Abstract
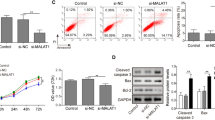
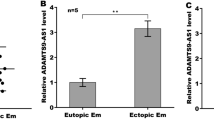
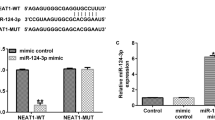
The present study identified a novel upstream long chain non-coding (lncRNA) NEAT1/miR-141-3p/HTRA1 axis that regulated the activation of NLR family pyrin domain containing 3 (NLRP3) inflammasome to modulate endometriosis (EM) development. Specifically, clinical data suggested that the expression of NLRP3 and apoptosis-associated speck-like protein containing CARD (ASC), the cleavage of caspase-1 and gasdermin D (GSDMD), and the production of inflammatory cytokines (interleukin (IL)-1β, IL-6, tumor necrosis factor (TNF)-α, and IL-18) were all significantly increased in the ectopic endometrium (EE) tissues, compared to the normal endometrium (NE) tissues. Then, through analyzing the datasets from GEO database (GSE2339, GSE58178, and GSE7305) using the GEO2R bioinformatics tools, we verified that HtrA Serine Peptidase 1 (HTRA1) was especially enriched in the EE tissues compared to the NE tissues. To further confirm the biological functions of HTRA1, HTRA1 was overexpressed or downregulated in primary human endometrial stromal cells (hESCs) isolated from NE tissues or EE tissues, respectively. The results showed that upregulation of HTRA1 activated NLRP3 inflammasome-mediated pyroptotic cell death and cellular inflammation in NE-derived hESCs, whereas silencing of HTRA1 played an opposite role in EE-derived hESCs. In addition, the lncRNA NEAT1/miR-141-3p axis was screened as the upstream regulator of HTRA1. Mechanistically, lncRNA NEAT1 sponged miR-141-3p to positively regulate HTRA1 in a competing endogenous RNA (ceRNA) mechanisms-dependent manner. The recovery experiments in hESCs from NE and EE tissues confirmed that lncRNA NEAT1 overexpression promoted NLRP3 inflammasome-mediated pyroptotic cell death through regulating the miR-141-3p/HTRA1 axis. Taken together, this study firstly uncovered the underlying mechanisms by which a novel lncRNA NEAT1/miR-141-3p/HTRA1-NLRP3 pathway contributed to the development of EM, which provided novel diagnostic and therapeutic biomarkers for this disease.







Similar content being viewed by others
Data availability
The data were all included in this manuscript. The original data are available from the corresponding author upon reasonable request.
References
Ahamed W, Yu RMC, Pan Y, Iwata T, Barathi VA, Wey YS, Tun SBB, Qiu B, Tan A, Wang X et al (2022) HTRA1 Regulates Subclinical Inflammation and Activates Proangiogenic Response in the Retina and Choroid. Int J Mol Sci 23(18):10206
Bai B, Yang Y, Wang Q, Li M, Tian C, Liu Y, Aung LHH, Li PF, Yu T, Chu XM (2020) NLRP3 inflammasome in endothelial dysfunction. Cell Death Dis 11(9):776
Bullon P, Navarro JM (2017) Inflammasome as a key pathogenic mechanism in endometriosis. Curr Drug Targets 18(9):997–1002
Chen M, Lai X, Wang X, Ying J, Zhang L, Zhou B, Liu X, Zhang J, Wei G, Hua F (2021) Long Non-coding RNAs and Circular RNAs: Insights Into Microglia and Astrocyte Mediated Neurological Diseases. Front Mol Neurosci 14:745066
Choi J, Jo M, Lee E, Kim SE, Lee DY, Choi D (2022) Inhibition of the NLRP3 inflammasome by progesterone is attenuated by abnormal autophagy induction in endometriotic cyst stromal cells: implications for endometriosis. Mol Hum Reprod 28(4):gaac007
Cui L, Chen S, Wang D, Yang Q (2021) LINC01116 promotes proliferation and migration of endometrial stromal cells by targeting FOXP1 via sponging miR-9-5p in endometriosis. J Cell Mol Med 25(4):2000–2012
Dentillo DB, Meola J, Rosa e Silva JC, Giuliatti S, Silva Junior WA Jr, Ferriani RA, Martelli L (2010) Deregulation of LOXL1 and HTRA1 gene expression in endometriosis. Reprod Sci 17(11):1016–1023
Falcone T, Flyckt R (2018) Clinical management of endometriosis. Obstet Gynecol 131(3):557–571
Gan L, Sun J, Sun J (2022a) Bioinformatical analysis identifies PDLIM3 as a potential biomarker associated with immune infiltration in patients with endometriosis. PeerJ 10:e13218
