Abstract
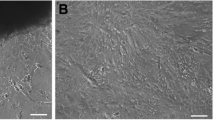
A new cell line, Asian sea bass brain (ASBB), was derived from the brain tissue of Asian sea bass Lates calcarifer. This cell line was maintained in Leibovitz L-15 media supplemented with 10% fetal bovine serum (FBS). The ASBB cell line was subcultured more than 60 times over a period of 15 mo. The ASBB cell line consists predominantly of fibroblastic-like cells and was able to grow at temperatures between 20°C and 30°C with an optimum temperature of 25°C. The growth rate of these cells increased as the proportion of FBS increased from 2% to 20% at 25°C with optimum growth at the concentrations of 10% or 15% FBS. Polymerase chain reaction products were obtained from ASBB cells and tissues of sea bass with primer sets of microsatellite markers of sea bass. An isolate of piscine nodavirus from juveniles of marine fish species tested positive by IQ2000 kit for viral nervous necrosis detection and was examined for its infectivity to a fish cell line of ASBB. A marine fish betanodavirus was tested to determine the susceptibility of this new cell line in comparison with commercial highly permissive SSN-1 cells. The ASBB cell line was found to be susceptible to nodavirus (RGNNV genotype), and the infection was confirmed by comparison cytopathic effect (CPE) with commercial SSN-1 and reverse transcriptase-polymerase chain reaction. A nodavirus was further elucidated by electron microscopy, and the virus tested was shown to induce CPE on ASBB cells with significant high titer. This suggests that the ASBB cell line has good potential for the isolation of fish viruses.








Similar content being viewed by others
References
Anderson M. J. Differences in growth of neurons from normal and regenerated teleost spinal cord in vitro. In Vitro Cell. Dev. Biol. 29A(2): 145–152; 1993.
Anderson M. J.; Waxman S. G. Neurogenesis in adult vertebrate spinal cord in situ and in vitro: a new model system. Ann. NY Acad. Sci. 457: 213–233; 1985.
Bejar J.; Borrego J. J.; Alvarez M. C. A continuous cell line from the cultured marine fish gilt-head seabream (Sparus aurata). Aquaculture 150: 143–153; 1997.
Breuil G.; Bonami J. R.; Pepin J. F.; Pichor Y. Viral infection (picrno-like virus) associated with mass mortalities in hatchery-reared seabass (Dicentrarchus labraz) larvae and juveniles. Aquaculture 97: 109–116; 1991.
Bryson S. P.; Joyce E. M.; Martell D. J.; Lee L. E.; Holt S. E.; Kales S. C.; Fujiki K.; Dixon B.; Bols N. C. A cell line (HEW) from embryos of haddock (Melanogrammus aeglefinius) and its capacity to tolerate environmental extremes. Mar. Biotechnol. 8: 641–653; 2006.
Buonocore F.; Forlenza M.; Randelli E.; Benedetti S.; Bossu P.; Meloni S.; Secombes C. J.; Mazzini M.; Scapigliati G. Biological activity of sea bass (Dicentrarchus labrax L.) recombinant interleukin-1beta. Mar. Biotechnol. 7: 609–617; 2005.
Chang S. F.; Ngoh G. H.; Kuch L. F. S.; Qin Q. W.; Chen C. L.; Lam T. J.; Sin Y. M. Developmental of a tropical marine fish cell line from Asian seabass (Lates calcarifer) for virus isolation. Aquaculture 192: 133–145; 2001.
Chapouton P.; Jagasia R.; Bally-Cuif L. Adult neurogenesis in non-mammalian vertebrates. BioEssays 29(8): 745–757; 2007.
Chenoweth S. F.; Hughes J. M.; Keenan C. P.; Lavery S. Concordance between dispersal and mitochondrial gene flow: isolation by distance in a tropical teleost Lates calcarifer (Australian barramundi). Heredity 80: 187–197; 1998.
Chi S. C.; Hu W. W.; Lo B. J. Establishment and characterization of a continuous cell line (GF-1) derived from grouper, Epinephelus coioides: a cell line susceptible to grouper nervous necrosis virus (GNNV). J. Fish Dis. 22: 173–182; 1999.
Chi S. C.; Wu Y. C.; Cheng T. M. Persistent infection of betanodavirus in a novel cell line derived from the brain tissue of barramundi Lates calcarifer. Dis Aquat Org 65(2): 91–98; 2005.
Cutrín J. M.; Dopazo C. P.; Thiéry R.; Leao P.; Olveira J. G.; Barja J. L.; Bandín I. Emergence of pathogenic betanodaviruses belonging to the SJNNV genogroup in farmed fish species from the Iberian Peninsula. J. Fish Dis. 30(4): 225–232; 2007.
