Abstract
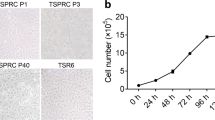
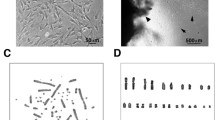
Common bottlenose dolphin (Tursiops truncatus) is a well-known cetacean species that inhabits temperate and tropical seas worldwide. Limited supply and poor quality of samples hinder the investigation of the effects of various pathogens and environmental pollutants on this cetacean species. Cultured cells are useful for experimental studies; however, no cell lines derived from cetaceans are generally available. Therefore, in this study, we established a novel kidney cell line, TK-ST, derived from T. truncatus. Primary cells exhibited the morphological characteristics of epithelial and fibroblast cells, but their immortalization and passaging resulted in a predominantly epithelial cell morphology. TK-ST was immortalized using the large T SV40 antigen and human telomerase reverse transcriptase and exhibited long-term stable cell growth. TK-ST cells are generally cultured in Dulbecco’s modified Eagle’s medium with 10% fetal bovine serum at 37°C and 5% CO2 but can also be cultured in 5–20% fetal bovine serum and several other classical media commonly used for common animal cell culture. TK-ST cells were found to be susceptible to several viruses, including the dolphin morbillivirus (most important virus in cetaceans), and exhibited cytopathic effects, facilitating the replication of the dolphin morbillivirus. Furthermore, mRNA expression levels of cytokine genes were increased in TK-ST cells after stimulation with lipopolysaccharides and poly(I:C). Therefore, the novel TK-ST cell line derived in this study can potentially be used for further in vitro studies on cetaceans.









Similar content being viewed by others
References
Alexopoulou L, Holt AC, Medzhitov R, Flavell RA (2001) Recognition of double-stranded RNA and activation of NF-κB by toll-like receptor 3. Nature 413:732–738
Ayral AM, Clarkson S, Cheeseman M, Wells S, Dear TN (2006) A panel of optimized primers and positive-control DNAs for PCR detection of common biological contaminants in mouse cell lines and tissue samples. Lab Anim (NY) 35(8):31–36. https://doi.org/10.1038/laban0906-31
Barrett T, Visser IKG, Mamaev L, Goatley L, Van Bressem M-F, Osterhaus A (1993) Dolphin and porpoise morbilliviruses are genetically distinct from phocine distemper virus. Virology 193(2):1010–1012. https://doi.org/10.1006/viro.1993.1217
Beineke A, Siebert U, Wohlsein P, Baumgärtner W (2010) Immunology of whales and dolphins. Vet Immunol Immunopathol 133:81–94. https://doi.org/10.1016/j.vetimm.2009.06.019
Bodnar AG, Ouellette M, Frolkis M, Holt SE, Chiu CP, Morin GB, Harley CB, Shay JW, Lichtsteiner S, Wright WE (1998) Extension of life-span by introduction of telomerase into normal human cells. Science (80- ) 279:349–352. https://doi.org/10.1126/science.279.5349.349
Bossart GD (2011) Marine mammals as sentinel species for oceans and human health. Vet Pathol 48:676–690. https://doi.org/10.1177/0300985810388525
Burkard M, Whitworth D, Schirmer K, Nash SB (2015) Establishment of the first humpback whale fibroblast cell lines and their application in chemical risk assessment. Aquat Toxicol 167:240–247. https://doi.org/10.1016/j.aquatox.2015.08.005
Carvan MJ, Santostefano M, Safe S, Busbee D (1994) Characterization of a bottlenose dolphin (Tursiops truncatus) kidney epithelial cell line. Mar Mam Sci 10(1):52–69. https://doi.org/10.1111/j.1748-7692.1994.tb00389.x
