Abstract
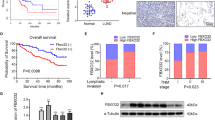
Palmitoylation is a post-translational modification occurring on cysteine residues, which process is catalyzed by a family of zinc finger Asp-His-His-Cys (DHHC) domain-containing (ZDHHC) protein acyltransferases. As a family member, ZDHHC9 plays a crucial role in varied malignancies by regulating protein stability via protein substrate palmitoylation. Based on the bioinformatic analysis of GEO gene microarray GSE75037 (|log2 fold change|> 1, P < 0.05), ZDHHC9 was defined as a significantly upregulated gene in lung adenocarcinoma (LUAD), which was also confirmed in our collected clinical specimens. It is necessary to explore the biological function of ZDHHC9 in LUAD cells. The follow-up functional experiments revealed that ZDHHC9 deficiency inhibited proliferation, migration, and invasion, while stimulated apoptosis in HCC827 cells. Besides, these malignant phenotypes could be accelerated by ZDHHC9 overexpression in A549. Moreover, we revealed that ZDHHC9 knockdown could promote PD-L1 protein degradation by reducing its palmitoylation level. The reduction of PD-L1 protein level could enhance anti-tumor immunity and inhibit the growth of LUAD cells. Therefore, our study uncovers the tumor-promoting role of ZDHHC9 in LUAD via regulating PD-L1 stability through palmitoylation, highlighting ZDHHC9 as a novel therapeutic target for LUAD.
Graphical abstract






Similar content being viewed by others
References
Anderson AM, Ragan MA (2016) Palmitoylation: a protein S-acylation with implications for breast cancer. NPJ Breast Cancer 2:16028
Chen X, Ma H, Wang Z, Zhang S, Yang H, Fang Z (2017) EZH2 Palmitoylation mediated by ZDHHC5 in p53-mutant glioma drives malignant development and progression. Cancer Res 77:4998–5010
Chen Z, Fillmore CM, Hammerman PS, Kim CF, Wong KK (2014) Non-small-cell lung cancers: a heterogeneous set of diseases. Nat Rev Cancer 14:535–546
Du W et al (2021) KPNB1-mediated nuclear translocation of PD-L1 promotes non-small cell lung cancer cell proliferation via the Gas6/MerTK signaling pathway. Cell Death Differ 28:1284–1300
Greaves J, Chamberlain LH (2014) New links between S-acylation and cancer. J Pathol 233:4–6
Jin J, Zhi X, Wang X, Meng D (2021) Protein palmitoylation and its pathophysiological relevance. J Cell Physiol 236:3220–3233
Ko PJ, Dixon SJ (2018) Protein palmitoylation and cancer. EMBO Rep 19
Kong T et al (2020) CD44 promotes PD-L1 expression and its tumor-intrinsic function in breast and lung cancers. Cancer Res 80:444–457
Lei Q, Wang D, Sun K, Wang L, Zhang Y (2020) Resistance mechanisms of anti-PD1/PDL1 therapy in solid tumors. Front Cell Dev Biol 8:672
Li CW et al (2016) Glycosylation and stabilization of programmed death ligand-1 suppresses T-cell activity. Nat Commun 7:12632
Liang J et al (2020) Mex3a interacts with LAMA2 to promote lung adenocarcinoma metastasis via PI3K/AKT pathway. Cell Death Dis 11:614
Linder ME, Deschenes RJ (2007) Palmitoylation: policing protein stability and traffic. Nat Rev Mol Cell Biol 8:74–84
Lu F, Shen SH, Wu S, Zheng P, Lin K, Liao J, Jiang X, Zeng G, Wei D (2022) Hypomethylation-induced prognostic marker zinc finger DHHC-type palmitoyltransferase 12 contributes to glioblastoma progression. Ann Transl Med 10:334
Mansilla F, Birkenkamp-Demtroder K, Kruhøffer M, Sørensen FB, Andersen CL, Laiho P, Aaltonen LA, Verspaget HW, Orntoft TF (2007) Differential expression of DHHC9 in microsatellite stable and instable human colorectal cancer subgroups. Br J Cancer 96:1896–1903
Meng S, Lu C, Zhang W, Shen W, Wei Y, Su D, Zhou F (2016) MMSA-1 expression pattern in multiple myeloma and its clinical significance. Clin Exp Med 16:599–609
