Abstract
Purpose
This study aimed to characterize the computed tomography (CT) findings of pseudolithiasis and investigate the outcomes and natural history in adult patients receiving CTRX therapy.
Methods
A total of 17 patients were diagnosed with CTRX-associated biliary pseudolithiasis on CT between April 2013 and March 2017. The medical records, characteristics, complications, treatment options, and outcomes of these patients were examined. Serial CT images and the form, density, and location of pseudolithiasis were reviewed by two radiologists.
Results
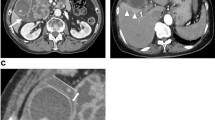
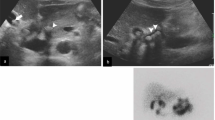
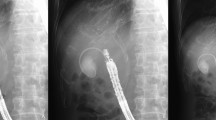
Of the 17 patients with CTRX-associated pseudolithiasis, seven were men and ten were women. The median patient age was 78 years (range 31–88 years). The median interval from CTRX administration to the diagnosis of pseudolithiasis was 10 days (range 4–32 days). The CT findings of pseudolithiasis included a sludge pattern (11 patients [64.7%]), stone pattern (two patients [11.8%]), and stone plus sludge pattern (four patients [23.5%]). Seven patients (41.2%) showed gall bladder enlargement along with a common bile duct (CBD) stone. Two patients with CBD stones underwent endoscopic CBD stone removal. The median time to pseudolithiasis resolution after CTRX cessation was 69 days.
Conclusion
The high-density sludge pattern is the most common typical CT finding of CTRX-associated pseudolithiasis in adults.



Similar content being viewed by others
References
Schaad U, Wedgwood-Krucko J, Tschaeppeler H. Reversible ceftriaxone-associated biliary pseudolithiasis in children. Lancet. 1988;332:1411–3.
Cometta A, Gallot-Lavallée-Villars S, Iten A, et al. Incidence of gallbladder lithiasis after ceftriaxone treatment. J Antimicrob Chemother. 1990;25:689–95.
Heim-Duthoy KL, Caperton EM, Pollock R, et al. Apparent biliary pseudolithiasis during ceftriaxone therapy. Antimicrob Agents Chemother. 1990;34:1146–9.
Kassner EG. Drug-related complications in infants and children: imaging features. AJR Am J Roentgenol. 1991;157:1039–49.
Lopez AJ, O'Keefe P, Morrissey M, et al. Ceftriaxone-induced cholelithiasis. Ann Intern Med. 1991;115:712–4.
Kirejczyk W, Crowe H, Mackay I, et al. Disappearing “gallstones”: biliary pseudolithiasis complicating ceftriaxone therapy. AJR Am J Roentgenol. 1992;159:329–30.
Ko CW, Sekijima JH, Lee SP. Biliary sludge. Ann Intern Med. 1999;130:301–11.
Alehossein M, Sotoudeh K, Nasoohi S, et al. Ceftriaxone induced biliary pseudolithiasis in children: report of 14 cases. Iran J Pediatr. 2008;18:31–7.
Palanduz A, Yalçın I, Tonguç E, et al. Sonographic assessment of ceftriaxone associated biliary pseudolithiasis in children. J Clin Ultrasound. 2000;28:166–8.
Riccabona M, Kerbl R, Schwinger W, et al. Ceftriaxone-induced cholelithiasis—a harmless side-effect? Klin Padiatr. 1993;205:421–3.
Shuichi A. A case of ceftriaxone-associated biliary pseudolithiasis in an elderly patient with renal dysfunction. IDCases [Internet]. 2017;9:62–4.
Nakajima H, Ota S, Hirose T, et al. Ceftriaxone-associated cholelithiasis in adult patients with bacterial meningitis. J Infect Dis Ther [Internet]. 2014;2(5):161.
Besim H, Arslan K. Ceftriaxone associated acute cholecystitis. Arch Balk Med Union. 2016;51:258–60.
Khurram D, Shamban L, Kornas R, et al. Marked Direct hyperbilirubinemia due to ceftriaxone in an adult with sickle cell disease. Case Rep Gastrointest Med [Internet]. 2015;2015:462165.
Prince JS, Senac MO. Ceftriaxone-associated nephrolithiasis and biliary pseudolithiasis in a child. Pediatr Radiol. 2003;33:648–51.
Barakos JA, Ralls P, Lapin SA, et al. Cholelithiasis: evaluation with CT. Radiology. 1987;162:415–8.
Shiffman ML, Keith FB, Moore EW. Pathogenesis of ceftriaxone-associated biliary sludge. Gastroenterology. 1990;99:1772–8.
Brogard J, Blickle J, Jehl F, et al. High biliary elimination of ceftriaxone in man. Int J Clin Pharmacol Ther Toxicol. 1988;26:167–72.
Schaad UB, Suter S, Gianella-Borradori A, et al. A comparison of ceftriaxone and cefuroxime for the treatment of bacterial meningitis in children. N Engl J Med. 1990;322:141–7.
Kimata T, Kaneko K, Takahashi M, et al. Increased urinary calcium excretion caused by ceftriaxone: possible association with urolithiasis. Pediatr Nephrol. 2012;27:605–9.
Palanivelu C, Jategaonkar P, Rangarajan M, et al. “Pseudo” cholelithiasis: sequelae of minimally invasive cholecystectomy with maximum surprise—an unusual case. Endoscopy. 2009;41:E186–187.
Papadopoulou F, Efremidis S, Karyda S, et al. Incidence of ceftriaxone associated gallbladder pseudolithiasis. Acta Paediatr. 1999;88:1352–5.
Murata S, Aomatsu T, Yoden A, et al. Fasting and bed rest, even for a relatively short period, are risk factors for ceftriaxone associated pseudolithiasis. Pediatr Int. 2015;57:942–6.
Hung WY, Lanfranco OA. Contemporary review of drug-induced pancreatitis: a different perspective. World J Gastrointest Pathophysiol. 2014;5:405–15.
Cohen D, Appel GB, Scully B, et al. Pharmacokinetics of ceftriaxone in patients with renal failure and in those undergoing hemodialysis. Antimicrob Agents Chemother. 1983;24:529–32.
Robertson FM, Crombleholme TM, Barlow SE, et al. Ceftriaxone choledocholithiasis. Pediatrics. 1996;98:133–5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflict of interest to declare.
Ethical approval
This study was approved by the institutional review board of our institution.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Yoshida, R., Yoshizako, T., Katsube, T. et al. Computed tomography findings of ceftriaxone-associated biliary pseudocholelithiasis in adults. Jpn J Radiol 37, 826–831 (2019). https://doi.org/10.1007/s11604-019-00893-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11604-019-00893-5