Cooper JK, Sykes G, King S, Cottrill K, Ivanova NV, Hanner R, Ikonomi P (2007) Species identification in cell culture: a two-pronged molecular approach. Vitr Cell Dev Biol - Anim 43:344–351. https://doi.org/10.1007/s11626-007-9060-2
Drexler HG, Uphoff CC (2002) Mycoplasma contamination of cell cultures: incidence, sources, effects, detection, elimination, prevention. Cytotechnology 39:75–90. https://doi.org/10.1023/A:1022913015916
Dulbecco R, Freeman G (1959) Plaque production by the polyoma virus. Virology 8(3):396–397. https://doi.org/10.1016/0042-6822(59)90043-1
Eagle H (1959) Amino acid metabolism in mammalian cell cultures. Science 130(3373):432–437. https://doi.org/10.1126/science.130.3373.432
Fridman AL, Tainsky MA (2008) Critical pathways in cellular senescence and immortalization revealed by gene expression profiling. Oncogene 27:5975–5987. https://doi.org/10.1038/onc.2008.213
Gough DJ, Messina NL, Clarke CJP, Johnstone RW, Levy DE (2012) Constitutive type I interferon modulates homeostatic balance through tonic signaling. Immunity 36(2):166–174. https://doi.org/10.1016/j.immuni.2012.01.011
Ham RG (1965) Clonal growth of mammalian cells in a chemically defined, synthetic medium. Proc Natl Acad Sci U S A 53(2):288–293. https://doi.org/10.1073/pnas.53.2.288
Harasawa R, Mizusawa H, Nozawa K, Nakagawa T, Asada K, Kato I (1993) Detection and tentative identification of dominant mycoplasma species in cell cultures by restriction analysis of the 16S–23S rRNA intergenic spacer regions. Res Microbiol 144:489–493. https://doi.org/10.1016/0923-2508(93)90057-9
Ishengoma E, Agaba M (2017) Evolution of toll-like receptors in the context of terrestrial ungulates and cetaceans diversification. BMC Evol Biol 17:54. https://doi.org/10.1186/s12862-017-0901-7
Iwasaki A, Medzhitov R (2004) Toll-like receptor control of the adaptive immune responses. Nat Immunol 5:987–995. https://doi.org/10.1038/ni1112
Jha KK, Banga S, Palejwala V, Ozer HL (1998) SV40-mediated immortalization. Exp Cell Res 245(1):1–7. https://doi.org/10.1006/excr.1998.4272
Jin W, Jia K, Yang L, Chen J, Wu Y, Yi M (2013) Derivation and characterization of cell cultures from the skin of the Indo-Pacific humpback dolphin Sousa chinensis. Vitr Cell Dev Biol - Anim 49:449–457. https://doi.org/10.1007/s11626-013-9611-7
Kawasaki T, Kawai T (2014) Toll-like receptor signaling pathways. Front Immunol 5. https://doi.org/10.3389/fimmu.2014.00461
Moore GE, Ito E, Ulrich K, Sandberg AA (1966) Culture of human leukemia cells. Cancer 19(5):713–723. https://doi.org/10.1002/1097-0142(196605)19:5%3C713::aid-cncr2820190518%3E3.0.co;2-y
Mühlebach MD, Mateo M, Sinn PL, Prüfer S, Uhlig KM, Leonard VHJ, Navaratnarajah CK, Frenzke M, Wong XX, Sawatsky B, Ramachandran S, McCray PB, Cichutek K, Von Messling V, Lopez M, Cattaneo R (2011) Adherens junction protein nectin-4 is the epithelial receptor for measles virus. Nature 480:530–533. https://doi.org/10.1038/nature10639
Nagasawa T, Uemori T, Asada K, Harasawa R (1992) Acholeplasma laidlawii has tRNA genes in the 16S–23S spacer of the rRNA operon. J Bacteriol 174(24):8163–8165. https://doi.org/10.1128/jb.174.24.8163-8165.1992
Nielsen O, Smith G, Weingartl H, Lair S, Measures L (2008) Use of a SLAM transfected Vero cell line to isolate and characterize marine mammal morbillivirus using an experimental ferret model. J Wildl Dis 44:600–611. https://doi.org/10.7589/0090-3558-44.3.600
