Abstract
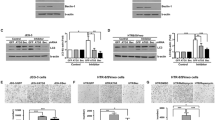
The maintenance of migration of trophoblast cells is beneficial to pregnancy, and its weakening can lead to preeclampsia (PE). CD142 is considered as a classical motility-promoting factor. Our research aimed to explore the role of CD142 in trophoblast cell migration and potential mechanism. Through fluorescence-activated cell sorting (FACS) and gene transduction assays, CD142 expression levels of mouse trophoblast cell lines were upregulated and downregulated respectively. Then, the migratory level was detected through Transwell assays in different groups of trophoblast cells. The corresponding chemokines were screened by ELISA in different sorted trophoblast cells. Based on gene overexpression and knockdown assays, the production mode of identified valuable chemokine was analyzed by detecting gene and protein expression in trophoblast cells. Finally, the contribution of autophagy response to specific chemokine regulated by CD142 was explored by combining different groups of cells and autophagy regulators. Our results showed that both CD142 positive sorting and CD142 overexpression promoted the migratory ability of trophoblast cells, and trophoblast cells with the highest level of CD142 had the strongest migratory ability. In addition, CD142+ cells contained the highest level of IL-8. Consistently, CD142 overexpression promoted IL-8 protein expression in trophoblast cells while CD142 silencing was contrary. However, both CD142 overexpression and CD142 silencing did not affect IL-8 mRNA expression. Moreover, both CD142+ and CD142-overexpressed cells showed higher BCL2 protein expression and poorer autophagic activity. Importantly, autophagy activation with TAT-Beclin1 recovered the increased IL-8 protein expression in CD142+ cells. Obviously, the migratory ability of CD142+ cells inhibited by TAT-Beclin1 was recovered by the addition of IL-8 recombinant factor. In conclusion, CD142 inhibits the degradation of IL-8 through the inhibition of BCL2-Beclin1-autophagy signal transduction, thereby promoting the migration of trophoblast cells.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Change history
29 November 2023
A Correction to this paper has been published: https://doi.org/10.1007/s11626-023-00829-4
References
Arderiu G, Peña E, Aledo R, Juan-Babot O, Badimon L (2011) Tissue factor regulates microvessel formation and stabilization by induction of chemokine (C-C motif) ligand 2 expression. Arterioscler Thromb Vasc Biol 31(11):2607–2615
Carneiro-Lobo TC, Lima MT, Mariano-Oliveira A, Dutra-Oliveira A, Oba-Shinjo SM, Marie SK, Sogayar MC, Monteiro RQ (2014) Expression of tissue factor signaling pathway elements correlates with the production of vascular endothelial growth factor and interleukin-8 in human astrocytoma patients. Oncol Rep 31(2):679–686
Chanakira A, Westmark PR, Ong IM, Sheehan JP (2017) Tissue factor-factor VIIa complex triggers protease activated receptor 2-dependent growth factor release and migration in ovarian cancer. Gynecol Oncol 145(1):167–175
Duhig K, Vandermolen B, Shennan A (2018) Recent advances in the diagnosis and management of pre-eclampsia. F1000Res 7:242
Duley L (2009) The global impact of pre-eclampsia and eclampsia. Semin Perinatol 33(3):130–137
Fang J, Gu L, Zhu N, Tang H, Alvarado CS, Zhou M (2008) Tissue factor/FVIIa activates Bcl-2 and prevents doxorubicin-induced apoptosis in neuroblastoma cells. BMC Cancer 88:69
Fisher SJ (2015) Why is placentation abnormal in preeclampsia? Am J Obstet Gynecol 213(4 Suppl):S115-122
Gao Q, Chen Z, He Y, Hou Z, Ye R, Xue W, Lin J, Tu X (2020) CD142 plays an important role in the mobility of colorectal cancer cells. Biosci Biotechnol Biochem 84(9):1856–1860
