Abstract
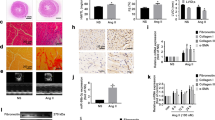
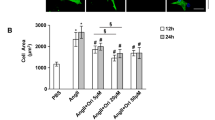
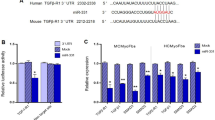
MicroRNAs (miRNAs) have emerged as essential regulators that could have pivotal roles in cardiac homeostasis and pathological remodeling of various cardiovascular diseases. We previously demonstrated that miRNA-122-5p overexpression exacerbated the process of vascular hypertrophy, fibrosis, and dysfunction in hypertensive rats and rat aortic adventitial fibroblasts. However, the exact roles and underlying mechanisms of miRNA-122-5p in myocardial fibroblasts remain largely unknown. In this work, neonatal rat cardiofibroblasts (CFs) were isolated and primarily cultured from the hearts of 2- to 3-d-old Sprague–Dawley rats. Stimulation of angiotensin II (Ang II) resulted in marked increases in cellular proliferation and migration and levels of collagen I, collagen III, CTGF, and TGF-β1 in cultured CFs. Furthermore, Ang II led to promoted expression of P62, Bax, and phosphorylated mTOR as well as downregulation of LC3II, beclin-1, and AMPK-phosphorylated levels, thereby contributing to imbalance of autophagy and apoptosis, and cellular injury in CFs, which were significantly ameliorated by treatment with miRNA-122-5p inhibitor. These changes were associated with decreased levels of collagen I, collagen III, CTGF, and TGF-β1. Furthermore, Ang II–induced loss of autophagy and promotion of apoptosis in CFs were prevented by the treatment with Pyr1-apelin-13 or AMPK agonist AICAR or mTOR inhibitor rapamycin, respectively. In contrast, administration of miRNA-122-5p mimics and autophagy inhibitor 3-methylademine reversed beneficial roles of Pyr1-apelin-13. Collectively, these data indicated that miRNA-122-5p is an essential regulator of autophagy and apoptosis in rat CFs via the apelin/AMPK/mTOR signaling pathway, which may be potentially used as a therapeutic target in myocardial fibrosis and related diseases.






Similar content being viewed by others
References
Ambros V (2004) The functions of animal microRNAs. Nature 431:350–355. https://doi.org/10.1038/nature02871
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297. https://doi.org/10.1016/s0092-8674(04)00045-5
Beaumont J, López B, Hermida N, Schroen B, San José G, Heymans S, Valencia F, Gómez-Doblas JJ, De Teresa E, Díez J, González A (2014) MicroRNA-122 down-regulation may play a role in severe myocardial fibrosis in human aortic stenosis through TGF-β1 up-regulation. Clin Sci (lond) 126:497–506. https://doi.org/10.1042/CS20130538
Chen LJ, Xu R, Yu HM, Chang Q, Zhong JC (2015) The ACE2/apelin signaling, microRNA, and hypertension. Int J Hypertens 2015:896861. https://doi.org/10.1155/2015/896861
Fan Y, Ma X, Li H, Gao Y, Huang Q, Zhang Y, Bao X, Du Q, Luo G, Liu K, Meng Q, Zhao C, Zhang X (2018) MiR-122 promotes metastasis of clear-cell renal cell carcinoma by downregulating Dicer. Int J Cancer 142:547–560. https://doi.org/10.1002/ijc.31050
Gao F, Chen J, Zhu H (2018) A potential strategy for treating atherosclerosis: improving endothelial function via AMP-activated protein kinase. Sci China Life Sci 61:1024–1029. https://doi.org/10.1007/s11427-017-9285-1
