Abstract
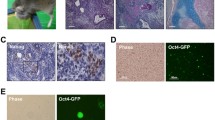
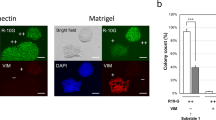
The aims of this study were to analyze the spontaneous differentiation of human embryonic stem cells in vitro and in vivo and to investigate the influence of in vitro partial differentiation on in vivo teratoma formation in immunodeficient mice. Standardized methods are needed for long-term cultivation of undifferentiated stem cells and the multilineage cells that spontaneously differentiate from them. Accordingly, SA002 human embryonic stem cells were cultured on irradiated mouse embryonic fibroblasts cells, on irradiated human foreskin fibroblasts, or were cultured feeder-free using matrigel. Expression of marker protein transcripts was analyzed in undifferentiated and differentiated stem cells using real-time PCR, and both types of stem cells were transplanted subcutaneously into immunodeficient NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ mice to test for teratoma formation. Teratoma histology and expression profiles were subsequently characterized. Cells cultured using different conditions and morphologically undifferentiated cells had comparable marker expression profiles, showing high expression levels of markers for pluripotency and low-to-moderate expression levels of germ layer markers. Cells showing spontaneous differentiation that were cultured in feeder-free conditions in the absence of basic fibroblast growth factor demonstrated slight upregulation of sex determining region Y-box 17, connexin 32, and albumin expression at early time points, as well as expression of octamer-binding transcription factor 4, proteoglycan epitopes on podocalyxin (Trafalgar), and alkaline phosphatase. At later time points, expression of hepatocyte nuclear factor-3-beta, and hepatocyte nuclear factor-4-alpha and alpha fetoprotein was upregulated, whereas beta-3-tubulin, chemokine receptor, nestin, sex-determining region Y-box 17, and connexin 32 were downregulated. Expression of pluripotency markers remained high, and hematopoetic markers were not expressed. SA002 cells that showed spontaneous partial differentiation in vitro had a low teratoma formation capacity in vivo. Cells that were partially differentiated led to slower growing teratomas with more uniform histology.



Similar content being viewed by others
References
Adewumi O.; Aflatoonian B.; hrlund-Richter L.; Amit M.; Andrews P. W.; Beighton G.; Bello P. A.; Benvenisty N.; Berry L. S.; Bevan S.; Blum B.; Brooking J.; Chen K. G. et al. Characterization of human embryonic stem cell lines by the International Stem Cell Initiative. Nat. Biotechnol. 25: 803–816; 2007.
Amit M.; Itskovitz-Eldor J. Derivation and spontaneous differentiation of human embryonic stem cells. J. Anat. 200: 225–232; 2002.
Amit M.; Itskovitz-Eldor J. Feeder-free culture of human embryonic stem cells. Methods Enzymol. 420: 37–49; 2006.
Applied Biosystem StepOne™ and StepOnePlus™ Real-Time PCR System Reagent Guide Part Number 4379704 Rev. B 04; 2007
Braam S. R.; Denning C.; Matsa E.; Young L. E.; Passier R.; Mummery C. L. Feeder-free culture of human embryonic stem cells in conditioned medium for efficient genetic modification. Nat. Protoc. 3(9): 1435–1443; 2008.
Brederlau A.; Correia A. S.; Anisimov S. V.; Elmi M.; Paul G.; Roybon L.; Morizane A.; Bergquist F.; Riebe I.; Nannmark U.; Carta M.; Hanse E.; Takahashi J.; Sasai Y.; Funa K.; Brundin P.; Eriksson P. S.; Li J. Y. Transplantation of human embryonic stem cell-derived cells to a rat model of Parkinson’s disease: effect of in vitro differentiation on graft survival and teratoma formation. Stem Cells. 24(6): 1433–1440; 2006.
Brivanlou A. H.; Gage F. H.; Jaenisch R.; Jessell T.; Melton D.; Rossant J. Stem cells. Setting standards for human embryonic stem cells. Science. 300(5621): 913–916; 2003.
Chen H. F.; Chuang C. Y.; Shieh Y. K.; Chang H. W.; Ho H. N.; Kuo H. C. Novel autogenic feeders derived from human embryonic stem cells (hESCs) support an undifferentiated status of hESCs in xeno-free culture conditions. Hum. Reprod. 24(5): 1114–1125; 2009.
Conner D. A. Mouse embryo fibroblast (MEF) feeder cell preparation. Curr. Protoc. Mol. Biol. 51: 23.2.1–23.2.7; 2001.
Cooke M. J.; Stojkovic M.; Przyborski S. A. Growth of teratomas derived from human pluripotent stem cells is influenced by the graft site. Stem Cells Dev. 15(2): 254–259; 2006.
