Abstract
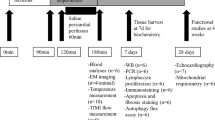
Though mild hypothermia displays an optimistic alleviation of contractive failure in the ischemia/reperfusion myocardium, we still lacked answers to many questions about its potential mechanisms. Our hypothesis is that hypothermia (32°C) induced in ischemia can ease mitochondrial injury resulting in improvement of myocardial contractility even under the condition of a normothermic reperfusion. Fifty newly born 1–2 d Sprague–Dawley rats were executed and the primary cardiomyocytes were obtained and cultivated in vitro. Myocytes were randomized into three groups and then subjected to ischemia either at 32°C or 37°C, both prior to undergoing reperfusion at 37°C. Contractility was presented as frequency and velocity. Ultrastructural alterations of cardiomyocytes and mitochondrion underwent semi-quantitative analysis with transmission electron microscopy and respiratory function of mitochondria was further assessed simultaneously. During cooling ischemia and following reperfusion, cardiomyocytes acquired a more immediate restoration to baseline level and had a significant difference as compared with those in normothermia (P < 0.05). Furthermore, hypothermia preserved the ultrastructure of myocytes and mitochondrion after ischemia. However, measurement on Heart Injury Score and form factor revealed no differences after 2-h reperfusion either in hypothermia or normothermia. On the contrary, the surface area and respiratory function of mitochondrion in reperfusion differed significantly in both groups (P < 0.05) which had an accordance with the variation on contractile performance. Hypothermia only induced in ischemia can bring contractility benefit even under a normothermia reperfusion in cultured cardiomyocytes





Similar content being viewed by others
References
Bernard S. A.; Gray T. W.; Buist M. D.; Jones B. M.; Silvester W.; Gutteridge G.; Smith K. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N. Engl. J. Med. 346: 557–563; 2002.
Fang X.; Huang Z.; Zhu J. et al. Ultrastructural evidence of mitochondrial abnormalities in postresuscitation myocardial dysfunction. Resuscitation 83(3): 386–394; 2012.
Fu J.; Gao J.; Pi R.; Liu P. An optimized protocol for culture of cardiomyocyte from neonatal rat. Cytotechnology 49: 109–116; 2005.
Gao W.-X.; Liu J.-Z. et al. Characteristics of energy metabolism in brain mitochondria of rats exposed to hypoxia. Chin. J. Pathophysiol. 16(10): 879–882; 2000.
Gazmuri R. J.; Weil M. H.; Bisera J.; Tang W.; Fukui M.; McKee D. Myocardial dysfunction after successful resuscitation from cardiac arrest. Critical Care Med. 24(6): 992–1000; 1996.
Gilbert N. F.; Meyer P. E.; Tauriainen M. P. et al. Effects of hypothermia on myocardial substrate selection. Ann. Thorac. Surg. 74: 1208–12; 2002.
Hale S. L.; Kolner R. A. Myocardial temperature reduction attenuates instituted after prolonged ischemia in rabbits. Cardiovasc. Res. 40: 502–507; 1998.
Heyndrickx G. R.; Millard R. W. Regional myocardial functional and electrophysiological alterations after brief coronary artery occlusion in conscious dogs. J. Clin. Invest. 56: 978–85; 1975.
Hypothermia after Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N. Engl. J. Med. 346: 549–556; 2002.
Kern K. B.; Hilwig R. W.; Rhee K. H.; Berg R. A. Myocardial dysfunction after resuscitation from cardiac arrest: an example of global myocardial stunning. J. Am. Coll. Cardiol. 28(1): 232–40; 1996.
Kuniyoshi Y.; Koja K. et al. Myocardial protective effect of hypothermia during extracorporeal circulation—by quantitative measurement of myocardial oxygen consumption. Ann. Thorac. Cardiovasc. Surg. 9: 155–62; 2003.
Laurent I.; Monchi M. et al. Reversible myocardial dysfunction in survivors of out-of-hospital cardiac arrest. J. Am. Coll. Cardiol. 40(12): 2110–6; 2002.
