Abstract
Background
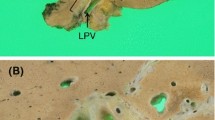
The significance of perineural invasion in extrahepatic cholangiocarcinoma has not been fully elucidated. This study aims to determine the prognostic impact of and optimal treatment strategy for perineural invasion in patients with extrahepatic cholangiocarcinoma.
Methods
Medical records of 133 patients with extrahepatic cholangiocarcinoma who underwent curative resection were reviewed retrospectively. Ninety-eight patients had perineural invasion and 35 patients did not. Univariate and multivariate survival analyses were performed to clarify the prognostic impact of and optimal treatment strategy for perineural invasion.
Results
Only tumor differentiation (P = 0.024) was independently associated with perineural invasion in the multivariate logistic regression model. Multivariate survival analysis revealed that perineural invasion (P = 0.002), resection margin status (P = 0.016), and International Union Against Cancer (UICC) pT factor (P = 0.015) were independent prognostic factors of overall survival. Overall 5-year survival rates for patients with and without perineural invasion were 28 and 74 %, respectively. Among 98 patients with perineural invasion, the use of adjuvant chemotherapy (P = 0.003), lymph node status (P = 0.015), resection margin status (P = 0.008), and UICC pT factor (P = 0.016) were independently associated with overall survival by multivariate analysis. Overall 5-year survival rates for patients with perineural invasion who did and did not receive adjuvant chemotherapy were 33 and 21 %, respectively (P = 0.023).
Conclusions
Perineural invasion is a potent prognostic factor in extrahepatic cholangiocarcinoma. Adjuvant chemotherapy may improve the overall survival of patients with perineural invasion.



Similar content being viewed by others
References
Nakeeb A, Pitt HA, Sohn TA, et al. Cholangiocarcinoma. A spectrum of intrahepatic, perihilar, and distal tumors. Ann Surg 1996; 224:463–473.
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin 2012; 62:10–29.
Bhuiya MR, Nimura Y, Kamiya J, et al. Clinicopathologic studies on perineural invasion of bile duct carcinoma. Ann Surg 1992; 215:344–349.
Kim S, Kim SW, Bang YJ, et al. Role of postoperative radiotherapy in the management of extrahepatic bile duct cancer. Int J Radiat Oncol Biol Phys 2002; 54:414–419.
Wakai T, Shirai Y, Moroda T, et al. Impact of ductal resection margin status on long-term survival in patients undergoing resection for extrahepatic cholangiocarcinoma. Cancer 2005; 103:1210–6.
Tamada S, Shibahara H, Higashi M, et al. MUC4 is a novel prognostic factor of extrahepatic bile duct carcinoma. Clin Cancer Res 2006; 12:4257–4264.
Oh D, do Lim H, Heo JS, et al. The role of adjuvant radiotherapy in microscopic tumor control after extrahepatic bile duct cancer surgery. Am J Clin Oncol 2007; 30:21–25.
Sasaki R, Takeda Y, Funato O, et al. Significance of ductal margin status in patients undergoing surgical resection for extrahepatic cholangiocarcinoma. World J Surg 2007; 31:1788–1796.
Itatsu K, Zen Y, Yamaguchi J, et al. Expression of matrix metalloproteinase 7 is an unfavorable postoperative prognostic factor in cholangiocarcinoma of the perihilar, hilar, and extrahepatic bile ducts. Hum Pathol 2008; 39:710–719.
Li S, Jo YS, Lee JH, et al. L1 cell adhesion molecule is a novel independent poor prognostic factor of extrahepatic cholangiocarcinoma. Clin Cancer Res 2009; 15:7345–7351.
Kitamura K, Hatano E, Higashi T, et al. Prognostic value of 18F-fluorodeoxyglucose positron emission tomography in patients with extrahepatic bile duct cancer. J Hepatobiliary Pancreat Sci 2011; 18:39–46.
Oshiro Y, Sasaki R, Kobayashi A, et al. Prognostic relevance of the lymph node ratio in surgical patients with extrahepatic cholangiocarcinoma. Eur J Surg Oncol 2011; 37:60–64.
Kim TH, Han SS, Park SJ, et al. Role of adjuvant chemoradiotherapy for resected extrahepatic biliary tract cancer. Int J Radiat Oncol Biol Phys 2011; 81:e853-859.
Park JH, Choi EK, Ahn SD, et al. Postoperative chemoradiotherapy for extrahepatic bile duct cancer. Int J Radiat Oncol Biol Phys 2011; 79:696–704.
Zhou L, He XD, Cui QC, Zhou WX et al. Expression of LAPTM4B-35: a novel marker of progression, invasiveness and poor prognosis of extrahepatic cholangiocarcinoma. Cancer Lett 2008; 264:209–217.
