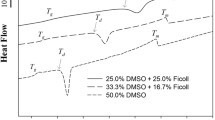
Abstract
With the development of cell-based assays and therapies, the purity of reagents used to grow and maintain cells has become much more important. In particular, the use of fetal calf serum for culturing cells presents a direct path for potential contamination of cell cultures. In recent years, much research has focused on the development of serum-free culturing systems, not only to alleviate difficulties due to availability and cost of fetal calf serum but also to prevent the transmission of potentially fatal diseases to human patients. Additionally, methods need to be developed for long-term storage of cell stocks that also reduce the risk of exposure to harmful diseases. As most methods employ fetal calf serum in their freezing formulations, solutions that avoid the use of fetal calf serum while providing equivalent or better recovery of cells upon thawing would be ideal. In this study, two vascular cell lines have been cryopreserved as adherent cell populations in two widely used cryoprotectants, dimethyl sulfoxide and 1,2-propanediol, and two vehicle solutions, Euro-Collins and Unisol-cryoprotectant vehicle specifically formulated for the maintenance of cell homeostasis at temperatures below 37° C. The addition of serum to these formulations was also evaluated to determine if its presence provided any additional benefit to the cells during cryopreservation. The results demonstrated that using vehicle solutions designed for lower temperatures produced viable cells that retained cell population viability values up to 75% of unfrozen controls. These results also demonstrated that including serum in the formulation provided no additional benefit to the cells and in some cases actually produced lower cell viability after cryopreservation. In conclusion, the development of solutions designed for low-temperature storage of cells provides a viable alternative to more conventional cryopreservation protocols and eliminates the necessity of including serum in these formulations.



Similar content being viewed by others
References
Asher, D. M. The transmissible spongiform encephalopathy agents: concerns and responses of United States regulatory agencies in maintaining the safety of biologics. Dev. Biol Stand. 100:103–118; 1999.
Baust, J. M. Molecular mechanisms of cellular demise associated with cryopreservation failure. Cell Preservation Technologies 1:17–32; 2002.
Baust, J. M.; van Buskirk, R. G.; Baust, J. G. Improved Cryopreservation Outcome is Facilitated by an Intracellular-Type medium and Inhibition of Apoptosis. Journal of the Biomedical Engineering Society 1:206–207; 1999.
Baust, J. M.; Van Buskirk, R.; Baust, J. G. Gene activation of the apoptotic caspase cascade following cryogenic storage. Cell Preservation Technologies 1:63–80; 2002a.
Baust, J. M.; Van Buskirk, R.; Baust, J. G. Modulation of the cryopreservation cap: elevated survival with reduced dimethyl sulfoxide concentration. Cryobiology 45:97–108; 2002b.
Baust, J. M.; Vogel, M. J.; van Buskirk, R. G.; Baust, J. G. A Molecular Basis of Cryopreservation Failure and its Modulation to Improve Cell Survival. Cell Transplantation 10:561–571; 2001.
Bielanski, A.; Bergeron, H.; Lau, P. C.; Devenish, J. Microbial contamination of embryos and semen during long term banking in liquid nitrogen. Cryobiology 46:146–152; 2003.
Bradley, R. Bovine spongiform encephalopathy and its relationship to the variant form of Creutzfeldt–Jakob disease. Contrib. Microbiol. 11:146–185; 2004.
Brown, P.; Will, R. G.; Bradley, R.; Asher, D. M.; Detwiler, L. Bovine spongiform encephalopathy and variant Creutzfeldt–Jakob disease: background, evolution, and current concerns. Emerg. Infect. Dis. 7:6–16; 2001.
Campbell, L. H.; Taylor, M. J.; Brockbank, K. G. M. U.S. Patent #6,596,531 Two stage method for thawing cryopreserved cells; 2003.
Committee for Veterinary Medicinal Products. Guideline on Requirements and Controls Applied to Bovine Serum used in the Production of Immunological Veterinary Medicinal Products. EMEA/CVMP/743-00; 2001.
Croes, E. A. and van Duijn, C. M. Variant Creutzfeldt–Jakob disease. Eur J Epidemiol 18:473–477; 2003.
Erickson, G. A.; Bolin, S. R.; Landgraf, J. G. Viral contamination of fetal bovine serum used for tissue culture: risks and concerns. Dev. Biol Stand. 75:173–175; 1991.
Erickson, G. A.; Landgraf, J. G.; Wessman, S. J.; Koski, T. A.; Moss, L. M. Detection and elimination of adventitious agents in continuous cell lines. Dev. Biol Stand. 70:59–66; 1989.
FDA International Conference on Harmonisation. Guidance on quality of biotechnological/biological products: derivation and characterization of cell substrates used for production of biotechnological/biological products; availability. Notice. Food and Drug Administration, HHS. Fed. Regist. 63:50244–50249; 1998.
Food and Drug Administration. BSE Estimating Risks for vCJD in Vaccines Using Bovine-Derived Materials. http://www.fda.gov/cber/bse/risk.htm; 2001.
Froud, S. J. The development, benefits and disadvantages of serum-free media. Dev. Biol Stand. 99:157–166; 1999.
Hubálek, Z. Protectants used in the cryopreservation of microorganisms. Cryobiology 46:205–229; 2003.
Irani, D. N. and Johnson, R. T. Diagnosis and prevention of bovine spongiform encephalopathy and variant Creutzfeldt–Jakob disease. Annu Rev Med 54:305–319; 2003.
Ironside, J. W. and Head, M. W. Variant Creutzfeldt–Jakob disease and its transmission by blood. J Thromb Haemost 1:1479–1486; 2003.
