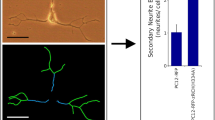
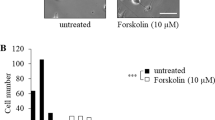
Abstract
The rat PC12 cell line has become a widely used research tool for many aspects of neurobiology. Nerve growth factor (NGF)-responsive PC12 cells were engineered to drive expression of doxycycline (Dox)-induced gene of interest in the Tet-On expression system that resulted in obtaining PC12-Tet-On cells. TrkA and TrkC are neurotrophin receptors derived from the tropomyosin-related kinase (Trk) family of receptor tyrosine kinases. TrkA receptor binds and is activated mainly by NGF, while TrkC receptor binds and is activated by neurotrophin 3 (NT3). The purpose of this research was to design and describe PC12-based neuronal cell model to study TrkC-triggered versus TrkA-triggered neurite outgrowth. The second-generation tetracycline-responsive promoter (P tight) was used in order to provide low basal expression in the absence of Dox and high-level Dox-induced expression of TrkC. The main advantage of presented model system is dependence of TrkC level on Dox concentration. It also allows to compare activation of intracellular signaling proteins and neurite outgrowth following activation of TrkA and TrkC receptors by NGF and NT3, respectively, in the context of the same quality and quantity of intracellular adaptor proteins, Ras proteins, protein kinases and phosphatases, and phospholipase Cγ1, as a difference in the activation of intracellular signaling network by these two distinct although related receptor tyrosine kinases is expected. The results of our studies suggest that despite slightly weaker activation of ERK1/2 mitogen-activated protein kinases, NT3-triggered TrkC seems to provide apparently stronger than NGF-triggered TrkA signal for neurite elongation in differentiating PC12 cells.



Similar content being viewed by others
References
Agha-Mohammadi S, Malley MO, Etemad A, Wang Z, Xiao X, Lotze MT (2004) Second-generation tetracycline-regulatable promoter: repositioned tet operator elements optimize transactivator synergy while shorter minimal promoter offers tight basal leakiness. J Gene Med 6:817–828. doi:10.1002/jgm.566
Belliveau DJ, Krivko I, Kohn J, Lachance C, Pozniak C, Rusakov D, Kaplan D, Miller FD (1997) NGF and neurotrophin-3 both activate TrkA on sympathetic neurons but differentially regulate survival and neuritogenesis. J Cell Biol 136:375–388. doi:10.1083/jcb.136.2.375
Bentley JK, Juilfs DM, Uhler MD (2001) Nerve growth factor inhibits PC12 cell PDE 2 phosphodiesterase activity and increases PDE 2 binding to phosphoproteins. J Neurochem 76:1252–1263. doi:10.1046/j.1471-4159.2001.00133.x
Bibel M, Barde YA (2000) Neurotrophins: key regulators of cell fate and cell shape in the vertebrate nervous system. Genes Dev 14:2919–2937. doi:10.1101/gad.841400
Ferrari A, Faraci P, Cecchini M, Beltram F (2010) The effect of alternative neuronal differentiation pathways on PC12 cell adhesion and neurite alignment to nanogratings. Biomaterials 31:2565–2573. doi:10.1016/j.biomaterials.2009.12.010
Gossen M, Bujard H (1992) Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc Natl Acad Sci U S A 89:5547–5551. doi:10.1073/pnas.89.12.5547
Gossen M, Freundlieb S, Bender G, Muller G, Hillen H, Bujard H (1995) Transcriptional activation by tetracyclines in mammalian cells. Science 268:1766–1769. doi:10.1126/science.7792603
Greene LA, Tischler AS (1976) Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc Natl Acad Sci U S A 73:2424–2428
Jörnvall H, Blokzijl A, ten Dijke P, Ibáñez CF (2001) The orphan receptor serine/threonine kinase ALK7 signals arrest of proliferation and morphological differentiation in a neuronal cell line. J Biol Chem 276:5140–5146. doi:10.1074/jbc.M005200200
Klopotowska D, Strzadala L, Matuszyk J (2008) Inducibility of doxycycline-regulated gene in neural and neuroendocrine cells strongly depends on the appropriate choice of a tetracycline-responsive promoter. Neurochem Int 52:221–229. doi:10.1016/j.neuint.2007.05.014
Lemmon MA, Schlessinger J (2000) Cell signaling by receptor tyrosine kinases. Cell 141:1117–1134. doi:10.1016/j.cell.2010.06.011
Toniatti C, Bujard H, Cortese R, Ciliberto G (2004) Gene therapy progress and prospects: transcription regulatory systems. Gene Ther 11:649–657. doi:10.1038/sj.gt.3302251
Tsoulfas P, Stephens RM, Kaplan DR, Parada LF (1996) TrkC isoforms with inserts in the kinase domain show impaired signaling responses. J Biol Chem 271:5691–5697. doi:10.1074/jbc.271.10.5691
Uren RT, Turbić A, Wong AW, Klein R, Murray SS, Turnley AM (2014) A novel role of suppressor of cytokine signaling-2 (SOCS2) in the regulation of TrkA neurotrophin receptor biology. J Neurochem 129:614–627. doi:10.1111/jnc.12671
Vaudry D, Stork PJ, Lazarovici P, Eiden LE (2002) Signaling pathways for PC12 cell differentiation: making the right connections. Science 296:1648–1649. doi:10.1126/science.1071552
Acknowledgment
This work was supported by the polish Ministry of Science and Higher Education grant no. N N401 063636.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: T. Okamoto
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary figure 1
Dose-dependent Dox-induced expression of trkC gene in clone 4.1. Cells were treated for 24 h with different Dox concentrations. Relative quantification of Dox-induced trkC mRNAs were determined by real-time PCR with SYBR Green and normalized to the hprt mRNAs. Data are representative from 4 independent experiments. (GIF 41 kb)
Supplementary figure 2
Expression of exogenous TrkC protein in clone 4.1. Cells were grown in the absence (A, B) or presence of 1 μg/mL Dox (C). Cells were immunostained for TrkC (red) with goat anti-TrkC primary antibody (B, C) plus anti-goat IgG secondary antibody conjugated to Cy3 (A-C). Cells were stained for nuclei (green) with YO-PRO-1 iodide and analyzed by immunofluorescence microscopy. Data are representative from 2 independent experiments. (GIF 82 kb)
Rights and permissions
About this article
Cite this article
Krawczyk, P., Twarog, E., Kurowska, E. et al. Establishment of a cellular model to study TrkC-dependent neuritogenesis. In Vitro Cell.Dev.Biol.-Animal 51, 241–248 (2015). https://doi.org/10.1007/s11626-014-9829-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-014-9829-z