Gan XG, Wang ZH, Xu HT (2022b) Mechanism of miRNA-141-3p in calcium oxalate-induced renal tubular epithelial cell injury via NLRP3-mediated pyroptosis. Kidney Blood Press Res 47(5):300–308
García-Gómez E, Vázquez-Martínez ER, Reyes-Mayoral C, Cruz-Orozco OP, Camacho-Arroyo I, Cerbón M (2019) Regulation of inflammation pathways and inflammasome by sex steroid hormones in endometriosis. Front Endocrinol (Lausanne) 10:935
Goteri G, Altobelli E, Tossetta G, Zizzi A, Avellini C, Licini C, Lorenzi T, Castellucci M, Ciavattini A, Marzioni D (2015) High temperature requirement A1, transforming growth factor beta1, phosphoSmad2 and Ki67 in eutopic and ectopic endometrium of women with endometriosis. Eur J Histochem 59(4):2570
Gu C, Meng Y, Meng Q, Fan W, Ye M, Zhang Q, Zhang N, Li L (2021) Exploring the potential key IncRNAs with endometriosis by construction of a ceRNA network. Int J Gen Med 14:4161–4170
Guo X, Xu X, Li T, Yu Q, Wang J, Chen Y, Ding S, Zhu L, Zou G, Zhang X (2021) NLRP3 inflammasome activation of mast cells by estrogen via the nuclear-initiated signaling pathway contributes to the development of endometriosis. Front Immunol 12:749979
Han SJ, Jung SY, Wu SP, Hawkins SM, Park MJ, Kyo S, Qin J, Lydon JP, Tsai SY, Tsai MJ et al (2015) Estrogen receptor β modulates apoptosis complexes and the inflammasome to drive the pathogenesis of endometriosis. Cell 163(4):960–974
Hooftman A, Angiari S, Hester S, Corcoran SE, Runtsch MC, Ling C, Ruzek MC, Slivka PF, McGettrick AF, Banahan K et al (2020) The immunomodulatory metabolite itaconate modifies NLRP3 and inhibits inflammasome activation. Cell Metab 32(3):468-478.e467
Huang C, Zhang C, Yang P, Chao R, Yue Z, Li C, Guo J, Li M (2020) Eldecalcitol inhibits LPS-Induced NLRP3 inflammasome-dependent pyroptosis in human gingival fibroblasts by activating the Nrf2/HO-1 signaling pathway. Drug Des Devel Ther 14:4901–4913
Huang J, Chen X, Lv Y (2021) HMGB1 mediated inflammation and autophagy contribute to endometriosis. Front Endocrinol (Lausanne) 12:616–696
Jiang L, Zhang M, Wang S, Xiao Y, Wu J, Zhou Y, Fang X (2020) LINC01018 and SMIM25 sponged miR-182–5p in endometriosis revealed by the ceRNA network construction. Int J Immunopathol Pharmacol 34:2058738420976309
Kaufmann FN, Costa AP, Ghisleni G, Diaz AP, Rodrigues ALS, Peluffo H, Kaster MP (2017) NLRP3 inflammasome-driven pathways in depression: clinical and preclinical findings. Brain Behav Immun 64:367–383
Kelley N, Jeltema D, Duan Y, He Y (2019) The NLRP3 inflammasome: an overview of mechanisms of activation and regulation. Int J Mol Sci 20(13):3328
Koninckx PR, Fernandes R, Ussia A, Schindler L, Wattiez A, Al-Suwaidi S, Amro B, Al-Maamari B, Hakim Z, Tahlak M (2021) Pathogenesis based diagnosis and treatment of endometriosis. Front Endocrinol (Lausanne) 12:745548
Lin S, Wen Z, Li S, Chen Z, Li C, Ouyang Z, Lin C, Kuang M, Xue C, Ding Y (2022) LncRNA Neat1 promotes the macrophage inflammatory response and acts as a therapeutic target in titanium particle-induced osteolysis. Acta Biomater 142:345–360
Liu Z, Chen B, Chang J, Feng L, Zhao X (2022) Melatonin regulates trophoblast pyroptosis, invasion and migration in preeclampsia by inhibiting HtrA1 transcription through the microRNA-520c-3p/SETD7 axis. Am J Reprod Immunol 87(4):e13523
Lu Z, Lin V, May A, Che B, Xiao X, Shaw DH, Su F, Wang Z, Du H, Shaw PX (2019) HTRA1 synergizes with oxidized phospholipids in promoting inflammation and macrophage infiltration essential for ocular VEGF expression. PLoS ONE 14(5):e0216808
Malvezzi H, Dobo C, Filippi RZ, Mendes do Nascimento H, Palmieri da Silva ESL, Meola J, Piccinato CA, Podgaec S (2022) Altered p16(Ink4a), IL-1β, and lamin b1 protein expression suggest cellular senescence in deep endometriotic lesions. Int J Mol Sci 23(5):2476
Moretti J, Blander JM (2021) Increasing complexity of NLRP3 inflammasome regulation. J Leukoc Biol 109(3):561–571
Su J, Gao C, Wang R, Xiao C, Yang M (2018) Genes associated with inflammation and the cell cycle may serve as biomarkers for the diagnosis and prognosis of acute myocardial infarction in a Chinese population. Mol Med Rep 18(2):1311–1322