De Boni U.; Seger M.; Scott J. W.; Crapper D. R. Neuron culture from adult goldfish. J. Neurobiol. 7: 495–512; 1976.
Fernandez R. D.; Yoshimizu M.; Ezura Y.; Kimura T. Comparative growth response of fish cell lines in different media, temperatures and sodium chloride concentrations. Gyobyo Kenkyu 28(1): 27–34; 1993.
Frerichs G. N.; Morgan D.; Hart D.; Skerrow C.; Roberts R. J.; Onions D. Spontaneously productive C-type retrovirus infection of fish cell lines. J. Gen. Virol. 72: 2537–2539; 1991.
Frerichs G. N.; Rodger H. D.; Peric Z. Cell culture isolation of piscine neuropathy nodavirus from juvenile sea bass, Dicentrarchus labrax. J. Gen. Virol. 77: 2067–2071; 1996.
Fryer J. L.; Lannon C. N. Three decades of fish cell culture: a current listing of cell lines derived from fish. J Tiss Cult Methods 16: 87–94; 1994.
Hinsch K.; Zupanc G. K. Isolation, cultivation, and differentiation of neural stem cells from adult fish brain. J Neurosci Methods 158: 75–88; 2006.
Hong Y.; Chen S. L.; Gui J. F.; Schartl M. Retention of the developmental pluriopotency inmedaka embroyonic stem cells after gene transfer and long-term drug selection towards for gene targeting in fish. Transgen Res 13: 41–50; 2004.
Hopert A.; Uphoff C. C.; Wirth M. et al. Specificity and sensitivity of polymerase chain reaction (PCR) in comparison with other methods for the detection of mycoplasma contamination in cell lines. J Immunol Methods 164: 91–100; 1993.
Kang M. S.; Oh M. J.; Kim Y. J.; Kawai K.; Jung S. J. Establishment and characterization of two cell lines derived from flounder, Paralichthys olivaceus (Temminck & Schlegel). J. Fish Dis. 26: 657–665; 2003.
Kaslin J.; Ganz J.; Brand M. Proliferation, neurogenesis and regeneration in the non-mammalian vertebrate brain. Philos. Trans. R. Soc. Lond. B Biol. Sci. 363(1489): 101–122; 2008.
Keenan C. P. Recent evolution of population-structure in Australian barramundi, Lates calcarifer (Bloch)—an example of isolation by distance in one-dimension. Austral J Mar Freshwater Res 45: 1123–1148; 1994.
Korsnes K. Nervous necrosis virus (NNV) in farmed Norwegian fish species. Ph.D. Thesis, Department of Molecular Biology, University of Bergen, Norway; 2008.
Lai Y. S.; John J. A.; Lin C. H.; Guo I. C.; Chen S. C.; Fang K.; Lin C. H.; Chang C. Y. Establishment of cell lines from a tropical grouper, Epinephelus awoara (Temminck & Schlegel), and their susceptibility to grouper irido- and nodaviruses. J. Fish Dis. 26(1): 31–42; 2003.
Larner A. J.; Johnson A. R.; Keynes R. J. Regeneration in the vertebrate central nervous system: phylogeny, ontogeny, and mechanisms. Biol. Rev. Camb. Philos. Soc. 70(4): 597–619; 1995.
Lo C. F.; Leu J. H.; Ho C. H.; Chen C. H.; Peng S. E.; Chen Y. T.; Yeh P. Y.; Huang C. J.; Wang C. H.; Kou G. H. Detection of baculovirus associated with white spot syndrome (WSSV) in penaeid shrimps using polymerase chain reaction. Dis Aquat Org 25: 133–141; 1996.
Munday B. L.; Langdon J. S.; Hyatt A.; Humphrey J. D. Mass mortality associated with a viral-induced vacuolating encephalopathy and retinopathy of larval and juvenile barramundi, Lates calcarifer Bloch. Aquaculture 103: 197–211; 1992.
Nicholson B. L.; Danner D. J.; Wu J. L. Three new continuous cell lines from marine fishes of Asia. In Vitro Cell. Dev. Biol. 23: 199–204; 1987.
Nishizawa T.; Mori K.; Nakai T.; Furusawa I.; Muroga K. Polymerase chain reaction (PCR) amplification of RNA of striped jack nervous necrosis virus (SJNNV). Dis. Aquat. Org. 18: 103–107; 1994.
