer, D. L. Fibrin activation of vascular endothelial cells: induction of IL-8 expression. J. Immunol. 155:867–876; 1995.
Schleimer, R. P.; Rutledge, B. K. Cultured human vascular endothelial cells acquire adhesiveness for neutrophils after stimulation with interleukin-1, endotoxin and tumor-promoting phorbol diesters. J. Immunol. 136:649–654; 1986.
Selvan, R. S.; Butterfield, J. H.; Krangel, M. S. Expression of multiple chemokine genes by human mast cell leukemia. J. Biol. Chem. 269:13893–13898; 1994.
Selvan, R. S.; Kapadia, H. B.; Platt, J. L. Complement-induced expression of chemokine genes in endothelium: regulation by IL-1-dependent and -independent mechanisms. J. Immunol. 161:4388–4395; 1998.
Sironi, M.; Breviario, F.; Proserpio, P., et al. IL-1 stimulates IL-6 production in endothelial cells. J. Immunol. 142:549–553; 1989.
Skalli, O.; Ropraz, P.; Trzeciak, A. A monoclonal antibody against alpha-smooth muscle actin: a new probe for smooth muscle differentiation. J. Cell Biol. 103(6 Pt 2):2787–2796; 1986.
Stevenson, F. T.; Burstein, S. L.; Fanton, C., et al. The 31-kDa precursor of interleukin-1α is myristoylated on specific lysines within the 16-kDa N-terminal propiece. Proc. Natl. Acad. Sci. USA 90:7245–7249; 1993.
Strieter, R. M.; Kunkel, S. L.; Showell, H. G., et al. Endothelial cell gene expression of a neutrophil chemotactic factor by TNF-alpha, LPS, and IL-1 beta. Science (Wash DC) 243:1467–1469; 1989.
Warner, S. J. C.; Auger, K. R.; Libby, P. Interleukin-1 induces interleukin-1. II: recombinant human interleukin-1 induces interleukin-1 production by adult human vascular endothelial cells. J. Immunol. 139:1911–1917; 1987.
Yu, C. L.; Haskard, D. O.; Cavender D, et al. Human gamma interferon increases the binding of T lymphocytes to endothelial cells. Clin. Exp. Immunol. 62:554–560; 1985.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Steen, M.B., Tuck, F.L. & Selvan, R.S. Spontaneous activation of endothelial cells: A central role for endogenous IL-1α. In Vitro Cell.Dev.Biol.-Animal 35, 327–332 (1999). https://doi.org/10.1007/s11626-999-0082-9
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11626-999-0082-9