Abstract
Background
Expression of CD3+ T cells, CD8+ cytotoxic T cells, CD45RO+ memory T cells, and FOXP3+ regulatory T cells at the invasive margin (IM) and tumor center (TC) has correlated with survival in gastric adenocarcinoma (GA) patients from East Asia, independent of anatomic staging. The reason for improved survival in East Asians compared with Western patients is a subject of debate. This study examined the immune profiles of a cohort of Western patients with GA, and their association with overall survival (OS).
Methods
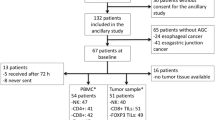
Immunohistochemistry (IHC) using antibodies to CD3, CD4, CD8, CD45RO, and FOXP3 was performed on a randomly selected resected GA specimens from 88 Western patients. Cutoffs for high or low expression of each marker were determined with maximally selected rank statistics, and multivariable Cox proportional-hazards models constructed to evaluate the relationship between OS and expression of each marker at the IM and TC.
Results
Immune cell density was independent of anatomic staging. High expression of CD3, CD4, CD8, and CD45RO at the IM along with CD4 and FOXP3 at the TC were associated with improved OS. A combined marker of CD3, CD8, CD45RO, and FOXP3 associated with OS in East Asian GA was also validated.
Discussion
This is the first report in US patients to demonstrate that high expression of multiple subsets of T lymphocytes in GA is associated with better OS independent of clinical factors and anatomic stage. Further evaluation of immune-modulating mechanisms may explain survival differences between Western and Eastern patients and provide opportunity for novel treatments.



Similar content being viewed by others
Abbreviations
- GA:
-
Gastric adenocarcinoma
- TIL:
-
Tumor-infiltrating lymphocyte
- IHC:
-
Immunohistochemistry
- OS:
-
Overall survival
- IM:
-
Invasive margin
- TC:
-
Tumor center
References
Noone AM H.N., Krapcho M, Miller D, Brest A, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, Chen HS, Feuer EJ, Cronin KA: SEER Cancer Statistics Review. 2018; https://seer.cancer.gov/csr/1975_2015/. Accessed December, 2018.
Kweon S.-S.: Updates on Cancer Epidemiology in Korea, 2018. Chonnam Medical Journal. 2018; 54:90–100.
Allemani C., Matsuda T., Di Carlo V., Harewood R., Matz M., Niksic M., et al.: Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet. 2018; 391:1023–1075.
Jun J.K., Choi K.S., Lee H.Y., Suh M., Park B., Song S.H., et al.: Effectiveness of the Korean National Cancer Screening Program in Reducing Gastric Cancer Mortality. Gastroenterology. 2017; 152:1319–1328 e1317.
Lee S., Jun J.K., Suh M., Park B., Noh D.K., Jung K.W., et al.: Gastric cancer screening uptake trends in Korea: results for the National Cancer Screening Program from 2002 to 2011: a prospective cross-sectional study. Medicine (Baltimore). 2015; 94:e533.
Saumoy M., Schneider Y., Shen N., Kahaleh M., Sharaiha R.Z., Shah S.C.: Cost Effectiveness of Gastric Cancer Screening According to Race and Ethnicity. Gastroenterology. 2018; 155:648–660.
Jiang Y., Zhang Q., Hu Y., Li T., Yu J., Zhao L., et al.: ImmunoScore Signature: a Prognostic and Predictive Tool in Gastric Cancer. Ann Surg. 2018; 267:504–513.
Lee J.S., Won H.S., Sun S., Hong J.H., Ko Y.H.: Prognostic role of tumor-infiltrating lymphocytes in gastric cancer: A systematic review and meta-analysis. Medicine (Baltimore). 2018; 97:e11769.
Amin M.B., American Joint Committee on Cancer, American Cancer Society. AJCC cancer staging manual. Eight edition/editor-in-chief, Mahul B. Amin, MD, FCAP; editors, Stephen B. Edge, MD, FACS and 16 others; Donna M. Gress, RHIT, CTR-Technical editor; Laura R. Meyer, CAPM-Managing editor. ed. Chicago IL: American Joint Committee on Cancer, Springer; 2017.
Zeng D., Zhou R., Yu Y., Luo Y., Zhang J., Sun H., et al.: Gene expression profiles for a prognostic immunoscore in gastric cancer. British Journal of Surgery. 2018; 105:1338–1348.
Pages F., Mlecnik B., Marliot F., Bindea G., Ou F.S., Bifulco C., et al.: International validation of the consensus Immunoscore for the classification of colon cancer: a prognostic and accuracy study. Lancet. 2018; 391:2128–2139.
Schuler T, Blankenstein T.: Cutting edge: CD8+ effector T cells reject tumors by direct antigen recognition but indirect action on host cells. J Immunol. 2003; 170:4427–4431
Kang B.W., Kim J.G., Lee I.H., Bae H.I., Seo A.N.: Clinical significance of tumor-infiltrating lymphocytes for gastric cancer in the era of immunology. World J Gastrointest Oncol. 2017; 9:293–299.
Zhou S., Shen Z., Wang Y., Ma H., Xu S., Qin J., et al.: CCR7 expression and intratumoral FOXP3+ regulatory T cells are correlated with overall survival and lymph node metastasis in gastric cancer. PLoS One. 2013; 8:e74430.
