Abstract
Background
Surgical resection is the only potentially curative treatment for patients with colorectal, liver, and pancreatic cancers. Although these procedures are performed with low mortality, rates of complications remain relatively high following hepatopancreatic and colorectal surgery.
Methods
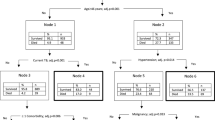
The American College of Surgeons (ACS) National Surgical Quality Improvement Program was utilized to identify patients undergoing liver, pancreatic and colorectal surgery from 2014 to 2016. Decision tree models were utilized to predict the occurrence of any complication, as well as specific complications. To assess the variability of the performance of the classification trees, bootstrapping was performed on 50% of the sample.
Results
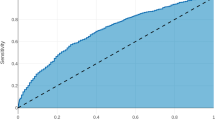
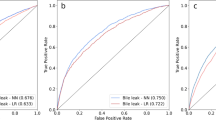
Algorithms were derived from a total of 15,657 patients who met inclusion criteria. The algorithm had a good predictive ability for the occurrence of any complication, with a C-statistic of 0.74, outperforming the ASA (C-statistic 0.58) and ACS-Surgical Risk Calculator (C-statistic 0.71). The algorithm was able to predict with high accuracy thirteen out of the seventeen complications analyzed. The best performance was in the prediction of stroke (C-statistic 0.98), followed by wound dehiscence, cardiac arrest, and progressive renal failure (all C-statistic 0.96). The algorithm had a good predictive ability for superficial SSI (C-statistic 0.76), organ space SSI (C-statistic 0.76), sepsis (C-statistic 0.79), and bleeding requiring transfusion (C-statistic 0.79).
Conclusion
Machine learning was used to develop an algorithm that accurately predicted patient risk of developing complications following liver, pancreatic, or colorectal surgery. The algorithm had very good predictive ability to predict specific complications and demonstrated superiority over other established methods.


Similar content being viewed by others
References
Merath K, Chen Q, Bagante F, Sun S, Akgul O, Idrees JJ, et al. Variation in the cost-of-rescue among medicare patients with complications following hepatopancreatic surgery. HPB (Oxford) [Internet]. 2018 Sep; Available from: https://www.ncbi.nlm.nih.gov/pubmed/30266495
Ghaferi AA, Birkmeyer JD, Dimick JB. Complications, failure to rescue, and mortality with major inpatient surgery in medicare patients. Ann Surg. 2009;250(6):1029–34.
Kohlnhofer BM, Tevis SE, Weber SM, Kennedy GD. Multiple complications and short length of stay are associated with postoperative readmissions. Am J Surg. 2014;207(4):449–56.
Mavros MN, de Jong M, Dogeas E, Hyder O, Pawlik TM. Impact of complications on long-term survival after resection of colorectal liver metastases. Br J Surg. 2013;100(5):711–8.
Healy MA, Mullard AJ, Campbell DA, Dimick JB. Hospital and payer costs associated with surgical complications. JAMA Surg. 2016;151(9):823–30.
Idrees JJ, Johnston FM, Canner JK, Dillhoff M, Schmidt C, Haut ER, et al. Cost of major complications after liver resection in the United States: are high-volume centers cost-effective? Ann Surg [Internet]. 2017; Available from: https://www.ncbi.nlm.nih.gov/pubmed/29232212
Tevis SE, Kennedy GD. Postoperative complications and implications on patient-centered outcomes. J Surg Res. 2013;181(1):106–13.
Silber JH, Rosenbaum PR, Trudeau ME, Chen W, Zhang X, Kelz RR, et al. Changes in prognosis after the first postoperative complication. Med Care. 2005;43(2):122–31.
Spolverato G, Yakoob MY, Kim Y, Alexandrescu S, Marques HP, Lamelas J, et al. Impact of complications on long-term survival after resection of intrahepatic cholangiocarcinoma. Cancer. 2015;121(16):2730–9.