Hjortoe GM, Petersen LC, Albrektsen T, Sorensen BB, Norby PL, Mandal SK, Pendurthi UR, Rao LV (2004) Tissue factor-factor VIIa-specific up-regulation of IL-8 expression in MDA-MB-231 cells is mediated by PAR-2 and results in increased cell migration. Blood 103(8):3029–3037
Ikeda U, Hojo Y, Shimada K (2001) Tissue factor overexpression in rat arterial neointima models: thrombosis and progression of advanced atherosclerosis. Circulation 103(10):E59
Illsley NP, DaSilva-Arnold SC, Zamudio S, Alvarez M, Al-Khan A (2020) Trophoblast invasion: lessons from abnormally invasive placenta (placenta accreta). Placenta 102:61–66
Liu J, Liu B, Diao G, Zhang Z (2022) Tissue factor promotes HCC carcinogenesis by inhibiting BCL2-dependent autophagy. Bull Cancer 109(7–8):795–804
Nitori N, Ino Y, Nakanishi Y, Yamada T, Honda K, Yanagihara K, Kosuge T, Kanai Y, Kitajima M, Hirohashi S (2005) Prognostic significance of tissue factor in pancreatic ductal adenocarcinoma. Clin Cancer Res 11(7):2531–2539
Oku K, Amengual O, Zigon P, Horita T, Yasuda S, Atsumi T (2013) Essential role of the p38 mitogen-activated protein kinase pathway in tissue factor gene expression mediated by the phosphatidylserine-dependent antiprothrombin antibody. Rheumatology (Oxford) 52(10):1775–1784
Ott I, Fischer EG, Miyagi Y, Mueller BM, Ruf W (1998) A role for tissue factor in cell adhesion and migration mediated by interaction with actin-binding protein 280. J Cell Biol 140(5):1241–1253
Pijnenborg R, Vercruysse L, Hanssens M (2006) The uterine spiral arteries in human pregnancy: facts and controversies. Placenta 27(9–10):939–958
Queiroz KC, Van ′t Veer C, Van Den Berg Y, Duitman J, Versteeg HH, Aberson HL, Groot AP, Verstege MI, Roelofs JJ, Te Velde AA, Spek CA (2011) Tissue factor-dependent chemokine production aggravates experimental colitis. Mol Med 17(9–10):1119–1126
Ridder A, Giorgione V, Khalil A, Thilaganathan B (2019) Preeclampsia: the relationship between uterine artery blood flow and trophoblast function. Int J Mol Sci 20(13):3263
Shigemori C, Wada H, Matsumoto K, Shiku H, Nakamura S, Suzuki H (1998) Tissue factor expression and metastatic potential of colorectal cancer. Thromb Haemost 80(6):894–898
Tieken C, Verboom MC, Ruf W, Gelderblom H, Bovée JV, Reitsma PH, Cleton-Jansen AM, Versteeg HH (2016) Tissue factor associates with survival and regulates tumour progression in osteosarcoma. Thromb Haemost 115(5):1025–1033
Wu J, Li A, Li Y, Li X, Zhang Q, Song W, Wang Y, Ogutu JO, Wang J, Li J, Tang R, Zhang F (2016) Chlorpromazine inhibits mitochondrial apoptotic pathway via increasing expression of tissue factor. Int J Biochem Cell Biol 70:82–91
Xu W, Chen B, Ke D, Chen X (2022) CD142 plays a key role in the carcinogenesis of gastric adenocarcinoma by inhibiting BCL2-dependent autophagy. Biochem Cell Biol 100(1):17–27
Funding
This work was supported by The Provincial Natural Science Foundation of China Hainan (819MS119, 821QN395, 822MS167) and The 530 Project of the National Natural Science Foundation of China (2021MSXM02, 2021MSXM12).
Author information
Authors and Affiliations
Contributions
Lei Shi and Linmei Zheng conceived and designed experiments. Linmei Zheng, Rong Tang, and Zhongyi Zhou performed experiments, analyzed data, and prepared figures. Jie Song and Zhicheng Lu assisted in data analysis. Lei Shi and Linmei Zheng wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Linmei Zheng and Rong Tang are considered as co-first authors.
The original version of this article was revised: The images presented in Fig. 1B in the article as originally published were incorrect and have been replaced by the correct ones.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, L., Tang, R., Shi, L. et al. CD142 promotes trophoblast cell migration by inhibiting BCL2-related autophagic degradation of IL-8. In Vitro Cell.Dev.Biol.-Animal 59, 131–141 (2023). https://doi.org/10.1007/s11626-023-00751-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-023-00751-9