Abstract
Background
Accurate preoperative assessment of hepatic functional reserve is essential for conducting a safe hepatectomy. In recent years, aspartate aminotransferase-to-platelet ratio index (APRI) has been used as a noninvasive model for assessing fibrosis stage, hepatic functional reserve, and prognosis after hepatectomy with a high level of accuracy. The purpose of this research was to evaluate the clinical value of combining APRI with standardized future liver remnant (sFLR) for predicting severe post-hepatectomy liver failure (PHLF) in patients with hepatocellular carcinoma (HCC).
Methods
Six hundred thirty-seven HCC patients who had undergone hepatectomy were enrolled in this study. The performance of the Child-Pugh (CP) grade, model for end-stage liver disease (MELD), APRI, sFLR, and APRI-sFLR in predicting severe PHLF was assessed using the area under the ROC curve (AUC).
Results
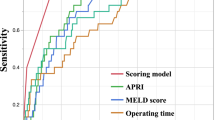
Severe PHLF was found to have developed in 101 (15.9%) patients. Multivariate logistic analyses identified that prealbumin, cirrhosis, APRI score, sFLR, and major resection were significantly associated with severe PHLF. The AUC values of the CP, MELD, APRI, and sFLR were 0.626, 0.604, 0.725, and 0.787, respectively, indicating that the APRI and sFLR showed significantly greater discriminatory abilities than CP and MELD (P < 0.05 for all). After APRI was combined with sFLR, the AUC value of APRI-sFLR for severe PHLF was 0.816, which greatly improved the prediction accuracy, compared with APRI or sFLR alone (P < 0.05 for all). Stratified analysis using the status of cirrhosis and extent of resection yielded similar results. Moreover, the incidence and grade of PHLF were significantly different among the three risk groups.
Conclusion
The combination of APRI and sFLR can be considered to be a predictive factor with increased accuracy for severe PHLF in HCC patients, compared with CP grade, MELD, APRI, or sFLR alone.



Similar content being viewed by others
Abbreviations
- HCC:
-
hepatocellular carcinoma
- PHLF:
-
post-hepatectomy liver failure
- CP:
-
Child-Pugh
- MELD:
-
model for end-stage liver disease
- APRI:
-
aspartate aminotransferase-to-platelet ratio index
- FLR:
-
future liver remnant
- eTLV:
-
estimated total liver volume
- sFLR:
-
standardized future liver remnant
- CSPH:
-
clinically significant portal hypertension
- ROC:
-
receiver operating characteristic
- AUC:
-
area under the ROC curve
References
Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin 2017;67(1):7–30.
Heimbach JK, Kulik LM, Finn RS, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 2018;67(1):358–80.
Peter R. Galle AF, Josep M, Llovet VM, Fabio Piscaglia JR, Peter Schirmacher VV. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol 2018;69(1):182–236.
Omata M, Cheng AL, Kokudo N, et al. Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: a 2017 update. Hepatol Int 2017;11(4):317–70.
Rahbari NN, Garden OJ, Padbury R, et al. Posthepatectomy liver failure: a definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery 2011;149(5):713–24.
Hernaez R, Solà E, Moreau R, Ginès P. Acute-on-chronic liver failure: an update. Gut 2017;66(3):541–53.
Wang YY, Zhong JH, Su ZY, et al. Albumin-bilirubin versus Child-Pugh score as a predictor of outcome after liver resection for hepatocellular carcinoma. Br J Surg 2016;103(6):725–34.
Cieslak KP, Runge JH, Heger M, Stoker J, Bennink RJ, van Gulik TM. New perspectives in the assessment of future remnant liver. Dig Surg 2014;31(4–5):255–68.
Durand F, Valla D. Assessment of the prognosis of cirrhosis: Child-Pugh versus MELD. J Hepatol 2005;42 Suppl(1):S100–7.
Pugh RN, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg 1973;60(8):646–9.
Wang YY, Zhao XH, Ma L, et al. Comparison of the ability of Child-Pugh score, MELD score, and ICG-R15 to assess preoperative hepatic functional reserve in patients with hepatocellular carcinoma. J Surg Oncol 2018;118(3):440–5.