Noyce RS, Bondre DG, Ha MN, Lin LT, Sisson G, Tsao MS, Richardson CD (2011) Tumor cell marker pvrl4 (nectin 4) is an epithelial cell receptor for measles virus. PLoS Pathog 7. https://doi.org/10.1371/journal.ppat.1002240
Ono K, Satoh M, Yoshida T, Ozawa Y, Kohara A, Takeuchi M, Mizusawa H, Sawada H (2007) Species identification of animal cells by nested PCR targeted to mitochondrial DNA. Vitr Cell Dev Biol - Anim 43:168–175. https://doi.org/10.1007/s11626-007-9033-5
Ono N, Tatsuo H, Hidaka Y, Aoki T, Minagawa H, Yanagi Y (2001) Measles viruses on throat swabs from measles patients use signaling lymphocytic activation molecule (CDw150) but not CD46 as a cellular receptor. J Virol 75:4399–4401. https://doi.org/10.1128/JVI.75.9.4399-4401.2001
Palumbi S, Martin A, Romano S, McMillan WO, Stice L, Grabowski G (1991) The simples fool’s guide to PCR. University of Hawaii, Honolulu
Parodi B, Aresu O, Bini D, Lorenzini R, Schena F, Visconti P, Cesaro M, Ferrera D, Andreotti V, Ruzzon T (2002) Species identification and confirmation of human and animal cell lines: a PCR-based method. Biotechniques 32:432–440. https://doi.org/10.2144/02322rr05
Pine M, Schroeder M, Greer K, Hokanson R, Busbee D (2004) Generation and partial characterization of a transformed cetacean cell line. Aquat Toxicol 67(2):195–202. https://doi.org/10.1016/j.aquatox.2004.01.003
Poltorak A, He X, Smirnova I, Liu M-Y, Huffel C Van, Du X, Birdwell D, Alejos E, Silva M, Galanos C, Freudenberg M, Ricciardi-Castagnoli P, Layton B, Beutler B (1998) Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science (80-. ) 282:2085–2088. https://doi.org/10.1126/science.282.5396.2085
Rosel PE, Dizon AE, Heyning JE (1994) Genetic analysis of sympatric morphotypes of common dolphins (genus Delphinus). Mar Biol 119:159–167. https://doi.org/10.1007/BF00349552
Seki F, Ono N, Yamaguchi R, Yanagi Y (2003) Efficient isolation of wild strains of canine distemper virus in Vero cells expressing canine SLAM (CD150) and their adaptability to Marmoset B95a cells. J Virol 77:9943–9950. https://doi.org/10.1128/JVI.77.18.9943-9950.2003
Sitt T, Bowen L, Blanchard MT, Smith BR, Gershwin LJ, Byrne BA, Stott JL (2008) Quantitation of leukocyte gene expression in cetaceans. Dev Comp Immunol 32:1253–1259. https://doi.org/10.1016/j.dci.2008.05.001
Suttle CA (2007) Marine viruses - major players in the global ecosystem. Nat Rev Microbiol 5:801–812. https://doi.org/10.1038/nrmicro1750
Suzuki M, Wakui H, Itou T, Segawa T, Inoshima Y, Maeda K, Kikuchi K (2016) Two isoforms of aquaporin 2 responsive to hypertonic stress in the bottlenose dolphin. J Exp Biol 219:1249–1258. https://doi.org/10.1242/jeb.132811
Takeda K, Akira S (2005) Toll-like receptors in innate immunity. Int Immunol 17:1–14. https://doi.org/10.1093/intimm/dxh186
Tatsuo H, Ono N, Tanaka K, Yanagi Y (2000) SLAM (CDw150) is a cellular receptor for measles virus. Nature 406:893–897. https://doi.org/10.1038/35022579
Tovey MG, Streulit M, Gressert I, Gugenheim J, Blanchardt B, Guymarhot J, Vignauxt F, Gigou M (1987) Interferon messenger RNA is produced constitutively in the organs of normal individuals. Proc Natl Acad Sci U S A 84:5038–5042. https://doi.org/10.1073/pnas.84.14.5038
Uphoff CC, Drexler HG (2014) Detection of mycoplasma contamination in cell cultures. Curr Protoc Mol Biol 106:1–14. https://doi.org/10.1002/0471142727.mb2804s106