Gyöngyösi M, Winkler J, Ramos I, Do QT, Firat H, McDonald K, González A, Thum T, Díez J, Jaisser F, Pizard A, Zannad F (2017) Myocardial fibrosis: biomedical research from bench to bedside. Eur J Heart Fail 19(2):177–191. https://doi.org/10.1002/ejhf.696
He L, Zhou Q, Huang Z, Xu J, Zhou H, Lv D, Lu L, Huang S, Tang M, Zhong J, Chen JX, Luo X, Li L, Chen L (2019) PINK1/parkin-mediated mitophagy promotes apelin-13-induced vascular smooth muscle cell proliferation by AMPKα and exacerbates atherosclerotic lesions. J Cell Physiol 234:8668–8682. https://doi.org/10.1002/jcp.27527
Levine B, Kroemer G (2019) Biological functions of autophagy genes: a disease perspective. Cell 176:11–42. https://doi.org/10.1016/j.cell.2018.09.048
Li S, Le W (2017) An insight review of autophagy biology and neurodegenerative diseases: machinery, mechanisms and regulation. Sci China Life Sci 60:1457–1459. https://doi.org/10.1007/s11427-017-9214-7
Li Y, Yang N, Dong B, Yang J, Kou L, Qin Q (2019) MicroRNA-122 promotes endothelial cell apoptosis by targeting XIAP: Therapeutic implication for atherosclerosis. Life Sci 232:116590. https://doi.org/10.1016/j.lfs.2019.116590
Liang D, Han D, Fan W, Zhang R, Qiao H, Fan M, Su T, Ma S, Li X, Chen J, Wang Y, Ren J, Cao F (2016) Therapeutic efficacy of apelin on transplanted mesenchymal stem cells in hindlimb ischemic mice via regulation of autophagy. Sci Rep 6:21914. https://doi.org/10.1038/srep21914
Long JK, Dai W, Zheng YW, Zhao SP (2019) MiR-122 promotes hepatic lipogenesis via inhibiting the LKB1/AMPK pathway by targeting Sirt1 in non-alcoholic fatty liver disease. Mol Med 25:26. https://doi.org/10.1186/s10020-019-0085-2
Montgomery RL, Hullinger TG, Semus HM, Dickinson BA, Seto AG, Lynch JM, Stack C, Latimer PA, Olson EN, van Rooij E (2011) Therapeutic inhibition of miR-208a improves cardiac function and survival during heart failure. Circulation 124:1537–1547. https://doi.org/10.1161/CIRCULATIONAHA.111.030932
Park S, Nguyen NB, Pezhouman A, Ardehali R (2019) Cardiac fibrosis: potential therapeutic targets. Transl Res 209:121–137. https://doi.org/10.1016/j.trsl.2019.03.001
Patel VB, Zhong JC, Grant MB, Oudit GY (2016) Role of the ACE2/angiotensin 1–7 axis of the renin-angiotensin system in heart failure. Circ Res 118:1313–1326. https://doi.org/10.1161/CIRCRESAHA.116.307708
Ren J, Zhang Y (2018) Targeting autophagy in aging and aging-related cardiovascular diseases. Trends Pharmacol Sci 39:1064–1076. https://doi.org/10.1016/j.tips.2018.10.005
Shirakabe A, Ikeda Y, Sciarretta S, Zablocki DK, Sadoshima J (2016) Aging and autophagy in the Heart. Circ Res 118:1563–1576. https://doi.org/10.1161/CIRCRESAHA
Skommer J, Rana I, Marques FZ, Zhu W, Du Z, Charchar FJ (2014) Small molecules, big effects: the role of microRNAs in regulation of cardiomyocyte death. Cell Death Dis 5:e1325. https://doi.org/10.1038/cddis.2014.287
Sun T, Li MY, Li PF, Cao JM (2018) MicroRNAs in cardiac autophagy: small molecules and big role. Cells 7:7080104. https://doi.org/10.3390/cells7080104
Ucar A, Gupta SK, Fiedler J, Erikci E, Kardasinski M, Batkai S, Dangwal S, Kumarswamy R, Bang C, Holzmann A, Remke J, Caprio M, Jentzsch C, Engelhardt S, Geisendorf S, Glas C, Hofmann TG, Nessling M, Richter K, Schiffer M, Carrier L, Napp LC, Bauersachs J, Chowdhury K, Thum T (2012) The miRNA-212/132 family regulates both cardiac hypertrophy and cardiomyocyte autophagy. Nat Commun 3:1078. https://doi.org/10.1038/ncomms2090
