Abstract
Objectives
To evaluate gender differences in initial presentation, pathology and outcomes with GC (GC).
Methods
The 1973–2013 Surveillance Epidemiology and End Results (SEER) 17-registry database was analysed for renal tumours from 1973 to 2013 coded as primary site “stomach”. After various exclusions, a final study group of 99,922 cases with complete data was obtained. Demographic variables analysed included age, sex, marital status and race. Tumour variables included size, stage at diagnosis, grade, primary site, treatment and histology. Primary outcome variables included overall survival (OS) and cancer-specific survival (CSS).
Results
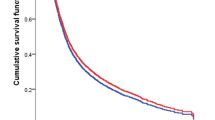
Overall, 96,501 gastric cancer patients were identified. Of those, 34,862 (36.2%) were women. For woman, log-rank test showed that OS and CSS were significantly longer in man (p < 0.0001). In Cox regression analysis, woman was associated with a significantly improved OS [(HR of death in 1973 to 2003 = 0.87, 95% CI = 0.85–0.89, P < 0.001) (HR of death in 2004 to 2013 = 0.94, 95% CI = 0.91–0.97, P < 0.001)] and cancer-specific survival [(HR of death in 1973 to 2003 = 0.90, 95% CI = 0.87–0.92, P < 0.001) (HR of death in 2004 to 2013 = 0.90, 95% CI = 0.87–0.93, P < 0.001)]. When performing a Kaplan-Meier curve analysis after propensity score matching, gender persisted to be a significant survival of woman for OS and CSS.
Conclusions
Men present with larger, higher stage, higher grade GC than women. OS and CSS are better in women, which is significantly different.




Similar content being viewed by others
References
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA: a cancer journal for clinicians. Jan-Feb 2014;64(1):9–29.
Matsuda T, Saika K. The 5-year relative survival rate of stomach cancer in the USA, Europe and Japan. Japanese journal of clinical oncology. Nov 2013;43(11):1157–1158.
Rawicz-Pruszynski K, van Sandick JW, Mielko J, Cisel B, Polkowski WP. Current challenges in gastric cancer surgery: European perspective. Surgical oncology. Dec 2018;27(4):650–656.
Karimi P, Islami F, Anandasabapathy S, Freedman ND, Kamangar F. Gastric cancer: descriptive epidemiology, risk factors, screening, and prevention. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. May 2014;23(5):700–713.
Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, et al. Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. European journal of cancer (Oxford, England : 1990). Apr 2013;49(6):1374–1403.
Leoncini E, Vukovic V, Cadoni G, et al. Tumour stage and gender predict recurrence and second primary malignancies in head and neck cancer: a multicentre study within the INHANCE consortium. European journal of epidemiology. Dec 2018;33(12):1205–1218.
Uhlig A, Seif Amir Hosseini A, Simon J, et al. Gender specific differences in disease-free, cancer specific and overall survival after radical cystectomy for bladder cancer: a systematic review and meta-analysis The Journal of urology. Jul 2018;200(1):48–60.
Fitchett EJA, Seale AC, Vergnano S, et al. Strengthening the Reporting of Observational Studies in Epidemiology for Newborn Infection (STROBE-NI): an extension of the STROBE statement for neonatal infection research. The Lancet. Infectious diseases. Oct 2016;16(10):e202-e213.
Aron M, Nguyen MM, Stein RJ, Gill IS. Impact of gender in renal cell carcinoma: an analysis of the SEER database. European urology. Jul 2008;54(1):133–140.
Mungan NA, Aben KK, Schoenberg MP, et al. Gender differences in stage-adjusted bladder cancer survival. Urology. Jun 2000;55(6):876–880.
Song M, Kang D, Yang JJ, et al. Age and sex interactions in gastric cancer incidence and mortality trends in Korea. Gastric cancer : official journal of the International Gastric Cancer Association and the Japanese Gastric Cancer Association. Jul 2015;18(3):580–589.