Jayme, D. W. An animal origin perspective of common constituents of serum-free medium formulations. Dev. Biol Stand. 99:181–187; 1999.
Jennings, A. Detecting viruses in sera: methods used and their merits. Dev. Biol Stand. 99:51–59; 1999.
Lakey, J. R. T.; Rajotte, R. V.; Fedorow, C. A.; Taylor, M. J. Islet Cryopreservation using Intracellular Preservation Solutions. Cell Transplant 10:583–589; 2001.
Levings, R. L. and Wessman, S. J. Bovine viral diarrhea virus contamination of nutrient serum, cell cultures and viral vaccines. Dev. Biol Stand. 75:177–181; 1991.
Makoschey, B.; van Gelder, P. T.; Keijsers, V.; Goovaerts, D. Bovine viral diarrhoea virus antigen in foetal calf serum batches and consequences of such contamination for vaccine production. Biologicals 31:203–208; 2003.
Merten, O. W. Safety issues of animal products used in serum-free media. Dev. Biol Stand. 99:167–180; 1999.
Nuttall, P. A.; Luther, P. D.; Stott, E. J. Viral contamination of bovine foetal serum and cell cultures. Nature 266:835–837; 1977.
Pegg, D. E.; Wusteman, M. C.; Boylan, S. Fractures in cryopreserved elastic arteries: mechanism and prevention. Cryobiology 33:658–659; 1996.
Schlumpf, R.; Morel, Ph.; Loveras, J. J.; Condie, R. M.; Matas, A.; Kurle, J.; Najarian, J. S.; Sutherland, D. E. R. Examination of the Role of the Colloids Hydroxyethylstarch, Dextran, Human Albumin, and Plasma Proteins in a Modified UW Solution. Transplantation Proceedings 23:2362–2365; 1991.
Shiiya, N.; Paul, M.; Bevenuti, C.; Astier, A.; Ferrer, M.-J.; Loisance, D. A. Lactobionate-based Extracellular-type Solution for Donor Heart Preservation. Journal of Heart and Lung Transplantation 12:476–483; 1993.
Southard, J. H.; van Gulik, T. M.; Ametani, M. S.; Vreugdenhil, P. K.; Lindell, S. L.; Pienaar, B. L.; Belzer, F. O. Important Components of the UW Solution. Transplantation 49:251–257; 1990.
Sumimoto, R.; Dohi, K.; Urushihara, T.; Jamieson, N. V.; Ito, H.; Sumimoto, K.; Fukuda, Y. An Examination of the Effects of Solutions Containing Histidine and Lactobionate for Heart, Pancreas, and Liver Preservation in the Rat. Transplantation 53:1206–1210; 1992.
Taylor, M. J. The Role of pH* and Buffer Capacity in the Recovery of Function of Smooth Muscle cooled to −13 C in Unfrozen Media. Cryobiology 19:585–601; 1982.
Taylor, M. J. Biology of cell survival in the cold: The Basis for Biopreservation of Tissues and Organs. In: Baust, J. G. ed. Advances in Biopreservation. Boca Raton: CRC Press; 2004.
Taylor, M. J.; Campbell, L. H.; Rutledge, R. N.; Brockbank, K. G. M. Comparison of Unisol(TM) with EuroCollins Solution as a Vehicle Solution for Cryoprotectants. Transplantation Proceedings 33:677–679; 2001.
Taylor, M. J. and Hunt, C. J. A new preservation solution for storage of corneas at low temperatures. Current Eye Research 4:963–973; 1985.
Taylor, M. J. and Pignat, Y. Practical acid dissociation constants, temperature coefficients and buffer capacities for some biological buffers in solutions containing dimethyl sulfoxide between 25 and −12° C. Cryobiology 19:99–109; 1982.
Taylor, M. J.; Walter, C. A.; Elford, B. C. The pH-dependent recovery of smooth muscle from storage at −13 C in unfrozen media. Cryobiology 15:452–460; 1978.
Trevitt, C. R. and Singh, P. N. Variant Creutzfeldt–Jakob disease: pathology, epidemiology, and public health implications. Am. J Clin. Nutr. 78:651S–656S; 2003.
Ward, H. J.; Head, M. W.; Will, R. G.; Ironside, J. W. Variant Creutzfeldt–Jakob disease. Clin. Lab Med 23:87–108; 2003.
Weber, D. J. Biosafety Considerations for Cell-based Therapies. BioPharm International; 2004.
Wessman, S. J. and Levings, R. L. Benefits and risks due to animal serum used in cell culture production. Dev. Biol Stand. 99:3–8; 1999.
Will, R. G.; Ironside, J. W.; Zeidler, M.; Cousens, S. N.; Estibeiro, K.; Alperovitch, A.; Poser, S.; Pocchiari, M.; Hofman, A.; Smith, P. G. A new variant of Creutzfeldt–Jakob disease in the UK. The Lancet 347:921–925; 1996.
Zabal, O.; Kobrak, A. L.; Lager, I. A.; Schudel, A. A.; Weber, E. L. Contamination of bovine fetal serum with bovine viral diarrhea virus. Rev Argent Microbiol. 32:27–32; 2000.
Acknowledgement
This work was supported in part by a cooperative agreement (97-07-0039) with the U.S. Department of Commerce, National Institute of Standards & Technology—Advanced Technology Program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Denry Sato
Rights and permissions
About this article
Cite this article
Campbell, L.H., Brockbank, K.G.M. Serum-free solutions for cryopreservation of cells. In Vitro Cell.Dev.Biol.-Animal 43, 269–275 (2007). https://doi.org/10.1007/s11626-007-9039-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-007-9039-z