Abstract
Background
Postoperative oral antiviral treatment with nucleoside or nucleotide analogues can suppress viral replication and reduce tumour recurrence for patients with hepatitis b virus–related hepatocellular carcinoma (HBV-related HCC) after curative resection. However, the superior antiviral treatment is still unclear. We conducted this study to investigate the different effects of nucleotide and nucleoside analogues on the prognosis of HBV-related HCC after curative resection.
Methods
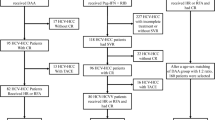
From February 2007 to February 2016, 487 consecutive patients with newly diagnosed HCC according to the Milan criteria who underwent R0 resection were enrolled according to the inclusion and exclusion criteria. According to their postoperative antiviral treatment, they were divided into the nucleotide group (NtA, n = 111) and the nucleoside group (NsA, n = 376).
Results
The baseline characteristics, serologic parameters, tumour characteristics, and operative data of the 2 groups were comparable. Nucleotide analogue use significantly decreased HCC recurrence (P = 0.028) and HCC-related death (P = 0.004), with hazard ratios (HRs) of 0.685 (95% CI, 0.484 to 0.971, P = 0.033) and 0.507 (95% CI, 0.310 to 0.830, P = 0.004), respectively, in multivariate Cox analyses. After the study patients were stratified according to three variables, we found that nucleotide analogue use was significantly associated with increased disease-free and overall survival among patients with cirrhosis, HBeAg-negative patients, and patients with positive HBV-DNA.
Conclusions
In patients with HBV-related HCC, nucleotide analogues but not nucleoside analogues significantly reduced HCC recurrence and improved overall survival after R0 hepatic resection.





Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA: a cancer journal for clinicians. 2011;61(2):69–90. https://doi.org/10.3322/caac.20107.
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F et al. Cancer statistics in China, 2015. CA: a cancer journal for clinicians. 2016;66(2):115–32. doi:https://doi.org/10.3322/caac.21338.
Roayaie S, Blume IN, Thung SN, Guido M, Fiel MI, Hiotis S et al. A system of classifying microvascular invasion to predict outcome after resection in patients with hepatocellular carcinoma. Gastroenterology. 2009;137(3):850–5. https://doi.org/10.1053/j.gastro.2009.06.003.
EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. Journal of hepatology. 2012;56(4):908–43. https://doi.org/10.1016/j.jhep.2011.12.001.
Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53(3):1020–2. https://doi.org/10.1002/hep.24199.
Hung IF, Poon RT, Lai CL, Fung J, Fan ST, Yuen MF. Recurrence of hepatitis B-related hepatocellular carcinoma is associated with high viral load at the time of resection. Am J Gastroenterol. 2008;103(7):1663–73.
Hoshida Y. Risk of recurrence in hepatitis B-related hepatocellular carcinoma: impact of viral load in late recurrence. Journal of hepatology. 2009;51(5):842–4. https://doi.org/10.1016/j.jhep.2009.08.003.
Yin J, Li N, Han Y, Xue J, Deng Y, Shi J et al. Effect of antiviral treatment with nucleotide/nucleoside analogs on postoperative prognosis of hepatitis B virus-related hepatocellular carcinoma: a two-stage longitudinal clinical study. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2013;31(29):3647–55. https://doi.org/10.1200/JCO.2012.48.5896.
Huang G, Lau WY, Wang ZG, Pan ZY, Yuan SX, Shen F et al. Antiviral therapy improves postoperative survival in patients with hepatocellular carcinoma: a randomized controlled trial. Annals of surgery. 2015;261(1):56–66. https://doi.org/10.1097/SLA.0000000000000858.
Wong JS, Wong GL, Tsoi KK, Wong VW, Cheung SY, Chong CN et al. Meta-analysis: the efficacy of anti-viral therapy in prevention of recurrence after curative treatment of chronic hepatitis B-related hepatocellular carcinoma. Alimentary pharmacology & therapeutics. 2011;33(10):1104–12. https://doi.org/10.1111/j.1365-2036.2011.04634.x.
Murata K, Tsukuda S, Suizu F, Kimura A, Sugiyama M, Watashi K et al. Immunomodulatory Mechanism of Acyclic Nucleoside Phosphates in Treatment of Hepatitis B Virus Infection. Hepatology. 2019. https://doi.org/10.1002/hep.30956.
EASL INTERNATIONAL CONSENSUS CONFERENCE ON HEPATITIS B13–14 September, 2002Geneva, Switzerland. Journal of hepatology. 2003;38(4):533–40. https://doi.org/10.1016/s0168-8278(03)00083-7.
EASL Clinical Practice Guidelines: Management of chronic hepatitis B. Journal of hepatology. 2009;50(2):227–42. https://doi.org/10.1016/j.jhep.2008.10.001.
Liaw YF, Leung N, Guan R, Lau GK, Merican I, McCaughan G et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2005 update. Liver international : official journal of the International Association for the Study of the Liver. 2005;25(3):472–89. https://doi.org/10.1111/j.1478-3231.2005.01134.x.
Liaw YF, Leung N, Kao JH, Piratvisuth T, Gane E, Han KH et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2008 update. Hepatology international. 2008;2(3):263–83. https://doi.org/10.1007/s12072-008-9080-3.
Liaw Y-F, Kao J-H, Piratvisuth T, Chan HLY, Chien R-N, Liu C-J et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2012 update. Hepatology international. 2012;6(3):531–61. https://doi.org/10.1007/s12072-012-9365-4.
Lok AS, McMahon BJ. Chronic hepatitis B. Hepatology. 2007;45(2):507–39. https://doi.org/10.1002/hep.21513.