van Nieuwenhoven FA, Turner NA (2013) The role of cardiac fibroblasts in the transition from inflammation to fibrosis following myocardial infarction. Vascul Pharmacol 58(3):182–188. https://doi.org/10.1016/j.vph.2012.07.003
Wang L, Tang W, Yan S, Zhou L, Shen T, Huang X, Dou L, Wang M, Yu S, Li J (2013) Efficient delivery of miR-122 to regulate cholesterol metabolism using a non-covalent peptide-based strategy. Mol Med Rep 8:1472–1478. https://doi.org/10.3892/mmr.2013.1691
Wang Y, Chang W, Zhang Y, Zhang L, Ding H, Qi H, Xue S, Yu H, Hu L, Liu D, Zhu W, Wang Y, Li P (2019) Circulating miR-22-5p and miR-122-5p are promising novel biomarkers for diagnosis of acute myocardial infarction. J Cell Physiol 234(4):4778–4786. https://doi.org/10.1002/jcp.27274
Xiong Y, Yepuri G, Forbiteh M, Yu Y, Montani JP, Yang Z, Ming XF (2014) ARG2 impairs endothelial autophagy through regulation of MTOR and PRKAA/AMPK signaling in advanced atherosclerosis. Autophagy 10:2223–2238. https://doi.org/10.4161/15548627
Xu R, Zhang ZZ, Chen LJ, Yu HM, Guo SJ, Xu YL, Oudit GY, Zhang Y, Chang Q, Song B, Chen DR, Zhu DL, Zhong JC (2016) Ascending aortic adventitial remodeling and fibrosis are ameliorated with apelin-13 in rats after TAC via suppression of the miRNA-122 and LGR4-β-catenin signaling. Peptides 86:85–94. https://doi.org/10.1016/j.peptides.2016.10.005
Yang Y, Lv SY, Lyu SK, Wu D, Chen Q (2015) The protective effect of apelin on ischemia/reperfusion injury. Peptides 63:43–46. https://doi.org/10.1016/j.peptides.2014.11.001
Zhang ZZ, Wang W, Jin HY, Chen X, Cheng YW, Xu YL, Song B, Penninger JM, Oudit GY, Zhong JC (2017) Apelin is a negative regulator of angiotensin II-mediated adverse myocardial remodeling and dysfunction. Hypertension 70:1165–1175. https://doi.org/10.1161/HYPERTENSIONAHA.117.10156
Zhong JC, Yu XY, Huang Y, Yung LM, Lau CW, Lin SG (2007) Apelin modulates aortic vascular tone via endothelial nitric oxide synthase phosphorylation pathway in diabetic mice. Cardiovasc Res 74:388–395. https://doi.org/10.1016/j.cardiores.2007.02.002
Zhong JC, Zhang ZZ, Wang W, McKinnie SMK, Vederas JC, Oudit GY (2017) Targeting the apelin pathway as a novel therapeutic approach for cardiovascular Diseases. Biochim Biophys Acta Mol Basis Dis 1863:1942–1950. https://doi.org/10.1016/j.bbadis.2016.11.007
Funding
This research program was supported by Natural Science Foundation of Beijing (#7222068) and the National Natural Science Foundation of China (#92168117; 81770253; 91849111; 91339108).
Author information
Authors and Affiliations
Contributions
YM and ZJC conceived the study and designed the experiments, and wrote the manuscript. SJJ contributed to data collection. YXC performed data analysis and interpreted the results. ZGZ and ZJC contributed to critical revision of the article. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Yang, M., Song, JJ., Yang, XC. et al. MiRNA-122-5p inhibitor abolishes angiotensin II–mediated loss of autophagy and promotion of apoptosis in rat cardiofibroblasts by modulation of the apelin-AMPK-mTOR signaling. In Vitro Cell.Dev.Biol.-Animal 58, 136–148 (2022). https://doi.org/10.1007/s11626-022-00651-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-022-00651-4