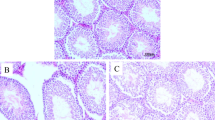
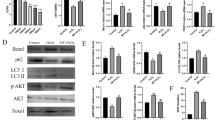
Abstract
Zinc deficiency has a huge impact on male reproduction. The zinc transporter (ZnT) family is involved in the maintenance of zinc homeostasis and testosterone synthesis. However, the underlying mechanisms remain to be investigated. Therefore, in this study, we aimed to determine the effect of zinc transporter 4 (ZnT4) on testosterone synthesis in male Kunming mice and mouse Leydig cells. The results of this study showed that compared with the zinc normal diet group (Con group), the zinc-deficient diet group (ZnD group) had decreased zinc content and increased ZnT4 expression in testicular tissues, and decreased serum testosterone levels, suggesting that ZnT4 may be involved in Leydig cell injury resulting from a zinc-deficient diet. Subsequently, mouse Leydig cell line TM3 cells were used to analyze the effect of ZnT4 downregulation on TM3 cell proliferation and apoptosis, on testosterone synthesis, and its underlying mechanisms. Here, we show that knockdown of ZnT4 can induce the accumulation of zinc, inhibit the viability, and induce apoptosis in TM3 cells. In addition, knockdown of ZnT4 downregulated testosterone concentration and expression of testosterone synthesis–related proteins steroidogenic acute regulatory protein (StAR) and 3β-hydroxysteroid dehydrogenase/D5-D4 isomerase (3β-HSD) in TM3 cells, while hCG could rescue their levels. We show that it is ZnT4 that plays a role in testosterone production through a mediated PI3K/Akt/mTOR autophagy pathway, whereas mTORC1 complex inhibitor (Rapa) blocks the decrease in testosterone levels caused by ZnT4 downregulation. In conclusion, the above results indicate that ZnT4 plays an important role in regulating testosterone synthesis.







Similar content being viewed by others
Data availability
Data supporting the findings of this study are available from the respective authors upon reasonable request.
References
Beigi Harchegani A, Dahan H, Tahmasbpour E, Bakhtiari Kaboutaraki H, Shahriary A (2020) Effects of zinc deficiency on impaired spermatogenesis and male infertility: the role of oxidative stress, inflammation and apoptosis. Hum Fertil (Camb) 23(1):5–16
Bose HS, Lingappa VR, Miller WL (2002) The steroidogenic acute regulatory protein, StAR, works only at the outer mitochondrial membrane. Endocr Res 28(4):295–308
Cavalieri J, Wang M (2015) Chemical sterilisation of Bos indicus bull calves following intratesticular injection of zinc acetate: effects on growth and concentrations of testosterone. Anim Reprod Sci 159:163–171
Chu Q, Chi ZH, Zhang X, Liang D, Wang X, Zhao Y, Zhang L, Zhang P (2016) A potential role for zinc transporter 7 in testosterone synthesis in mouse Leydig tumor cells. Int J Mol Med 37(6):1619–1626
Ding H, Zhao J, Liu H, Wang J, Lu W (2020) BMAL1 knockdown promoted apoptosis and reduced testosterone secretion in TM3 Leydig cell line. Gene 15(747):144672
Egwurugwu JN, Ifedi CU, Uchefuna RC, Ezeokafor EN, Alagwu EA (2013) Effects of zinc on male sex hormones and semen quality in rats. Niger J Physiol Sci 28(1):17–22
Ewing LL, Wing TY, Cochran RC, Kromann N, Zirkin BR (1983) Effect of luteinizing hormone on Leydig cell structure and testosterone secretion. Endocrinology. 112(5):1763–1769
Fang J, Li DH, Yu XQ et al (2016) Formaldehyde exposure inhibits the expression of mammalian target of rapamycin in rat testis. Toxicol Ind Health 32(11):1882–1890
Gao F, Li G, Liu C et al (2018) Autophagy regulates testosterone synthesis by facilitating cholesterol uptake in Leydig cells. J Cell Biol 217(6):2103–2119
Gao H, Liu C, Li W (2019) Assessing autophagy in the Leydig cells. Methods Mol Biol 1854:71–85
González CR, Muscarsel Isla ML, Vitullo AD (2018) The balance between apoptosis and autophagy regulates testis regression and recrudescence in the seasonal-breeding South American plains vizcacha, Lagostomus maximus. PLoS One 13(1):e0191126
Gu YP, Yang XM, Luo P et al (2017) Inhibition of acrolein-induced autophagy and apoptosis by a glycosaminoglycan from Sepia esculenta ink in mouse Leydig cells. Carbohydr Polym 163:270–279
Heidelbaugh JJ (2016) Endocrinology update: testicular hypogonadism. FP Essent 451:31–41