Lok AS, McMahon BJ. Chronic hepatitis B: update 2009. Hepatology. 2009;50(3):661–2. https://doi.org/10.1002/hep.23190.
Poon D, Anderson BO, Chen LT, Tanaka K, Lau WY, Van Cutsem E et al. Management of hepatocellular carcinoma in Asia: consensus statement from the Asian Oncology Summit 2009. The Lancet Oncology. 2009;10(11):1111–8. https://doi.org/10.1016/S1470-2045(09)70241-4.
EASL–EORTC Clinical Practice Guidelines: Management of hepatocellular carcinoma. Journal of hepatology. 2012;56(4):908–43. https://doi.org/10.1016/j.jhep.2011.12.001.
Heimbach J, Kulik LM, Finn R, Sirlin CB, Abecassis M, Roberts LR et al. Aasld guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2017. https://doi.org/10.1002/hep.29086.
Murata K, Asano M, Matsumoto A, Sugiyama M, Nishida N, Tanaka E et al. Induction of IFN–lambda3 as an additional effect of nucleotide, not nucleoside, analogues: a new potential target for HBV infection. Gut. 2016. https://doi.org/10.1136/gutjnl-2016-312653.
Samuel CE. Antiviral actions of interferons. Clinical microbiology reviews. 2001;14(4):778–809, table of contents. https://doi.org/10.1128/CMR.14.4.778-809.2001.
Wang L, Tang ZY, Qin LX, Wu XF, Sun HC, Xue Q et al. High-dose and long-term therapy with interferon-alfa inhibits tumor growth and recurrence in nude mice bearing human hepatocellular carcinoma xenografts with high metastatic potential. Hepatology. 2000;32(1):43–8. https://doi.org/10.1053/jhep.2000.8525.
Wang L, Wu WZ, Sun HC, Wu XF, Qin LX, Liu YK et al. Mechanism of interferon alpha on inhibition of metastasis and angiogenesis of hepatocellular carcinoma after curative resection in nude mice. Journal of gastrointestinal surgery : official journal of the Society for Surgery of the Alimentary Tract. 2003;7(5):587–94.
von Marschall Z, Scholz A, Cramer T, Schafer G, Schirner M, Oberg K et al. Effects of interferon alpha on vascular endothelial growth factor gene transcription and tumor angiogenesis. J Natl Cancer I. 2003;95(6):437–48.
Dinney CP, Bielenberg DR, Perrotte P, Reich R, Eve BY, Bucana CD et al. Inhibition of basic fibroblast growth factor expression, angiogenesis, and growth of human bladder carcinoma in mice by systemic interferon-alpha administration. Cancer research. 1998;58(4):808-14.
Kotenko SV. IFN-lambdas. Current opinion in immunology. 2011;23(5):583–90. https://doi.org/10.1016/j.coi.2011.07.007.
Kotenko SV, Gallagher G, Baurin VV, Lewis-Antes A, Shen M, Shah NK et al. IFN-lambdas mediate antiviral protection through a distinct class II cytokine receptor complex. Nature immunology. 2003;4(1):69–77. https://doi.org/10.1038/ni875.
Huang G, Yang Y, Shen F, Pan ZY, Fu SY, Lau WY et al. Early viral suppression predicts good postoperative survivals in patients with hepatocellular carcinoma with a high baseline HBV-DNA load. Annals of surgical oncology. 2013;20(5):1482–90. https://doi.org/10.1245/s10434-012-2803-7.
Wai CT, Chu CJ, Hussain M, Lok AS. HBV genotype B is associated with better response to interferon therapy in HBeAg(+) chronic hepatitis than genotype C. Hepatology. 2002;36(6):1425–30.
Yang HI, Yeh SH, Chen PJ, Iloeje UH, Jen CL, Su J et al. Associations between hepatitis B virus genotype and mutants and the risk of hepatocellular carcinoma. J Natl Cancer I. 2008;100(16):1134–43. https://doi.org/10.1093/jnci/djn243.
Brunetto MR, Marcellin P, Cherubini B, Yurdaydin C, Farci P, Hadziyannis SJ et al. Response to peginterferon alfa-2a (40KD) in HBeAg-negative CHB: on-treatment kinetics of HBsAg serum levels vary by HBV genotype. Journal of hepatology. 2013;59(6):1153–9. https://doi.org/10.1016/j.jhep.2013.07.017.
Acknowledgements
The authors thank American Journal of Experts (http://www.aje.com/) for English language editing.
Funding
This work was in part supported by grant from the National Science and Technology Key Projects (2017ZX10203207–003-002), the National Natural Science Foundation of China (81900463), the Science and Technological Supports Project of Sichuan Province (2020YFS0133), the Science and Technology Project of Chengdu (2018-YF05–01460-SN), and Post-Doctor Research Project, West China Hospital, Sichuan University (2019HXBH004).
Author information
Authors and Affiliations
Contributions
Conception: Zhang XY, Wen TF, and Li C. Acquisition of data: Zhang XY and Li C. Analysis of data: all authors. Writing: Zhang XY. Critical revision: all authors. Final approval: all authors.
Corresponding author
Ethics declarations
This study complied with the standards of the Helsinki Declaration and current ethical guidelines and was approved by the Ethics Committee of West China Hospital, Sichuan University.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 16.6 MB)
Rights and permissions
About this article
Cite this article
Zhang, X., Li, C., Wen, T. et al. The Different Effects of Nucleotide and Nucleoside Analogues on the Prognosis of HBV-Related HCC After Curative Resection. J Gastrointest Surg 25, 1419–1429 (2021). https://doi.org/10.1007/s11605-020-04633-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-020-04633-3