Dorcaratto D, Mazzinari G, Fernandez M, Muñoz E, Garcés-Albir M, Ortega J, et al. Impact of postoperative complications on survival and recurrence after resection of colorectal liver metastases: systematic review and meta-analysis. Ann Surg. 2019;
Merath K, Chen Q, Bagante F, Akgul O, Idrees JJ, Dillhoff M, et al. Synergistic effects of perioperative complications on 30-day mortality following hepatopancreatic surgery. Journal of Gastrointestinal Surgery. 2018;22(10):1715–23.
Ejaz A, Kim Y, Spolverato G, Taylor R, Hundt J, Pawlik TM. Understanding drivers of hospital charge variation for episodes of care among patients undergoing hepatopancreatobiliary surgery. HPB (Oxford). 2015;17(11):955–63.
Abdelsattar ZM, Birkmeyer JD, Wong SL. Variation in Medicare payments for colorectal cancer surgery. J Oncol Pract. 2015;11(5):391–5.
Pradarelli JC, Healy MA, Osborne NH, Ghaferi AA, Dimick JB, Nathan H. Variation in Medicare expenditures for treating perioperative complications: the cost of rescue. JAMA Surg. 2016;151(12):e163340.
Copeland GP, Jones D, Walters M. POSSUM: a scoring system for surgical audit. Br J Surg. 1991;78(3):355–60.
Gawande AA, Kwaan MR, Regenbogen SE, Lipsitz SA, Zinner MJ. An Apgar score for surgery. J Am Coll Surg. 2007;204(2):201–8.
Bilimoria KY, Liu Y, Paruch JL, Zhou L, Kmiecik TE, Ko CY, et al. Development and evaluation of the universal ACS NSQIP surgical risk calculator: a decision aid and informed consent tool for patients and surgeons. J Am Coll Surg. 2013;217(5):833–842.e1-3.
Beal EW, Saunders ND, Kearney JF, Lyon E, Wei L, Squires MH, et al. Accuracy of the ACS NSQIP online risk calculator depends on how you look at it: results from the United States Gastric Cancer Collaborative. Am Surg. 2018;84(3):358–64.
Gleeson EM, Shaikh MF, Shewokis PA, Clarke JR, Meyers WC, Pitt HA, et al. WHipple-ABACUS, a simple, validated risk score for 30-day mortality after pancreaticoduodenectomy developed using the ACS-NSQIP database. Surgery. 11;160(5):1279–87.
Beal EW, Lyon E, Kearney J, Wei L, Ethun CG, Black SM, et al. Evaluating the American College of Surgeons National Surgical Quality Improvement project risk calculator: results from the U.S. Extrahepatic Biliary Malignancy Consortium. HPB (Oxford). 2017;19(12):1104–11.
Bertsimas D, Dunn J, Velmahos GC, Kaafarani HMA. Surgical risk is not linear: derivation and validation of a novel, user-friendly, and machine-learning-based Predictive OpTimal Trees in Emergency Surgery Risk (POTTER) Calculator. Annals of Surgery. 2018;268(4):574–83.
Carlos RC, Kahn CE, Halabi S. Data science: big data, machine learning, and artificial intelligence. J Am Coll Radiol. 2018;15(3 Pt B):497–8.
Linden A, Yarnold PR. Modeling time-to-event (survival) data using classification tree analysis. J Eval Clin Pract. 2017;23(6):1299–308.
Hu Y-J, Ku T-H, Jan R-H, Wang K, Tseng Y-C, Yang S-F. Decision tree-based learning to predict patient controlled analgesia consumption and readjustment. BMC Med Inform Decis Mak. 2012;12:131.
Khuri SF, Daley J, Henderson W, Hur K, Demakis J, Aust JB, et al. The Department of Veterans Affairs’ NSQIP: the first national, validated, outcome-based, risk-adjusted, and peer-controlled program for the measurement and enhancement of the quality of surgical care. National VA Surgical Quality Improvement Program. Ann Surg. 1998;228(4):491–507.