Parameswaran V.; Ishaq Ahmed V. P.; Shukla R.; Bhonde R. R.; Sahul Hameed A. S. Development and characterization of two new cell lines from milkfish (Chanos chanos) and grouper (Epinephelus coioides) for virus isolation. Mar. Biotechnol. 9(2): 281–91; 2007.
Parameswaran V.; Shukla R.; Bhonde R. R.; Sahul Hameed A. S. Establishment of embryonic cell line from sea bass (Lates calcarifer) for virus isolation. J Virol Methods 137: 309–316; 2006a.
Parameswaran V.; Shukla R.; Bhonde R. R.; Sahul Hameed A. S. Splenic cell line from sea bass, Lates calcarifer: establishment and characterization. Aquaculture 261: 43–53; 2006b.
Payment P.; Trudel M. Methods and techniques in virology. Marcel Dekker Inc, New York, USA, p 33; 1993.
Sahul Hameed A. S.; Parameswaran V.; Bright Singh I. S.; Thirunavukkarasu A. R.; Bhonde R. R. Establishment and characterization of India's first marine fish cell line (SISK) from the kidney of sea bass (Lates calcarifer). Aquaculture 257: 92–103; 2006.
Tong S. L.; Lee H.; Miao H. Z. The establishment and partial characterization of a continuous fish cell line FG-9307 from the gill of flounder Paralichthys olivaceus. Aquaculture 156: 327–333; 1997.
Tong S. L.; Miao H. Z.; Li H. Three new continuous fish cell lines of SPH, SPS and RSBF derived from sea perch (Lateolabrax japaonicus) and red sea bream (Pagrosomus major). Aquaculture 169: 143–151; 1998.
Uphoff C. C.; Drexler H. G. Comparative PCR analysis for detection of mycoplasma infection in continuous cell lines. In Vitro Cell. Dev. Biol. Anim. 38: 79–85; 2002.
Wen C. M.; Cheng Y. H.; Huang Y. F.; Wang C. S. Isolation and characterization of a neural progenitor cell line from tilapia brain. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 149(2): 167–180; 2008.
Yue G. H.; Li Y.; Chao T. M.; Chou R.; Orban L. Novel microsatellites from Asian Sea Bass (Lates calcarifer) and their application to Broodstock analysis. Mar. Biotechnol. 4: 503–511; 2002.
Yue G. H.; Li Y.; Orban L. Characterization of microsatellites in the IGF-2 and GH genes of Asian seabass (Lates calcarifer). Mar. Biotechnol. 3: 1–3; 2001.
Cross References
Blin N.; Stafford D. W. A general method for isolation of high molecular weight DNA from eukaryoties. Nucleic Acids Res. 3: 2303–2308; 1976.
Reed L. J.; Muench H. A simple method of estimating fifty per cent end-points. Am J Hyg 27: 493–497; 1938.
Yu Z. N.; Kong X. Y.; Xie Z. Y. Studies on karyotypes of fishes of economic importance in coastal waters of Shandong Peninsula. J Fish Sci China 22(21): l–6; 1995.
Acknowledgments
The authors thank the National fisheries research center (Nafish), Penang, Malaysia for providing the nodavirus-infected fish samples. The authors thank also the laboratory staff of virology lab and Aquatic Animal Health Unit for providing the facilities to carry out this work. This work was funded by University Putra Malaysia (UPM) as a part of Ph.D. program at the Faculty of Veterinary Medicine, UPM University, Serdang, Selangor, Malaysia.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Denry Sato
Appendix
Appendix
0.075 M potassium chloride (hypotonic solution)
2.79 g KCl
500 ml dH2O
Phosphate buffer for Giemsa stain
0.469 g NaH2PO4
0.937 g NaH2PO4
1,000 ml dH2O
pH = 7
NTE
Tris–HCl 10 mM
NaCl 100 mM
EDTA 1.0 mM
Phenol:chloroform:iso-amyl alcohol (25:24:1)
10 ml Tris–phenol (yellow phase)
10 ml chloroform iso-amyl alcohol
Proteinase K (20 mg/ml)
100 mg proteinase K
5 ml DEPC treated water
Store at −20°C
Glutaraldehyde (GTA) 5%
Add 20 ml of 25% GTA to 80 ml of 0.2 M sodium cacodylate buffer, pH 7.4.
Rights and permissions
About this article
Cite this article
Hasoon, M.F., Daud, H.M., Abdullah, A.A. et al. Development and partial characterization of new marine cell line from brain of Asian sea bass Lates calcarifer for virus isolation. In Vitro Cell.Dev.Biol.-Animal 47, 16–25 (2011). https://doi.org/10.1007/s11626-010-9348-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-010-9348-5