Malinchoc M, Kamath PS, Gordon FD, Peine CJ, Rank J, ter B PC. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology 2000;31(4):864–71.
Liu PH, Hsu CY, Hsia CY, et al. ALBI and PALBI grade predict survival for HCC across treatment modalities and BCLC stages in the MELD Era. J Gastroenterol Hepatol 2017;32(4):879–86.
Wiesner R, Edwards E, Freeman R, et al. Model for end-stage liver disease (MELD) and allocation of donor livers. Gastroenterology 2003;124(1):91–6.
Wai CT, Greenson JK, Fontana RJ, et al. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology 2003;38(2):518–26.
Lin ZH, Xin YN, Dong QJ, et al. Performance of the aspartate aminotransferase-to-platelet ratio index for the staging of hepatitis C-related fibrosis: an updated meta-analysis. Hepatology 2011;53(3):726–36.
Shen SL, Fu SJ, Chen B, et al. Preoperative aspartate aminotransferase to platelet ratio is an independent prognostic factor for hepatitis B-induced hepatocellular carcinoma after hepatic resection. Ann Surg Oncol 2014;21(12):3802–9.
Cheng J, Zhao P, Liu J, Liu X, Wu X. Preoperative aspartate aminotransferase-to-platelet ratio index (APRI) is a predictor on postoperative outcomes of hepatocellular carcinoma. Medicine (Baltimore) 2016;95(48):e5486.
Ichikawa T, Uenishi T, Takemura S, et al. A simple, noninvasively determined index predicting hepatic failure following liver resection for hepatocellular carcinoma. J Hepatobiliary Pancreat Surg 2009;16(1):42–8.
Xiao G, Yang J, Yan L. Comparison of diagnostic accuracy of aspartate aminotransferase to platelet ratio index and fibrosis-4 index for detecting liver fibrosis in adult patients with chronic hepatitis B virus infection: a systemic review and meta-analysis. Hepatology 2015;61(1):292–302.
Hann HW, Wan S, Lai Y, et al. Aspartate aminotransferase to platelet ratio index as a prospective predictor of hepatocellular carcinoma risk in patients with chronic hepatitis B virus infection. J Gastroenterol Hepatol 2015;30(1):131–8.
Hagström H, Talbäck M, Andreasson A, Walldius G, Hammar N. Ability of Noninvasive Scoring Systems to Identify Individuals in the Population at Risk for Severe Liver Disease. Gastroenterology 2019.
Tang T, Qiu JL, Li GW, et al. Aspartate aminotransferase-to-platelet ratio predicts response to transarterial chemoembolisation and prognosis in hepatocellular carcinoma patients. Clin Radiol 2018;73(3):259–65.
Mai RY, Ye JZ, Long ZR, et al. Preoperative aspartate aminotransferase-to-platelet-ratio index as a predictor of posthepatectomy liver failure for resectable hepatocellular carcinoma. Cancer Manag Res 2019;11:1401–14.
Schindl MJ, Redhead DN, Fearon KC, Garden OJ, Wigmore SJ. The value of residual liver volume as a predictor of hepatic dysfunction and infection after major liver resection. Gut 2005;54(2):289–96.
Kishi Y, Abdalla EK, Chun YS, et al. Three hundred and one consecutive extended right hepatectomies: evaluation of outcome based on systematic liver volumetry. Ann Surg 2009;250(4):540–8.
Berzigotti A, Reig M, Abraldes JG, Bosch J, Bruix J. Portal hypertension and the outcome of surgery for hepatocellular carcinoma in compensated cirrhosis: a systematic review and meta-analysis. Hepatology 2015;61(2):526–36.
Pol B, Campan P, Hardwigsen J, Botti G, Pons J, Le TYP. Morbidity of major hepatic resections: a 100-case prospective study. Eur J Surg 1999;165(5):446–53.
Camp RL, Dolled-Filhart M, Rimm DL. X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res 2004;10(21):7252–9.
Lai J, Wang H, Peng J, Chen P, Pan Z. Establishment and external validation of a prognostic model for predicting disease-free survival and risk stratification in breast cancer patients treated with neoadjuvant chemotherapy. Cancer Manag Res 2018;10:2347–56.