Huang L, Gitschier J (1997) A novel gene involved in zinc transport is deficient in the lethal milk mouse. Nat Genet 17(3):292–297
Huang T, Yan G, Guan M (2020) Zinc homeostasis in bone: zinc transporters and bone diseases. Int J Mol Sci 21(4):1236
Kambe T, Yamaguchi-Iwai Y, Sasaki R, Nagao M (2004) Overview of mammalian zinc transporters. Cell Mol Life Sci 61(1):49–68
Kukic I, Lee JK, Coblentz J et al (2013) Zinc-dependent lysosomal enlargement in TRPML1-deficient cells involves MTF-1 transcription factor and ZnT4 (Slc30a4) transporter. Biochem J 451(2):155–163
Liuzzi JP, Narayanan V, Doan H, Yoo C (2018) Effect of zinc intake on hepatic autophagy during acute alcohol intoxication. Biometals 31(2):217–232
Michalczyk AA, Allen J, Blomeley RC et al (2002) Constitutive expression of hZnT4 zinc transporter in human breast epithelial cells. Biochem J 364(Pt 1):105–113
Midzak AS, Chen H, Papadopoulos V, Zirkin BR (2009) Leydig cell aging and the mechanisms of reduced testosterone synthesis. Mol Cell Endocrinol 299(1):23–31
Murgia C, Espignani V, Cerase J et al (1999) Cloning, expression, and vesicular localization of zinc transporter Dri 27/ZnT4 in intestinal tissue and cells. Am J Phys 277(6):G1231–G1239
Nguyen TT, Trieu TS, Tran TO, Luong TLA (2019) Evaluation of sperm DNA fragmentation index, zinc concentration and seminal parameters from infertile men with varicocele. Andrologia 51(2):e13184
Ni H, Feng X, Xiao ZJ, Tao LY, Jin MF (2011) Dynamic pattern of gene expression of ZnT-4, caspase-3, LC3, and PRG-3 in rat cerebral cortex following flurothyl-induced recurrent neonatal seizures. Biol Trace Elem Res 143(3):1607–1615
Omu AE, Al-Azemi MK, Al-Maghrebi M, Mathew CT, Omu FE, Kehinde EO, Anim JT, Oriowo MA, Memon A (2015) Molecular basis for the effects of zinc deficiency on spermatogenesis: an experimental study in the Sprague-Dawley rat model. Indian J Urol 31(1):57–64
Quan C, Wang C, Duan P et al (2017) Bisphenol A induces autophagy and apoptosis concurrently involving the Akt/mTOR pathway in testes of pubertal SD rats. Environ Toxicol 32(8):1977–1989
Raucci F, D’Aniello A, Di Fiore MM (2014) Stimulation of androgen production by D-aspartate through the enhancement of StAR, P450scc and 3β-HSD mRNA levels in vivo rat testis and in culture of immature rat Leydig cells. Steroids 84:103–110
Stocco DM, Clark BJ (1996) Regulation of the acute production of steroids in steroidogenic cells. Endocr Rev 17(3):221–244
Su Y, Quan C, Li X, Shi Y, Duan P, Yang K (2018) Mutual promotion of apoptosis and autophagy in prepubertal rat testes induced by joint exposure of bisphenol A and nonylphenol. Environ Pollut 243(Pt A):693–702
Sun B, Ma J, Te L, Zuo X, Liu J, Li Y, Bi J, Wang S (2023) Zinc-deficient diet causes imbalance in zinc homeostasis and impaired autophagy and impairs semen quality in mice. Biol Trace Elem Res 201(5):2396–2406
Yang JY, Zhang YF, Li YX, Guan GP, Kong XF, Liang AM, Ma KW, Da Li G, Bai XF (2016) Effects of T-2 toxin on the regulation of steroidogenesis in mouse Leydig cells. Toxicol Ind Health 32(10):1801–1807
Zhang X, Guan T, Yang B, Chi Z, Wang ZY, Gu HF (2018) A novel role for zinc transporter 8 in the facilitation of zinc accumulation and regulation of testosterone synthesis in Leydig cells of human and mouse testicles. Metabolism 88:40–50
Acknowledgements
Heartfelt thanks to all the members of the Key Laboratory of Reproductive Medicine of Hebei Reproductive Health Hospital (Hebei Reproductive Health Research Institute).
Funding
This study was funded by the Hebei Natural Science Foundation (Grant No. H2021314001; No. H2022314001); Hebei Medical Science Research Project Plan (20231201).
Author information
Authors and Affiliations
Contributions
Huanhuan Li, Yuejia Li, Junsheng Liu, Xuan Liu, and Yuanjing Li collected samples and performed experiments. Huanhuan Li and Jing Ma analyzed the data and wrote the manuscript. Jing Ma and Shusong Wang conceived the idea, designed the study, raised funding, and revised the manuscript. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, H., Li, Y., Liu, J. et al. Knockdown of ZnT4 Induced Apoptosis, Inhibited Proliferation and testosterone synthesis of TM3 cells. In Vitro Cell.Dev.Biol.-Animal 59, 565–574 (2023). https://doi.org/10.1007/s11626-023-00804-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-023-00804-z