Hall BL, Hamilton BH, Richards K, Bilimoria KY, Cohen ME, Ko CY. Does surgical quality improve in the American College of Surgeons National Surgical Quality Improvement Program: an evaluation of all participating hospitals. Ann Surg. 2009;250(3):363–76.
Fink AS, Campbell DA, Mentzer RM, Henderson WG, Daley J, Bannister J, et al. The National Surgical Quality Improvement Program in non-veterans administration hospitals: initial demonstration of feasibility. Ann Surg. 2002;236(3):344–53; discussion 353–354.
Hastie T, Tibshirani R, Friedman JH. The elements of statistical learning: data mining, inference, and prediction. 2nd ed. New York, NY: Springer; 2009. 745 p. (Springer series in statistics).
Pencina MJ, D’Agostino RB. Evaluating discrimination of risk prediction models: the C statistic. JAMA. 2015;314(10):1063–4.
Merath K, Bagante F, Beal EW, Lopez-Aguiar AG, Poultsides G, Makris E, et al. Nomogram predicting the risk of recurrence after curative-intent resection of primary non-metastatic gastrointestinal neuroendocrine tumors: an analysis of the U.S. Neuroendocrine Tumor Study Group. J Surg Oncol [Internet]. 2018; Available from: https://www.ncbi.nlm.nih.gov/pubmed/29448303
Efron B, Tibshirani R. An introduction to the bootstrap. New York: Chapman & Hall; 1993. 436 p. (Monographs on statistics and applied probability).
Obermeyer Z, Emanuel EJ. Predicting the future - big data, machine learning, and clinical medicine. N Engl J Med. 2016;375(13):1216–9.
Zhao B, Waterman RS, Urman RD, Gabriel RA. A machine learning approach to predicting case duration for robot-assisted surgery. J Med Syst. 2019;43(2):32.
Harvin JA, Green CE, Pedroza C, Tyson JE, Moore LJ, Wade CE, et al. Using machine learning to identify change in surgical decision making in current use of damage control laparotomy. J Am Coll Surg. 2019;228(3):255–64.
Canchi T, Kumar SD, Ng EYK, Narayanan S. A review of computational methods to predict the risk of rupture of abdominal aortic aneurysms. Biomed Res Int. 2015;2015:861627.
Yeong E-K, Hsiao T-C, Chiang HK, Lin C-W. Prediction of burn healing time using artificial neural networks and reflectance spectrometer. Burns. 2005;31(4):415–20.
Chen JH, Asch SM. Machine learning and prediction in medicine - beyond the peak of inflated expectations. N Engl J Med. 2017;376(26):2507–9.
James G, Witten D, Hastie T, Tibshirani R, editors. An introduction to statistical learning: with applications in R. New York: Springer; 2013. 426 p. (Springer texts in statistics).
Parikh SA, Gomez R, Thirugnanasambandam M, Chauhan SS, De Oliveira V, Muluk SC, et al. Decision tree-based classification of abdominal aortic aneurysms using geometry quantification measures. Ann Biomed Eng. 2018;46(12):2135–47.
Wynter-Blyth V, Moorthy K. Prehabilitation: preparing patients for surgery. BMJ. 8;358:j3702.
Minnella EM, Liberman AS, Charlebois P, Stein B, Scheede-Bergdahl C, Awasthi R, et al. The impact of improved functional capacity before surgery on postoperative complications: a study in colorectal cancer. Acta Oncol. 2019;1–6.
Raval MV, Pawlik TM. Practical guide to surgical data sets: national surgical quality improvement program (NSQIP) and pediatric NSQIP. JAMA Surg [Internet]. 2018; Available from: https://www.ncbi.nlm.nih.gov/pubmed/29617521
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Merath, K., Hyer, J.M., Mehta, R. et al. Use of Machine Learning for Prediction of Patient Risk of Postoperative Complications After Liver, Pancreatic, and Colorectal Surgery. J Gastrointest Surg 24, 1843–1851 (2020). https://doi.org/10.1007/s11605-019-04338-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-019-04338-2