Abstract
Background
The present study was designed to specifically investigate the clinicopathological role of expression of cortactin, as well as the correlation with clinical outcomes in stages II–III colorectal cancer (CRC).
Methods
Two hundred and five stages II–III CRC patients were included in this study. Formalin-fixed paraffin-embedded specimens were stained for cortactin and the correlation between the staining, its clinicopathological parameters, and its prognostic power were analyzed statistically.
Results
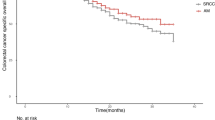
Of the 205 patients studied, 113 cases (55.1%) were strongly positive for cortactin. Cortactin expression correlated with tumor invasion (P = 0.018), histological grade (P = 0.004), and preoperative CEA level (P < 0.001). In univariate analysis, tumor invasion, American Joint Committee on Cancer (AJCC) stage, lymphovascular invasion, preoperative CEA level, and cortactin expression were significant prognostic factors for disease-free survival (P = 0.034, 0.009, 0.043, 0.004, and 0.004, respectively), while for overall survival, tumor invasion, AJCC stage, pathologic grade, preoperative CEA level, and cortactin expression were significant prognostic factors (P = 0.003, 0.008, 0.038, 0.017, and <0.001, respectively). In multivariate analysis, tumor invasion, preoperative CEA level, and cortactin expression maintained their independent prognostic influence on disease-free survival (P = <0.001, 0.003, and 0.008, respectively). However, tumor invasion, AJCC stage, and cortactin expression influenced overall survival (P = 0.036, <0.001, and 0.004, respectively).
Conclusions
Cortactin may be a good biomarker to be applied in the clinical setting to predict the prognosis of patients with completely resected pathologic stages II–III CRC.




Similar content being viewed by others
References
Ottaiano A, Franco R, Aiello Talamanca A, et al. Overexpression of both CXC chemokine receptor 4 and vascular endothelial growth factor proteins predicts early distant relapse in stage II-III colorectal cancer patients. Clin Cancer Res. 2006;12:2795–803.
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71–96.
Obrand DI, Gordon PH. Incidence and patterns of recurrence following curative resection for colorectal carcinoma. Dis Colon Rectum. 1997;40:15–24.
Cerottini JP, Caplin S, Pampallona S, et al. Prognostic factors in colorectal cancer. Oncol Rep. 1999;6:409–14.
Zafirellis K, Agrogiannis G, Zachaki A, et al. Prognostic significance of VEGF expression evaluated by quantitative immunohistochemical analysis in colorectal cancer. J Surg Res. 2008;147:99–107.
van Rossum AG, Moolenaar WH, Schuuring E. Cortactin affects cell migration by regulating intercellular adhesion and cell spreading. Exp Cell Res. 2006;312:1658–70.
Weaver AM, Karginov AV, Kinley AW, et al. Cortactin promotes and stabilizes Arp2/3-induced actin filament network formation. Curr Biol. 2001;11:370–4.
Weaver AM. Cortactin in tumor invasiveness. Cancer Lett. 2008;265:157–66.
Ormandy CJ, Musgrove EA, Hui R, et al. Cyclin D1, EMS1 and 11q13 amplification in breast cancer. Breast Cancer Res Treat. 2003;78:323–35.
Schuuring E. The involvement of the chromosome 11 q13 region in human malignancies: cyclin D1 and EMS1 are two new candidate oncogenes—a review. Gene. 1995;159:83–96.
Tsai WC, Jin JS, Chang WK, et al. Association of cortactin and fascin-1 expression in gastric adenocarcinoma: correlation with clinicopathological parameters. J Histochem Cytochem. 2007;55:955–62.
Yuan BZ, Zhou X, Zimonjic DB, et al. Amplification and overexpression of the EMS 1 oncogene, a possible prognostic marker, in human hepatocellular carcinoma. J Mol Diagn. 2003;5:48–53.
Greer RO Jr, Said S, Shroyer KR, et al. Overexpression of cyclin D1 and cortactin is primarily independent of gene amplification in salivary gland adenoid cystic carcinoma. Oral Oncol. 2007;43:735–41.
Lee YY, Yu CP, Lin CK, et al. Expression of survivin and cortactin in colorectal adenocarcinoma: association with clinicopathological parameters. Dis Markers. 2009;26:9–18.
Schuuring E, Verhoeven E, van Tinteren H, et al. Amplification of genes within the chromosome 11q13 region is indicative of poor prognosis in patients with operable breast cancer. Cancer Res. 1992;52:5229–34.
McCarty KS Jr, Szabo E, Flowers JL, et al. Use of a monoclonal anti-estrogen receptor antibody in the immunohistochemical evaluation of human tumors. Cancer Res. 1986;46:4244s–8s.
Cox DR. Regression models and life-tables. J R Stat Soc Ser B. 1972;34:187–220.
Levy M, Visokai V, Lipska L, et al. Tumor markers in staging and prognosis of colorectal carcinoma. Neoplasma. 2008;55:138–42.
Zhang LH, Tian B, Diao LR, et al. Dominant expression of 85-kDa form of cortactin in colorectal cancer. J Cancer Res Clin Oncol. 2006;132:113–20.
Rodrigo JP, García-Carracedo D, García LA, et al. Distinctive clinicopathological associations of amplification of the cortactin gene at 11q13 in head and neck squamous cell carcinomas. J Pathol. 2009;217:516–23.