D’Amour K. A.; Agulnick A. D.; Eliazer S.; Kelly O. G.; Kroon E.; Baetge E. E. Efficient differentiation of human embryonic stem cells to definitive endoderm. Nat. Biotechnol. 23(12): 1534–1541; 2005.
Drukker M.; Katchman H.; Katz G.; Even-Tov F. S.; Shezen E.; Hornstein E.; Mandelboim O.; Reisner Y.; Benvenisty N. Human embryonic stem cells and their differentiated derivatives are less susceptible to immune rejection than adult cells. Stem Cells. 24(2): 221–229; 2006.
Dziadek M.; Adamson E. Localization and synthesis of alphafoetoprotein in post-implantation mouse embryos. J. Embryol. Exp. Morphol. 43: 289–313; 1978.
Eakin G. S.; Hadjantonakis A. K.; Papaioannou V. E.; Behringer R. R. Developmental potential and behavior of tetraploid cells in the mouse embryo. Dev. Biol. 288(1): 150–159; 2005.
Ellerstrom C.; Strehl R.; Noaksson K.; Hyllner J.; Semb H. Facilitated expansion of human embryonic stem cells by single-cell enzymatic dissociation. Stem Cells. 25(7): 1690–1696; 2007.
Fong C. Y.; Gauthaman K.; Bongso A. Teratomas from pluripotent stem cells: a clinical hurdle. J. Cell Biochem. 111: 769–781; 2010.
Ghosh Z.; Huang M.; Hu S.; Wilson K. D.; Dey D.; Wu J. C. Dissecting the oncogenic and tumorigenic potential of differentiated human induced pluripotent stem cells and human embryonic stem cells. Cancer Res. 71(14): 5030–5039; 2011.
Ginzinger D. G. Gene quantification using real-time quantitative PCR: an emerging technology hits the mainstream. Exp. Hematol. 30(6): 503–512; 2002.
Greber B.; Lehrach H.; Adjaye J. Fibroblast growth factor 2 modulates transforming growth factor beta signaling in mouse embryonic fibroblasts and human ESCs (hESCs) to support hESC self-renewal. Stem Cells. 25(2): 455–464; 2007.
Habich A.; Jurga M.; Markiewicz I.; Lukomska B.; Bany-Laszewicz U.; Domanska-Janik K. Early appearance of stem/progenitor cells with neural-like characteristics in human cord blood mononuclear fraction cultured in vitro. Exp. Hematol. 34(7): 914–925; 2006.
Hannoun Z.; Fletcher J.; Greenhough S.; Medine C.; Samuel K.; Sharma R.; Pryde A.; Black J. R.; Ross J. A.; Wilmut I.; Iredale J. P.; Hay D. C. The comparison between conditioned media and serum-free media in human embryonic stem cell culture and differentiation. Cell Reprogram. 12(2): 133–140; 2010.
Heins N.; Englund M. C.; Sjoblom C.; Dahl U.; Tonning A.; Bergh C.; Lindahl A.; Hanson C.; Semb H. Derivation, characterization, and differentiation of human embryonic stem cells. Stem Cells. 22(3): 367–376; 2004.
Heins N.; Lindahl A.; Karlsson U.; Rehnstrom M.; Caisander G.; Emanuelsson K.; Hanson C.; Semb H.; Bjorquist P.; Sartipy P.; Hyllner J. Clonal derivation and characterization of human embryonic stem cell lines. J. Biotechnol. 122(4): 511–520; 2006.
Iida M.; Heike T.; Yoshimoto M.; Baba S.; Doi H.; Nakahata T. Identification of cardiac stem cells with FLK1, CD31, and VE-cadherin expression during embryonic stem cell differentiation. FASEB J. 19(3): 371–378; 2005.
King F. W.; Ritner C.; Liszewski W.; Kwan H. C.; Pedersen A.; Leavitt A. D.; Bernstein H. S. Subpopulations of human embryonic stem cells with distinct tissue-specific fates can be selected from pluripotent cultures. Stem Cells Dev. 18(10): 1441–1450; 2009.
Klimanskaya I.; Chung Y.; Becker S.; Lu S. J.; Lanza R. Human embryonic stem cell lines derived from single blastomeres. Nature. 444(7118): 481–485; 2006.
Klimanskaya I.; Chung Y.; Meisner L.; Johnson J.; West M. D.; Lanza R. Human embryonic stem cells derived without feeder cells. Lancet. 365(9471): 1636–1641; 2005.
Knoepfler P. S. Deconstructing stem cell tumorigenicity: a roadmap to safe regenerative medicine. Stem Cells. 27(5): 1050–1056; 2009.
