Abstract
Introduction
The aim of this study was to investigate the role of Mad1/c-Myc in telomerase regulation in gastric cancer cells in order to gain insight into telomerase activity and to evaluate PinX1 as a putative inhibitor of gastric cancer.
Methods
PinX1 and PinX1siRNA eukaryotic expression vectors were constructed by recombinant technology and transfected into gastric carcinoma cells using Lipofectamine™ 2000. Telomerase activity was measured by the telomeric repeat amplification protocol. Apoptosis of gastric cancer cells was analyzed by flow cytometry and transmission electron microscopy. Reverse transcription-polymerase chain reaction and Western blotting were used to assess the expression levels of PinX1 and Mad1/c-Myc.
Results
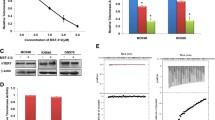
We found that PinX1-negative gastric cancer cells showed significantly higher telomerase activity than did the PinX1-postive cells. PinX1-transfection reduced telomerase activity in PinX1-negative gastric cancer cells and exhibited an upregulation of Mad1 and downregulation of c-Myc expression. Pinx1 RNAi treatment led to downregulation of Mad1 and upregulation of c-Myc.
Conclusion
Suppression of telomerase activity mediated by PinX1 is involved in the Mad1/c-Myc pathway.




Similar content being viewed by others
References
Feng J, Funk W.D, Wang S.S, Weinrich S.L, Avilion A.A, Chiu C.P, Adams R.R, Chang E, Allsopp R.C, Yu J, Le S, West M.D; Harley C.B, Andrews W.H, Greider C.W, Villeponteau B. The RNA component of human telomerase. Science. 1995; 269: 1236–1241.
Nakamura T.M, Morin G.B, Chapman K.B, Weinrich S.L, Andrews W.H, Lingner J, Harley C.B, Cech T.R. Telomerase catalytic subunit homologs from fission yeast and human. Science. 1997; 277: 955–959.
Harrington L, McPhail T, Mar V, Zhou W, Oulton R, Bass M.B, Arruda I, Robinson M.O. A mammalian telomerase-associated protein. Science. 1997; 275: 973–977.
Counter C.M, Meyerson M, Eaton E.N, Ellisen L.W, Caddle S.D, Haber D.A, Weinberg R.A. Telomerase activity is restored in human cells by ectopic expression of hTERT (hEST2), the catalytic subunit of telomerase. Oncogene. 1998; 16: 1217–1222.
Meyerson M, Counter C.M, Eaton E.N, Ellisen L.W, Steiner P, Caddle S.D, Ziaugra L, Beijersbergen R.L, Davidoff M.J, Liu Q, Bacchetti S, Haber D.A, Weinberg R.A. hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell. 1997; 90: 785–795.
Poole J.C, Andrews L.G, Tollefsbol T.O. Activity, function, and gene regulation of the catalytic subunit of telomerase (hTERT).Gene. 2001; 269: 1–12.
Bièche I, Noguès C, Paradis V, Olivi M, Bedossa P, Lidereau R, Vidaud M. Quantitation of hTERT gene expression in sporadic breast tumors with a real-time reverse transcription-polymerase chain reaction assay. Clin Cancer Res. 2000; 6: 452–459.
Shay J.W, Bacchetti S. A survey of telomerase activity in human cancer. Eur J Cancer. 1997; 33: 787–791.
Kim N.W, Piatyszek M.A, Prowse K.R, Harley C.B, West M.D, Ho P.L, Coviello G.M, Wright W.E, Weinrich S.L, Shay J.W. Specific association of human telomerase activity with immortal cells and cancer. Science. 1994; 266: 2011–2015.
Quante M, Heeg S, Werder A, Goessel G, Fulda C, Doebele M, Nakagawa H, Beijersbergen R, Blum H.E, Opitz O.G. Differential transcriptional regulation of human telomerase in a cellular model representing important genetic alterations in esophageal squamous carcinogenesis. Carcinogenesis. 2005; 26: 1879–1889.
Yang S.M, Fang D.C, Luo Y.H, Lu R, Battle P.D, Liu W.W. Alterations of telomerase activity and terminal restriction fragment in gastric cancer and its premalignant lesions. to gain an insight into the biology of telomerase. J Gastroenterol Hepatol. 2001; 16: 876–882.
Fang D.C, Young J, Luo Y.H, Lu R, Jass J. Detection of telomerase activity in biopsy samples of colorectal cancer. J Gastroenterol Hepatol. 1999; 14: 328–332.
Yang S.M, Fang D.C, Yang J.L, Chen L, Luo Y.H, Liang G.P. Antisense human telomerase reverse transcriptase could partially reverse malignant phenotypes of gastric carcinoma cell line in vitro. Eur J Cancer Prev. 2008; 17: 209–217.
Beitzinger M, Oswald C, Beinoraviciute-Kellner R, Stiewe T. Regulation of telomerase activity by the p53 family member p73. Oncogene. 2006; 25: 813–826.
Ducrest A.L, Szutorisz H, Lingner J, Nabholz M. Regulation of the human telomerase reverse transcriptase gene. Oncogene. 2002; 21: 541–552.
Kanaya T, Kyo S, Hamada K, Takakura M, Kitagawa Y, Harada H, Inoue M. Adenoviral expression of p53 represses telomerase activity through down-regulation of human telomerase reverse transcriptase transcription. Clin Cancer Res. 2000; 6: 1239–1247.