Hofman P, Butori C, Havet K, et al. Prognostic significance of cortactin levels in head and neck squamous cell carcinoma: comparison with epidermal growth factor receptor status. Br J Cancer. 2008;98:956–64.
Daly RJ. Cortactin signalling and dynamic actin networks. Biochem J. 2004;382:13–25.
Norton L, Massagué J. Is cancer a disease of self-seeding? Nat Med. 2006;12:875–8.
Wood CB, Ratcliffe JG, Burt RW, et al. The clinical significance of the pattern of elevated serum carcinoembryonic antigen (CEA) levels in recurrent colorectal cancer. Br J Surg. 1980;67:46–8.
Wichmann MW, Muller C, Lau-Werner U, et al. The role of carcinoembryonic antigen for the detection of recurrent disease following curative resection of large-bowel cancer. Langenbecks Arch Surg. 2000;385:271–5.
Chau I, Allen MJ, Cunningham D, et al. The value of routine serum carcino-embryonic antigen measurement and computed tomography in the surveillance of patients after adjuvant chemotherapy for colorectal cancer. J Clin Oncol. 2004; 22:1420–9.
McCall JL, Black RB, Toouli J, et al. The value of serum carcinoembryonic antigen in predicting recurrent disease following curative resection of colorectal cancer. Dis Colon Rectum. 1994;37:875–81.
Wanebo HJ, Rao B, Pinsky CM. The use of preoperative carcinoembryonic antigen level as a prognostic indicator to complement pathological staging. N Engl J Med. 1978;299: 448–51.
Wichmann MW, Lau-Werner U, Mu¨ ller C, et al. Carcinoembryonic antigen for the detection of recurrent disease following curative resection of colorectal cancer. Anticancer Res. 2000;20:4953–5.
Goldstein MJ, Mitchell EP. Carcinoembryonic antigen in the staging and follow-up of patients with colorectal cancer. Cancer Invest. 2005;23:338–51.
Lipská L, Visokai V, Levý M, et al. Tumor markers in patients with relapse of colorectal carcinoma. Anticancer Res. 2007;27:1901–5.
Watine J, Miedouge M, Friedberg B. Carcinoembryonic antigen as an independent prognostic factor of recurrence and survival in patients resected for colorectal liver metastases: a systematic review. Dis Colon Rectum. 2001;44:1791–9.
Wiratkapun S, Kraemer M, Eu KW, et al. High preoperative serum carcinoembryonic antigen predicts metastatic recurrence in potentially curative colonic cancer: results of a five-year study. Dis Colon Rectum. 2004;44:231–5.
Takagawa R, Fujii S, Ohta M, et al. Preoperative serum carcinoembryonic antigen level as a predictive factor of recurrence after curative resection of colorectal cancer. Ann Surg Oncol. 2008;15:3433–9.
Newland RC, Chapuis PH, Pheils MT, et al. The relationship of survival to staging and grading of colorectal carcinoma: a prospective study of 503 cases. Cancer. 1981;47:1424–9.
Compton C, Fenoglio-Preiser CM, Pettigrew N, et al. American joint committee on cancer prognostic factors consensus conference: colorectal working group. Cancer. 2000;88:1739–57.
Chuma M, Sakamoto M, Yasuda J, et al. Overexpression of cortactin is involved in motility and metastasis of hepatocellular carcinoma. J Hepatol. 2004;41:629–36.
Gibcus JH, Mastik MF, Menkema L, et al. Cortactin expression predicts poor survival in laryngeal carcinoma. Br J Cancer. 2008;98:950–5.
Li X, Zheng H, Hara T, et al. Aberrant expression of cortactin and fascin are effective markers for pathogenesis, invasion, metastasis and prognosis of gastric carcinomas. Int J Oncol. 2008;33:69–79.
Lin CK, Su HY, Tsai WC, et al. Association of cortactin, fascin-1 and epidermal growth factor receptor (EGFR) expression in ovarian carcinomas: correlation with clinicopathological parameters. Dis Markers. 2008;25:17–26.
Hsu NY, Yeh KT, Chiang IP, et al. Cortactin overexpression in the esophageal squamous cell carcinoma and its involvement in the carcinogenesis. Dis Esophagus. 2008;21:402–8.
Rodrigo JP, García-Carracedo D, García LA, et al. Distinctive clinicopathological associations of amplification of the cortactin gene at 11q13 in head and neck squamous cell carcinomas. J Pathol. 2009;217:516–23.
Hsu KF, Lin CK, Yu CP, et al. Cortactin, fascin, and survivin expression associated with clinicopathological parameters in esophageal squamous cell carcinoma. Dis Esophagus. 2009;22:402–8.
Lin CK, Chao TK, Yu CP, et al. The expression of six biomarkers in the four most common ovarian cancers: correlation with clinicopathological parameters. APMIS. 2009;117:162–75.
Hirakawa H, Shibata K, Nakayama T. Localization of cortactin is associated with colorectal cancer development. Int J Oncol. 2009;35:1271–6.
Funding
The research was supported in part by the National Natural Science Foundation of China (No.30772106).
Conflict of interest statement
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have no commercial, proprietary, or financial interest in the products or companies described in this article.
Rights and permissions
About this article
Cite this article
Cai, Jh., Zhao, R., Zhu, Jw. et al. Expression of Cortactin Correlates with a Poor Prognosis in Patients with Stages II–III Colorectal Adenocarcinoma. J Gastrointest Surg 14, 1248–1257 (2010). https://doi.org/10.1007/s11605-010-1247-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-010-1247-2