Mitchell DA, Vasudevan A, Linder ME, Deschenes RJ (2006) Protein palmitoylation by a family of DHHC protein S-acyltransferases. J Lipid Res 47:1118–1127
Qu M, Zhou X, Wang X, Li H (2021) Lipid-induced S-palmitoylation as a vital regulator of cell signaling and disease development. Int J Biol Sci 17:4223–4237
Salaun C, Greaves J, Chamberlain LH (2010) The intracellular dynamic of protein palmitoylation. J Cell Biol 191:1229–1238
Sharma C, Wang HX, Li Q, Knoblich K, Reisenbichler ES, Richardson AL, Hemler ME (2017) Protein acyltransferase DHHC3 regulates breast tumor growth, oxidative stress, and senescence. Cancer Res 77:6880–6890
Sun C, Mezzadra R, Schumacher TN (2018) Regulation and function of the PD-L1 checkpoint. Immunity 48:434–452
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249
Tang F, Yang C, Li FP, Yu DH, Pan ZY, Wang ZF, Li ZQ (2022) Palmitoyl transferases act as potential regulators of tumor-infiltrating immune cells and glioma progression. Mol Ther Nucleic Acids 28:716–731
Tang J et al (2021) Cancer cells escape p53’s tumor suppression through ablation of ZDHHC1-mediated p53 palmitoylation. Oncogene 40:5416–5426
Touz MC, Conrad JT, Nash TE (2005) A novel palmitoyl acyl transferase controls surface protein palmitoylation and cytotoxicity in Giardia lamblia. Mol Microbiol 58:999–1011
Xu F, Chen JX, Yang XB, Hong XB, Li ZX, Lin L, Chen YS (2020) Analysis of lung adenocarcinoma subtypes based on immune signatures identifies clinical implications for cancer therapy. Mol Ther Oncolytics 17:241–249
Yang Y et al (2019) Palmitoylation stabilizes PD-L1 to promote breast tumor growth. Cell Res 29:83–86
Yao H et al (2019) Inhibiting PD-L1 palmitoylation enhances T-cell immune responses against tumours. Nat Biomed Eng 3:306–317
Yao Y, Zhou Y, Hua Q (2021) circRNA hsa_circ_0018414 inhibits the progression of LUAD by sponging miR-6807-3p and upregulating DKK1. Mol Ther Nucleic Acids 23:783–796
Young E, Zheng ZY, Wilkins AD, Jeong HT, Li M, Lichtarge O, Chang EC (2014) Regulation of Ras localization and cell transformation by evolutionarily conserved palmitoyltransferases. Mol Cell Biol 34:374–385
Yuan M et al (2020) ZDHHC12-mediated claudin-3 S-palmitoylation determines ovarian cancer progression. Acta Pharm Sin B 10:1426–1439
Zhang D, Jiang Q, Ge X, Shi Y, Ye T, Mi Y, Xie T, Li Q, Ye Q (2021a) RHOV promotes lung adenocarcinoma cell growth and metastasis through JNK/c-Jun pathway. Int J Biol Sci 17:2622–2632
Zhang Y, Li F, Fu K, Liu X, Lien IC, Li H (2021b) Potential role of S-palmitoylation in cancer stem cells of lung adenocarcinoma. Front Cell Dev Biol 9:734897
Zhu C, Zhang S, Liu D, Wang Q, Yang N, Zheng Z, Wu Q, Zhou Y (2021) A novel gene prognostic signature based on differential DNA methylation in breast cancer. Front Genet 12:742578
Zhu P, Kang G, Jiao Y, Gui C, Fan H, Li X, Jia Y, Zhang L, Ma X (2022) The α5-nAChR/PD-L1 axis facilitates lung adenocarcinoma cell migration and invasion. Hum Cell 35:1207–1218
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
11626_2023_755_MOESM1_ESM.docx
Supplementary file1 Figure S1. Efficiency detection of ZDHHC9 overexpressing or silencing. The expression of ZDHHC9 in A549 cells (A) and HCC827 cells (B) was detected by real-time PCR. (C) The protein levels of ZDHHC9 were tested by western blot. Data were assayed in triplicate and analyzed by one-way ANOVA followed by Tukey’s multiple comparison tests. ***P < 0.001. (DOCX 226 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Z., Jiang, D., Liu, F. et al. Involvement of ZDHHC9 in lung adenocarcinoma: regulation of PD-L1 stability via palmitoylation. In Vitro Cell.Dev.Biol.-Animal 59, 193–203 (2023). https://doi.org/10.1007/s11626-023-00755-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-023-00755-5