Van Bressem M-F, Duignan P, Banyard A, Barbieri M, Colegrove K, De Guise S, Di Guardo G, Dobson A, Domingo M, Fauquier D, Fernandez A, Goldstein T, Grenfell B, Groch K, Gulland F, Jensen B, Jepson P, Hall A, Kuiken T, Mazzariol S, Morris S, Nielsen O, Raga J, Rowles T, Saliki J, Sierra E, Stephens N, Stone B, Tomo I, Wang J, Waltzek T, Wellehan J (2014) Cetacean morbillivirus: current knowledge and future directions. Viruses 6:5145–5181. https://doi.org/10.3390/v6125145
Viricel M, Rosel PE (2012) Evaluating the utility of cox1 for cetacean species identification. Mar Mamm Sci 28:37–62. https://doi.org/10.1111/j.1748-7692.2010.00460.x
Wells R, Rhinehart H, Hansen L, Sweeney J, Townsend F, Stone R, Casper DR, Scott M, Hohn A, Rowles T (2004) Bottlenose dolphins as marine ecosystem sentinels: developing a health monitoring system. EcoHealth 1:246–254. https://doi.org/10.1007/s10393-004-0094-6
Xu S, Tian R, Lin Y, Yu Z, Zhang Z, Niu X, Wang X, Yang G (2019) Widespread positive selection on cetacean TLR extracellular domain. Mol Immunol 106:135–142. https://doi.org/10.1016/j.molimm.2018.12.022
Yajing S, Rajput IR, Ying H, Fei Y, Sanganyado E, Ping L, Jingzhen W, Wenhua L (2018) Establishment and characterization of pygmy killer whale (Feresa attenuata) dermal fibroblast cell line. PLoS One 13:1–15. https://doi.org/10.1371/journal.pone.0195128
Yu J, Kindy MS, Ellis BC, Baatz JE, Peden-Adams M, Ellingham TJ, Wolff DJ, Fair PA, Gattoni-Celli S (2005) Establishment of epidermal cell lines derived from the skin of the Atlantic bottlenose dolphin (Tursiops truncatus). Anat Rec Part A Discov Mol Cell Evol Biol 287A:1246–1255. https://doi.org/10.1002/ar.a.20266
Zhou P, Tachedjian M, Wynne JW, Boyd V, Cui J, Smith I, Cowled C, Ng JHJ, Mok L, Michalski WP, Mendenhall IH, Tachedjian G, Wang LF, Baker ML (2016) Contraction of the type i IFN locus and unusual constitutive expression of IFN-α in bats. Proc Natl Acad Sci U S A 113:2696–2701. https://doi.org/10.1073/PNAS.1518240113/-/DCSUPPLEMENTAL/PNAS.201518240SI.PDF
Acknowledgements
We would like to thank Dr. Makoto Sugiyama (Gifu University, Gifu, Japan) for providing the dolphin morbillivirus Muc strain. We are also grateful to the cooperation of the Taiji Fisheries Cooperative Union (Wakayama, Japan) and the Fisheries Agency of Japan for providing the specimens used in this study. We would also like to thank the staff of the Shinagawa Aquarium for their assistance in collecting the blood samples used in this study.
Funding
This research was funded by a Grant-in-Aid for Scientific Research C (18K05831) from the Japan Society for the Promotion of Science (KAKENHI).
Author information
Authors and Affiliations
Contributions
Kaede Tashiro, Takao Segawa, and Takuya Itou conceptualized the study. Kaede Tashiro and Takuya Itou conducted the experiments. Kaede Tashiro, Taketo Futami, Miwa Suzuki, and Takuya Itou processed samples. Kaede Tashiro, Takao Segawa, Miwa Suzuki, and Takuya Itou performed the data analysis; Kaede Tashiro drafted, revised, and edited the manuscript. All authors read the manuscript and approved the final submitted manuscript for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tashiro, K., Segawa, T., Futami, T. et al. Establishment and characterization of a novel kidney cell line derived from the common bottlenose dolphin. In Vitro Cell.Dev.Biol.-Animal 59, 536–549 (2023). https://doi.org/10.1007/s11626-023-00786-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-023-00786-y