Günes C, Lichtsteiner S, Vasserot A.P, Englert C. Expression of the hTERT gene is regulated at the level of transcriptional initiation and repressed by Mad1. Cancer Res. 2000; 60: 2116–2121.
Zhou X.Z, Lu K.P. The Pin2/TRF1-interacting protein PinX1 is a potent telomerase inhibitor. Cell. 2001; 107: 347–359.
Zhang B, Bai Y.X, Ma H.H, Feng F, Jin R, Wang Z.L, Lin J, Sun S.P, Yang P, Wang X.X, Huang P.T, Huang C.F, Peng Y, Chen Y.C, Kung H.F, Huang J.J. Silencing PinX1 compromises telomere length maintenance as well as tumorigenicity in telomerase-positive human cancer cells. Cancer Res. 2009; 69: 75–83.
Kondo T, Oue N, Mitani Y, Kuniyasu H, Noguchi T, Kuraoka K, Nakayama H, Yasui W. Loss of heterozygosity and histone hypoacetylation of the PinX1 gene are associated with reduced expression in gastric carcinoma. Oncogene. 2005; 24: 157–164.
Ma Y, Wu L, Liu C, Xu L, Li D, Li J.C. The correlation of genetic instability of PINX1 gene to clinico-pathological features of gastric cancer in the Chinese population. J Cancer Res Clin Oncol. 2009; 135: 431–437.
Wu K.J, Grandori C, Amacker M, Simon-Vermot N, Polack A, Lingner J, Dalla-Favera R. Direct activation of TERT transcription by c-MYC. Nat Genet. 1999; 21: 220–224.
Wang J, Xie L.Y, Allan S, Beach D, Hannon G.J. Myc activates telomerase. Genes Dev. 1998; 12: 1769–1774.
Hu X, Yu N, Fu L, Su W, Huang G, Wu Y, Huang H, Huang H. Downregulation of human telomerase reverse transcriptase through anti-C-myc siRNA in human colon cancer Colo 320 cells. Nucleotides Nucleic Acids. 2009; 28: 1–11.
Lin S.Y, Elledge S.J. Multiple tumor suppressor pathways negatively regulate telomerase. Cell. 2003; 113: 881–889.
Xu D, Popov N, Hou M, Wang Q, Björkholm M, Gruber A, Menkel AR, Henriksson M. Switch from Myc/Max to Mad1/Max binding and decrease in histone acetylation at the telomerase reverse transcriptase promoter during differentiation of HL60 cells. Proc Natl Acad Sci U S A. 2001; 98: 3826–3831.
Hahn W.C, Weinberg R.A. Rules for making human tumor cells. N Engl J Med. 2002; 347: 1593–1603.
Hanahan D, Weinberg R.A. The hallmarks of cancer. Cell. 2000; 100: 57–70.
Banik S.S, Counter C.M.Characterization of Interactions between PinX1 and Human Telomerase Subunits hTERT and hTR. J Biol Chem. 2004; 279: 51745–51748.
Horikawa I, Barrett J.C. Transcriptional regulation of the telomerase hTERT gene as a target for cellular and viral oncogenic mechanisms. Carcinogenesis. 2003; 24: 1167–1176.
Kyo S, Takakura M, Taira T, Kanaya T, Itoh H, Yutsudo M, Ariga H, Inoue M. Sp1 cooperates with c-Myc to activate transcription of the human telomerase reverse transcriptase gene (hTERT). Nucleic Acids Res. 2000; 28: 669–677.
Greenberg R.A, O'Hagan R.C, Deng H, Xiao Q, Hann S.R, Adams R.R, Lichtsteiner S, Chin L, Morin G.B, DePinho R.A. Telomerase reverse transcriptase gene is a direct target of c-Myc but is not functionally equivalent in cellular transformation Oncogene. 1999; 18: 1219–1226.
Takakura M, Kyo S, Kanaya T, Hirano H, Takeda J, Yutsudo M, Inoue M. Cloning of human telomerase catalytic subunit (hTERT) gene promoter and identification of proximal core promoter sequences essential for transcriptional activation in immortalized and cancer cells. Cancer Res. 1999; 59: 551–557.
Oh S, Song Y.H, Kim U.J, Yim J, Kim T.K. In vivo and in vitro analyses of Myc for differential promoter activities of the human telomerase (hTERT) gene in normal and tumor cells. Biochem Biophys Res Commun. 1999; 263: 361–365.
Flores I, Evan G, Blasco M.A. Genetic Analysis of Myc and Telomerase Interactions In Vivo.Molecular and Cellular Biology. 2006; 26: 6130–6138.
Nam C.W, Park N.H, Park B.R, Shin J.W, Jung S.W, Na Y.W, Seo J.H. Mitotic checkpoint gene MAD1 in hepatocellular carcinoma is associated with tumor recurrence after surgical resection. J Surg Oncol. 2008; 97: 567–571.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Hb., Wang, Xw., Zhou, G. et al. PinX1 Inhibits Telomerase Activity in Gastric Cancer Cells Through Mad1/c-Myc Pathway. J Gastrointest Surg 14, 1227–1234 (2010). https://doi.org/10.1007/s11605-010-1253-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-010-1253-4