Tamang S, Acharya V, Roy D, Sharma R, Aryaa A, Sharma U, Khandelwal A, Prakash H, Vasquez KM, Jain A (2019) SNHG12: an LncRNA as a potential therapeutic target and biomarker for human cancer. Front Oncol 9:901
Taylor HS, Kotlyar AM, Flores VA (2021) Endometriosis is a chronic systemic disease: clinical challenges and novel innovations. Lancet 397(10276):839–852
Xie C, Yin Z, Liu Y (2022) Analysis of characteristic genes and ceRNA regulation mechanism of endometriosis based on full transcriptional sequencing. Front Genet 13:902329
Xu S, Wang J, Jiang J, Song J, Zhu W, Zhang F, Shao M, Xu H, Ma X, Lyu F (2020) TLR4 promotes microglial pyroptosis via lncRNA-F630028O10Rik by activating PI3K/AKT pathway after spinal cord injury. Cell Death Dis 11(8):693
Yang J, Cheng M, Gu B, Wang J, Yan S, Xu D (2020) CircRNA_09505 aggravates inflammation and joint damage in collagen-induced arthritis mice via miR-6089/AKT1/NF-κB axis. Cell Death Dis 11(10):833
Yu SY, Li XL (2021) Pyroptosis and inflammasomes in obstetrical and gynecological diseases. Gynecol Endocrinol 37(5):385–391
Zhan JF, Huang HW, Huang C, Hu LL, Xu WW (2020) Long non-coding RNA NEAT1 regulates pyroptosis in diabetic nephropathy via mediating the miR-34c/NLRP3 axis. Kidney Blood Press Res 45(4):589–602
Zhang M, Zheng Y, Sun Y, Li S, Chen L, Jin X, Hou X, Liu X, Chen Q, Li J et al (2019a) Knockdown of NEAT1 induces tolerogenic phenotype in dendritic cells by inhibiting activation of NLRP3 inflammasome. Theranostics 9(12):3425–3442
Zhang P, Cao L, Zhou R, Yang X, Wu M (2019b) The lncRNA Neat1 promotes activation of inflammasomes in macrophages. Nat Commun 10(1):1495
Zhao W, Ma L, Cai C, Gong X (2019) Caffeine inhibits NLRP3 inflammasome activation by suppressing MAPK/NF-κB and A2aR signaling in LPS-induced THP-1 macrophages. Int J Biol Sci 15(8):1571–1581
Zheng M, Kanneganti TD (2020) The regulation of the ZBP1-NLRP3 inflammasome and its implications in pyroptosis, apoptosis, and necroptosis (PANoptosis). Immunol Rev 297(1):26–38
Zhou F, Li C, Zhang SY (2020a) NLRP3 inflammasome: a new therapeutic target for high-risk reproductive disorders? Chin Med J (engl) 134(1):20–27
Zhou Y, Tong Z, Jiang S, Zheng W, Zhao J, Zhou X (2020b) The roles of endoplasmic reticulum in NLRP3 inflammasome activation. Cells 9(5):1219
Acknowledgements
Not applicable.
Funding
The research was funded by Yunnan Provincial Clinical Research Center for Obstetrics and Gynecology (Project No.2022YJZX-FC07), the Doctor Research Fund of The First People’s Hospital of Yunnan Province (KHBS-2020-003) and the Yunnan Provincial Department of Science and Technology-Kunming Medical University Joint Special Project on Applied Basic Research (202201AY070001-229).
Author information
Authors and Affiliations
Contributions
All the authors contributed equal work in this research.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

11626_2023_760_MOESM1_ESM.jpg
Supplementary file1 Venn diagram of overlapping down-regulated genes from 3 GSE dataset of EM (GSE23339, GSE58178 and GSE7305). (JPG 769 KB)

11626_2023_760_MOESM2_ESM.jpg
Supplementary file2 (A) Cell morphology of NE-derived hESCs transfected with pcDNA-NEAT1, miR-141-3p mimic or siRNA- HTRA1 was observed under a microscope (scale bar = 100µm). (B) Cell morphology of EE-derived hESCs transfected with siRNA-NEAT1, miR-141-3p inhibitor or pcDNA- HTRA1 (scale bar = 100µm). (C) Cell viability of NE-derived hESCs transfected with pcDNA-NEAT1, miR-141-3p mimic or siRNA- HTRA1 was determined by cell counting Kit-8 (CCK-8) assay. (C) Cell viability of EE-derived hESCs transfected with siRNA-NEAT1, miR-141-3p inhibitor or pcDNA- HTRA1. (nsP>0.05 , *P<0.05). (JPG 2962 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, L., Ye, K. & Wang, D. Upregulation of HTRA1 mediated by the lncRNA NEAT1/miR-141-3p axis contributes to endometriosis development through activating NLRP3 inflammasome-mediated pyroptotic cell death and cellular inflammation. In Vitro Cell.Dev.Biol.-Animal 59, 166–178 (2023). https://doi.org/10.1007/s11626-023-00760-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-023-00760-8