Abstract
Background
Prosthetic mesh infection is one of the most challenging complications after hernia repair. We evaluate the efficacy of soaking mesh in antibiotics to prevent prosthetic infection in an animal model of clean–contaminated ventral hernia repair (VHR).
Material and Methods
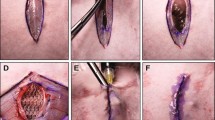
Rats underwent an acute VHR with one of four synthetic meshes (composite multifilament polyester (Parietex PCO), multifilament polyester (Parietex TET), composite monofilament polypropylene (Ventralight), or monofilament polypropylene (SoftMesh)). Prior to implantation, mesh was soaked in saline or 10 mg/ml of vancomycin for 15 min. Following implantation, meshes were contaminated with 104 CFU of methicillin-resistant Staphylococcus aureus (MRSA) bacteria. Thirty days after implantation, mesh samples were cultured and evaluated under scanning electron microscope for biofilm formation.
Results
Presoaking meshes significantly improves bacterial clearance in composite meshes and multifilament polyester mesh. MRSA clearance was as follows for all meshes (saline-soaked vs. vanco-soaked): Parietex PCO (0 vs. 56 %, p = 0.006), Parietex TET (0 vs. 50 %, p = 0.01), Ventralight (20 vs. 78 %, p = 0.012), and SoftMesh (70 vs. 80 %, p = 0.6). MRSA biofilm formation was consistent with bacterial growth.
Conclusion
Presoaking multifilament and composite mesh in vancomycin solution reduces MRSA bacterial growth. Its implementation may reduce the risk of mesh infection in clean–contaminated cases, although further investigation with human trials should be performed.


Similar content being viewed by others

References
den Hartoq D, Dur AH, Tuinebreijer WE, Kreis RW. Open surgical procedures for incisional hernias. Cochrane Database Syst Rev 2008;16(3):CD006438.
Leber GE, Garb JL, Alexander AI, Reed WP. Long-term complications associated with prosthetic repair of incisional hernia. Arch Surg 1998;133:378–382.
Cobb WS, Carbonell AM, Kalbaugh CL, et al. Infection risk of open placement of intraperitoneal composite mesh. Am Surg 2009;75(9):762–767; discussion 767–768.
Machairas A, Misiakos EP, Liakakos T, Karatzas G. Incisional hernioplasty with extraperitoneal onlay polyester mesh. Am Surg 2004;70(8):726–729.
Halaweish I, Harth K, Broome A-M, et al. Novel in vitro model for assessing susceptibility of synthetic hernia repair meshes to Staphylococcus aureus infection using green fluorescent protein-label bacteria and modern imaging techniques. Surg Infect 2010;11(5):449–454.
Engelsman AF, van der Mei HC, Busscher HJ, Ploeg RJ. Morphological aspects of surgical meshes as a risk factor for bacterial colonization. Br J Surg 2008;95:1051–1059.
Yuehuei H, Friedman RJ. Concise review of mechanisms of bacterial adhesion to biomaterial surfaces. J Biomed Mater Res 1998;43:338–348.
Harth KC, Rosen MJ, Thatiparti TR, et al. Antibiotic-releasing mesh coating to reduce prosthetic sepsis: an in vivo study. J Surg Res 2010;163(2):337–343.
Carbonell AM, Matthews BD, Dréau D, et al. The susceptibility of prosthetic biomaterial to infection. Surg Endosc 2005;19:430–435.
Cobb WS, Paton BL, Novitsky YW, et al. Intra-abdominal placement of antimicrobial-impregnated mesh is associated with noninfectious fever. Am Surg 2006; 72(12):1205–1208. Discussion: 1208–1209.
Engelsman AF, van der Mei HC, Ploeg RJ, Busscher HJ. The phenomenon of infection with abdominal wall reconstruction. Biomaterials 2007;28:2314–2327.
Hetrick EM, Schoenfisch MH. Reducing implant-related infection: active release strategies. Chem Soc Rev 2006;35(9):780–789.
Aydinuraz K, Agalar C, Agalar F, et al. In vitro S. epidermidis and S. aureus adherence to composite and lightweight polypropylene grafts. J Surg Res 2009;157:79–86
Klinge U, Junge K, Spellerberg B, et al. Do multifilament alloplastic meshes increase the infection rate? Analysis of the polymeric surface, the bacteria adherence, and the in vivo consequences in a rat model. J Biomed Mater Res 2002;63(6):765–771.
Sanchez-Manuel FJ, Lozano-García J, Seco-Gil JL. Antibiotic prophylaxis for hernia repair. Cochrane Database Syst Rev 2012;2:CD003769.
Lazorthes F, Chiotasso P, Massip P, et al. Local antibiotics prophylaxis in inguinal hernia repair. Surg Gynecol Obstet 1992;1755:69–570.
Troy MG, Dong QS, Dobrin PB, Hecht D. Do topical antibiotics provide prophylasis against bacterial growth in the presence of polypropylene mesh? Am J Surg 1996:171(4):391–393.
Goëau-Brissonnière O, Leflon V, Letort M, Nicolas MH. Resistance of antibiotics-bonded gelatin-coated polymer mesh to Staphylococcus aureus in a rabbit subcutaneous pouch model. Biomaterials 1999;20:229–232.
Díaz-Godoy A, García-Ureña MA, López-Monclus J, et al. Searching for the polypropylene mesh to be used in bowel contamination. Hernia 2011;15:173–179.
Poelstra KA, Barekzi NA, Rediske AM, et al. Prophylactic treatment of gram-positive and gram-negative abdominal implant infections using locally delivered polyclonal antibodies. Biomed Mater Res 2002;60(1):206–215
Raad I, Mohamed JA, Reitzel RA, et al. Improved antibiotic-impregnated catheters with extended-spectrum activity against resistant bacteria and fungi. Antimicrob Agents Chemother 2012;56(2):935–941.
Jang CH, Park H, Cho YB. Effect of vancomycin-coated tympanostomy tubes on Methicillin-resistant Staphylococcus aureus biofilm formation: in vitro study. J Laryngol Otol 2010;214(6):594:598.
Acknowledgments
This work was funded through an internal department of surgery grant at Case Western Reserve University.
Disclosures
Michael Rosen and Yuri Novitsky are speakers for Davol Inc. and Lifecell.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sadava, E.E., Krpata, D.M., Gao, Y. et al. Does Presoaking Synthetic Mesh in Antibiotic Solution Reduce Mesh Infections? An Experimental Study. J Gastrointest Surg 17, 562–568 (2013). https://doi.org/10.1007/s11605-012-2099-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-012-2099-8