Lavon N.; Benvenisty N. Study of hepatocyte differentiation using embryonic stem cells. J. Cell Biochem. 96(6): 1193–1202; 2005.
Lee D. S.; Yu K.; Rho J. Y.; Lee E.; Han J. S.; Koo D. B.; Cho Y. S.; Kim J.; Lee K. K.; Han Y. M. Cyclopamine treatment of human embryonic stem cells followed by culture in human astrocyte medium promotes differentiation into nestin- and GFAP-expressing astrocytic lineage. Life Sci. 80(2): 154–159; 2006.
Li H. Y.; Chien Y.; Chen Y. J.; Chen S. F.; Chang Y. L.; Chiang C. H.; Jeng S. Y.; Chang C. M.; Wang M. L.; Chen L. K.; Hung S. I.; Huo T. I.; Lee S. D.; Chiou S. H. Reprogramming induced pluripotent stem cells in the absence of c-Myc for differentiation into hepatocyte-like cells. Biomaterials. 32: 5994–6005; 2011.
Lim J. W.; Bodnar A. Proteome analysis of conditioned medium from mouse embryonic fibroblast feeder layers which support the growth of human embryonic stem cells. Proteomics. 2(9): 1187–1203; 2002.
Lu J.; Hou R.; Booth C. J.; Yang S. H.; Snyder M. Defined culture conditions of human embryonic stem cells. Proc. Natl. Acad. Sci. U.S.A. 103(15): 5688–5693; 2006.
Michalska A.E. Isolation and propagation of mouse embryonic fibroblasts and preparation of mouse embryonic feeder layer cells. Curr. Protoc. Stem Cell Biol. Chapter 1: Unit1C.3.; 2007
Nitsche A.; Becker M.; Junghahn I.; Aumann J.; Landt O.; Fichtner I.; Wittig B.; Siegert W. Quantification of human cells in NOD/SCID mice by duplex real-time polymerase-chain reaction. Haematologica. 86(7): 693–699; 2001.
O’Connor M. D.; Kardel M. D.; Eaves C. J. Functional assays for human embryonic stem cell pluripotency. Methods Mol. Biol. 690: 67–80; 2011.
Odorico J. S.; Kaufman D. S.; Thomson J. A. Multilineage differentiation from human embryonic stem cell lines. Stem Cells. 19(3): 193–204; 2001.
Park Y. B.; Kim Y. Y.; Oh S. K.; Chung S. G.; Ku S. Y.; Kim S. H.; Choi Y. M.; Moon S. Y. Alterations of proliferative and differentiation potentials of human embryonic stem cells during long-term culture. Exp. Mol. Med. 40(1): 98–108; 2008.
Skottman H.; Hovatta O. Culture conditions for human embryonic stem cells. Reproduction. 132(5): 691–698; 2006.
Spagnoli F. M.; Hemmati-Brivanlou A. Guiding embryonic stem cells towards differentiation: lessons from molecular embryology. Curr. Opin. Genet. Dev. 16(5): 469–475; 2006.
Stojkovic P.; Lako M.; Przyborski S.; Stewart R.; Armstrong L.; Evans J.; Zhang X.; Stojkovic M. Human-serum matrix supports undifferentiated growth of human embryonic stem cells. Stem Cells. 23(7): 895–902; 2005.
Wesselschmidt R. L. The teratoma assay: an in vivo assessment of pluripotency. Methods Mol. Biol. 767: 231–241; 2011.
Xu C.; Inokuma M. S.; Denham J.; Golds K.; Kundu P.; Gold J. D.; Carpenter M. K. Feeder-free growth of undifferentiated human embryonic stem cells. Nat. Biotechnol. 19(10): 971–974; 2001.
Zhou J.; Ou-Yang Q.; Li J.; Zhou X. Y.; Lin G.; Lu G. X. Human feeder cells support establishment and definitive endoderm differentiation of human embryonic stem cells. Stem Cells Dev. 17(4): 737–749; 2008.
Acknowledgments
This study was supported in part by the Federal Ministry of Education and Research, Germany (01GN0526, I.F.). SA002 cells, which are listed in the NIH Human Embryonic Stem Cell Registry, were supplied by Cellartis AB, Göteborg, Sweden. Mrs. Monika Becker provided expert technical assistance for this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: T. Okamoto
Rights and permissions
About this article
Cite this article
Keil, M., Siegert, A., Eckert, K. et al. Transcriptional expression profile of cultured human embryonic stem cells in vitro and in vivo. In Vitro Cell.Dev.Biol.-Animal 48, 165–174 (2012). https://doi.org/10.1007/s11626-012-9487-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-012-9487-y