Kim HW, Kim JH, Lim BJ, et al. Sex disparity in gastric cancer: female sex is a poor prognostic factor for advanced gastric cancer. Annals of surgical oncology. Dec 2016;23(13):4344–4351.
Yang Y, Wang G, He J, et al. Gender differences in colorectal cancer survival: a meta-analysis. International journal of cancer. Nov 15 2017;141(10):1942–1949.
Gan L, He J, Zhang X, et al. Expression profile and prognostic role of sex hormone receptors in gastric cancer. BMC cancer. Dec 2 2012;12:566.
Fang Z, Zhang T, Dizeyi N, et al. Androgen receptor enhances p27 degradation in prostate cancer cells through rapid and selective TORC2 activation. The Journal of biological chemistry.Jan 13 2012;287(3):2090–2098.
Liu X, Gao Y, Ye H, et al. Positive feedback loop mediated by protein phosphatase 1alpha mobilization of P-TEFb and basal CDK1 drives androgen receptor in prostate cancer. Nucleic acids research. Apr 20 2017;45(7):3738–3751.
Yoshida S, Aihara K, Ikeda Y, et al. Androgen receptor promotes sex-independent angiogenesis in response to ischemia and is required for activation of vascular endothelial growth factor receptor signaling. Circulation. Jul 2 2013;128(1):60–71.
Ozguroglu M, Demir G. Expression of p53 protein and resistance to preoperative chemotherapy in locally advanced gastric carcinoma. Cancer. Aug 1 1999;86(3):547–549.
Koivisto PA, Rantala I. Amplification of the androgen receptor gene is associated with P53 mutation in hormone-refractory recurrent prostate cancer. The Journal of pathology. Jan 1999;187(2):237–241.
Wu W, Karelia D, Pramanik K, et al. Phenylbutyl isoselenocyanate induces reactive oxygen species to inhibit androgen receptor and to initiate p53-mediated apoptosis in LNCaP prostate cancer cells. Molecular carcinogenesis. Aug 2018;57(8):1055–1066.
Feng H, Cheng AS, Tsang DP, et al. Cell cycle-related kinase is a direct androgen receptor-regulated gene that drives beta-catenin/T cell factor-dependent hepatocarcinogenesis. The Journal of clinical investigation. Aug 2011;121(8):3159–3175.
Grumolato L, Liu G, Haremaki T, et al. beta-Catenin-independent activation of TCF1/LEF1 in human hematopoietic tumor cells through interaction with ATF2 transcription factors. PLoS genetics. 2013;9(8):e1003603.
Austin PC. The performance of different propensity-score methods for estimating differences in proportions (risk differences or absolute risk reductions) in observational studies. Statistics in medicine. Sep 10 2010;29(20):2137–2148.
Author information
Authors and Affiliations
Contributions
CZ and HL conceived and designed the study. ZW, CW, WC and YH performed the statistical analysis. HL and ZW wrote the paper. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
All authors declare that they have no conflict of interest.
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Huafu Li and Zhewei Wei are joint first authors.
Electronic Supplementary Material
ESM 1
Univariate Cox Analyses of GC Patients According to Various Clinicopathological Variables. A and B, Analyses of OS in 1973–2003 and 2004–2013. C and D, Analyses of CSS in 1973–2003 and 2004–2013. (ZIP 8.89 mb)
ESM 2
Multivariate Cox Analyses of GC Patients According to Various Clinicopathological Variables. A and B, Analyses of OS in 1973–2003 and 2004–2013. C and D, Analyses of CSS in 1973–2003 and 2004–2013. (ZIP 7.33 mb)
Rights and permissions
About this article
Cite this article
Li, H., Wei, Z., Wang, C. et al. Gender Differences in Gastric Cancer Survival: 99,922 Cases Based on the SEER Database. J Gastrointest Surg 24, 1747–1757 (2020). https://doi.org/10.1007/s11605-019-04304-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-019-04304-y