Kamath PS, Wiesner RH, Malinchoc M, et al. A model to predict survival in patients with end-stage liver disease. Hepatology 2001;33(2):464–70.
Yang T, Liu K, Liu CF, et al. Impact of postoperative infective complications on long-term survival after liver resection for hepatocellular carcinoma. Br J Surg 2019;106(9):1228–36.
Miyazaki S, Takasaki K, Yamamoto M, Tsugita M, Otsubo T. Liver regeneration and restoration of liver function after partial hepatectomy: the relation of fibrosis of the liver parenchyma. Hepatogastroenterology 1999;46(29):2919–24.
Shirabe K, Shimada M, Gion T, et al. Postoperative liver failure after major hepatic resection for hepatocellular carcinoma in the modern era with special reference to remnant liver volume. J Am Coll Surg 1999;188(3):304–9.
Olthof PB, Wiggers JK, Groot KB, et al. Postoperative Liver Failure Risk Score: Identifying Patients with Resectable Perihilar Cholangiocarcinoma Who Can Benefit from Portal Vein Embolization. J Am Coll Surg 2017;225(3):387–94.
Urata K, Kawasaki S, Matsunami H, et al. Calculation of child and adult standard liver volume for liver transplantation. Hepatology 1995;21(5):1317–21.
Zou H, Wen Y, Yuan K, Miao XY, Xiong L, Liu KJ. Combining albumin-bilirubin score with future liver remnant predicts post-hepatectomy liver failure in HBV-associated HCC patients. Liver Int 2018;38(3):494–502.
Funding
The study was supported by the National Science Foundation of China Youth Fund Project (81803007), National Natural Science Foundation of China (81460426), 66th Chinese Post-Doctoral Science Foundation Project (2019M663412), Project of GuangXi Natural Science Foundation (2019JJA140151), Regional Science Fund Project of China Natural Science Foundation (81660498), Youth Talent Fund Project of Guangxi Natural Science Foundation (Nos. 2016GXNSFBA380090, 2018GXNSFBA281030, and 2018GXNSFBA281091), and Guangxi Medical and Health Appropriate Technology Development and Application Project (Nos. S2017101 and S2018062).
Author information
Authors and Affiliations
Contributions
Conception: Rong-yun Mai, Jie Zeng, Le-qun Li, and Jia-zhou Ye; study design: Rong-yun Mai, Guo-bin Wu, Fei-xiang Wu, Liang Ma, Bang-de Xiang, and Jia-zhou Ye; administrative support: Le-qun Li; data collection and acquisition: Rong-yun Mai, Jie Zeng, Hua-ze Lu, Xue-min Piao, Xing Gao; data analysis: Rong-yun Mai, Jie Zeng, Hua-ze Lu, Hua-ze Lu, Rong Liang, and Yan Lin; manuscript preparation: Rong-yun Mai, Jie Zeng, and Hua-ze Lu; critical revision: Rong-yun Mai and Jia-zhou Ye; final approval of manuscript: all authors.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 32 kb)
Supplementary Figure 1
Three-dimensional reconstruction and virtual hepatectomy. Red, hepatic artery; dark blue, hepatic vein; lake blue, portal vein; khaki, tumor; brown, hepatic parenchyma; dark green, gall bladder. (A) Computed tomography scan of the left HCC. (B) Volume of the re-constructional liver. (C) Virtual hepatectomy prior to surgery. (D) Remnant liver volume after virtual hepatectomy. Abbreviation: HCC, hepatocellular carcinoma. (PNG 3740 kb)
Supplementary Figure 2
Relationship of incidence and PHLF grade based on risk group stratification assessed using the APRI-sFLR score in the subgroups of HCC patients. Abbreviation: APRI-sFLR, combination of APRI and sFLR; PHLF, posthepatectomy liver failure; HCC, hepatocellular carcinoma. (PNG 507 kb)
Rights and permissions
About this article
Cite this article
Mai, Ry., Zeng, J., Lu, Hz. et al. Combining Aspartate Aminotransferase-to-Platelet Ratio Index with Future Liver Remnant to Assess Preoperative Hepatic Functional Reserve in Patients with Hepatocellular Carcinoma. J Gastrointest Surg 25, 688–697 (2021). https://doi.org/10.1007/s11605-020-04575-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-020-04575-w