Wakatsuki K., Sho M., Yamato I., Takayama T., Matsumoto S., Tanaka T., et al.: Clinical impact of tumor-infiltrating CD45RO(+) memory T cells on human gastric cancer. Oncol Rep. 2013; 29:1756–1762.
Lee H.E., Chae S.W., Lee Y.J., Kim M.A., Lee H.S., Lee B.L., et al.: Prognostic implications of type and density of tumour-infiltrating lymphocytes in gastric cancer. Br J Cancer. 2008; 99:1704–1711.
Perrone G., Ruffini P.A., Catalano V., Spino C., Santini D., Muretto P., et al.: Intratumoural FOXP3-positive regulatory T cells are associated with adverse prognosis in radically resected gastric cancer. Eur J Cancer. 2008; 44:1875–1882.
Zheng X., Song X., Shao Y., Xu B., Chen L., Zhou Q., et al.: Prognostic role of tumor-infiltrating lymphocytes in gastric cancer: a meta-analysis. Oncotarget. 2017; 8:57386–57398.
Yuan L., Xu B., Yuan P., Zhou J., Qin P., Han L., et al.: Tumor-infiltrating CD4+ T cells in patients with gastric cancer. Cancer Cell International. 2017; 17:114.
Liu K., Yang K., Wu B., Chen H., Chen X., Chen X., et al.: Tumor-Infiltrating Immune Cells Are Associated With Prognosis of Gastric Cancer. Medicine (Baltimore). 2015; 94:e1631.
Breart B, Lemaitre F, Celli S, Bousso P.: Two-photon imaging of intratumoral CD8+ T cell cytotoxic activity during adoptive T cell therapy in mice. J Clin Invest. 2008; 118:1390–1397.
Ostroumov D., Fekete-Drimusz N., Saborowski M., Kühnel F., Woller N.: CD4 and CD8 T lymphocyte interplay in controlling tumor growth. Cell Mol Life Sci. 2018; 75:689–713.
Szylberg L., Karbownik D., Marszalek A.: The Role of FOXP3 in Human Cancers. Anticancer Res. 2016; 36:3789–3794.
Ma G.F., Chen S.Y., Sun Z.R., Miao Q., Liu Y.M., Zeng X.Q., et al.: FOXP3 inhibits proliferation and induces apoptosis of gastric cancer cells by activating the apoptotic signaling pathway. Biochem Biophys Res Commun. 2013; 430:804–809.
Hao Q., Zhang C., Gao Y., Wang S., Li J., Xue X., Li W., Zhang W., Zhang Y.: FOXP3 inhibits NF-kB activity and hence COX2 expression in gastric cancer cells. Cell Signal. 2014; 26:564–569.
Choi H.S., Ha S.Y., Kim H.M., Ahn S.M., Kang M.S., Kim K.M., et al.: The prognostic effects of tumor infiltrating regulatory T cells and myeloid derived suppressor cells assessed by multicolor flow cytometry in gastric cancer patients. Oncotarget. 2016; 7:7940–7951.
Beckhove P., Feuerer M., Dolenc M., Schuetz F., Choi C., Sommerfeldt N., et al: Specifically activated memory T cell subsets from cancer patients recognize and reject xenotransplanted autologous tumors. J Clin Invest. 2004; 114:67–76.
Coutzac C., Pernotab S., Chaput N., Zaanan A. Immunotherapy in advanced gastric cancer, is it the future? Crit Rev Oncol Hematol. 2019;133:25–32.
Strong V.E., Song K.Y., Park C.H., Jacks L.M., Gonen M., Shah M., et al.: Comparison of gastric cancer survival following R0 resection in the United States and Korea using an internationally validated nomogram. Ann Surg. 2010;251:640–6.
Wang J., Sun Y., Bertagnolli M.M. Comparison of gastric cancer survival between Caucasian and Asian patients treated in the United States: results from the Surveillance Epidemiology and End Results (SEER) database. Ann Surg Oncol. 2015;22:2965–71.
Strong V.E., Wu A.W., Selby L.V., Gonen M., Hsu M., Song K.Y., et al.: Differences in gastric cancer survival between the U.S. and China. J Surg Oncol. 2015;112:31–7.
Lin S.J., Gagnon-Bartsch J.A., Tan I.B., Earle S., Ruff L., Pettinger K., et al.: Signatures of tumour immunity distinguish Asian and non-Asian gastric adenocarcinomas.Gut. 2015;64:1721–31.
Funding
This study was funded by the Gastrointestinal Research Program, John Wayne Cancer Institute.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work. AU drafted the work, and all other authors revised it critically for content. All authors approve the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
All authors have no conflicts of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This study was presented as a Plenary at the 2019 Society for the Surgery of the Alimentary Tract, San Diego, CA, May 21, 2019.
Electronic Supplementary Material
ESM 1
(XLSX 20.7 kb)
Rights and permissions
About this article
Cite this article
Uppal, A., Dehal, A., Chang, SC. et al. The Immune Microenvironment Impacts Survival in Western Patients with Gastric Adenocarcinoma. J Gastrointest Surg 24, 28–38 (2020). https://doi.org/10.1007/s11605-019-04403-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-019-04403-w