Cho JY, Han HS, Yoon YS, et al. Preoperative cholangitis and metastatic lymph node have a negative impact on survival after resection of extrahepatic bile duct cancer. World J Surg 2012; 36:1842–1847.
Oshikiri T, Miyamoto M, Shichinohe T, et al. Prognostic value of intratumoral CD8+ T lymphocyte in extrahepatic bile duct carcinoma as essential immune response. J Surg Oncol 2003; 84:224–228.
Yoshikawa D, Ojima H, Iwasaki M, et al. Clinicopathological and prognostic significance of EGFR, VEGF, and HER2 expression in cholangiocarcinoma. Br J Cancer 2008; 98:418–425.
Hong SM, Cho H, Lee OJ, et al. The number of metastatic lymph nodes in extrahepatic bile duct carcinoma as a prognostic factor. Am J Surg Pathol 2005; 29:1177–1183.
Peng J, Sheng W, Huang D, et al. Perineural invasion in pT3N0 rectal cancer: the incidence and its prognostic effect. Cancer 2011; 117:1415–1421.
Chatterjee D, Katz MH, Rashid A, et al. Perineural and intraneural invasion in posttherapy pancreaticoduodenectomy specimens predicts poor prognosis in patients with pancreatic ductal adenocarcinoma. Am J Surg Pathol 2012; 36:409–417.
Feng FY, Qian Y, Stenmark MH, et al. Perineural invasion predicts increased recurrence, metastasis, and death from prostate cancer following treatment with dose-escalated radiation therapy. Int J Radiat Oncol Biol Phys 2011; 81:e361-367.
Sudo T, Murakami Y, Uemura K, et al. Prognostic impact of perineural invasion following pancreatoduodenectomy with lymphadenectomy for ampullary carcinoma. Dig Dis Sci 2008; 53:2281–2286.
Shirai K, Ebata T, Oda K, et al. Perineural invasion is a prognostic factor in intrahepatic cholangiocarcinoma. World J Surg 2008; 32:2395–2402.
Yamaguchi R, Nagino M, Oda K, et al. Perineural invasion has a negative impact on survival of patients with gallbladder carcinoma. Br J Surg 2002; 89:1130–1136.
Bassi C, Dervenis C, Butturini G, et al. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery 2005; 138:8–13.
Wente MN, Bassi C, Dervenis C, et al. Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 2007; 142:761–768.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications. A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004; 240:205–213.
Japanese Society of Biliary Surgery. General Rules for Surgical and Pathological Studies on Cancer of the Biliary Tract. 5th ed. Tokyo: Kanehara; 2003.
Sobin LH, Gospodarowicz MK, Wittekind C (eds). Internatinal Union Against Cancer (UICC): TNM Classification of Malignant Tumors. 7th ed. New York: Wiley-Blackwell; 2010.
Murakami Y, Uemura K, Sudo T, et al. Adjuvant gemcitabine plus S-1 chemotherapy improves survival after aggressive surgical resection for advanced biliary carcinoma. Ann Surg 2009; 250:950–956.
Murakami Y, Uemura K, Sudo T, et al. Prognostic factors after surgical resection for intrahepatic, hilar, and distal cholangiocarcinoma. Ann Surg Oncol 2011; 18:651–658.
Murakami Y, Uemura K, Sudo T, et al. Adjuvant chemotherapy with gemcitabine and S-1 after surgical resection for advanced biliary carcinoma: outcomes and prognostic factors. J Hepatobiliary Pancreat Sci 2012; 19:306–313.
Kobayashi H, Murakami Y, Uemura K, et al. Human equilibrative nucleoside transporter 1 expression predicts survival of advanced cholangiocarcinoma patients treated with gemcitabine-based adjuvant chemotherapy after surgical resection. Ann Surg 2012; 256:288–296.
Murakami Y, Uemura K, Sudo T, et al. Prognostic factors of patients with advanced gallbladder carcinoma following aggressive surgical resection. J Gastrointest Surg 2011; 15:1007–1016.
Murakami Y, Uemura K, Sudo T, et al. Gemcitabine-based adjuvant chemotherapy improves survival after aggressive surgery for hilar cholangiocarcinoma. J Gastrointest Surg 2009; 13:1470–1479.
Shen FZ, Zhang BY, Feng YJ, et al. Current research in perineural invasion of cholangiocarcinoma. J Exp Clin Cancer Res 2010; 10;29:24.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Murakami, Y., Uemura, K., Sudo, T. et al. Perineural Invasion in Extrahepatic Cholangiocarcinoma: Prognostic Impact and Treatment Strategies. J Gastrointest Surg 17, 1429–1439 (2013). https://doi.org/10.1007/s11605-013-2251-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-013-2251-0