Miki T.; Liu G. S.; Cohen M. V. et al. Mild hypothermia reduces infarct size in the beating rabbit heart: a practical intervention for acute myocardial infarction. Basic Res. Cardiol. 93: 372–383; 1998.
Negovsky V. A. Postresuscitation disease. Crit. Care Med. 16: 942–6; 2009.
Neumar R. W.; Nolan J. P. Post-cardiac arrest syndrome. Circulation 118: 2452–83; 2008.
Ning X.; Chengsu Xu; Song Y. et al. Hypothermia preserves function and signaling for mitochondrial biogenesis during subsequent ischemia. Am. J. Physiol. Heart Circ. Physiol. 274: 786–793; 1998.
Ozisik K.; Yildirim E. et al. Ultrastructural changes of rat cardiac myocytes in a time-dependent manner after traumatic brain injury. Am. J. Transplant. 4: 900–904; 2004.
Petrosillo G.; Ruggiero F. et al. Role of reactive oxygen species and cardiolipin in the release of cytochrome c from mitochondria. FASEB J. 17(15): 2202–2208; 2003.
Ristagno G.; Tantillo S.; Sun S.; HarryWeil M.; Tang W. Hypothermia improves ventricular myocyte contractility under conditions of normal perfusion and after an interval of ischemia. Resuscitation 81: 898–903; 2010.
Shao Z.-H.; Chang W.-T.; Chan K. C. et al. Hypothermia-induced cardioprotection using extended ischemia and early reperfusion cooling. Am. J. Physiol. Heart Circ. Physiol. 292: H1995–2003; 2007.
Sims N. R. Rapid isolation of metabolically active mitochondria from rat brain and subregions using Percoll density gradient centrifugation. J. Neurochem. 55(2): 698–707; 1990.
Song F.; Shan Y.; Cappello F. et al. Apoptosis is not involved in the mechanism of myocardial dysfunction after resuscitation in a rat model of cardiac arrest and cardiopulmonary resuscitation. Critical Care Med. 38: 1329–1334; 2010.
Tissier R.; Couvreu N.; Ghaleh B. et al. Rapid cooling preserves the ischaemic myocardium against mitochondrial damage and left ventricular dysfunction. Cardiovasc. Res. 83: 345–353; 2009.
Tsai M.-S.; Barbut D.; Wang H. et al. Intra-arrest rapid head cooling improves postresuscitation myocardial function in comparison with delayed postresuscitation surface cooling. Critical Care Med. 36: S434–439; 2008.
Webster C. M.; Kelly S.; Koike M. A. et al. Inflammation and NFκB activation is decreased by hypothermia following global cerebral ischemia. Neurobiol. Dis. 33(2): 301–312; 2009.
Xu T.; Tang W. et al. Postresuscitation myocardial diastolic dysfunction following prolonged ventricular fibrillation and cardiopulmonary resuscitation. Critical Care Med. 36: 188–192; 2008.
Zhu C.; Wang X.; Cheng X. et al. Post-ischemic hypothermia-induced tissue protection and diminished apoptosis after neonatal cerebral hypoxia–ischemia. Brain Res. 996: 67–75; 2004.
Acknowledgment
Our best appreciations should be sincerely sent to Haibo Chen (Analysis and Testing Center, Dalian Medical College) for his technical assistance on Image Pro Plus software and constructive advice of morphometric measurement on mitochondrion.
Funding sources
This study was supported by a research grant from National Natural Science Foundation of China (30700303), the Fundamental Research Funds for the Central Universities and Yat-Sen Scholarship for Young Scientists.
Conflict of interest
The authors have no conflict of financial interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: T. Okamoto
H. Li and X. Fang contributed equally to this study.
Rights and permissions
About this article
Cite this article
Li, H., Fang, X., Yang, Z. et al. Ischemia hypothermia improved contractility under normothermia reperfusion in the model of cultured cardiomyocyte. In Vitro Cell.Dev.Biol.-Animal 48, 284–292 (2012). https://doi.org/10.1007/s11626-012-9497-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-012-9497-9