Abstract
Purpose
To evaluate CT density of liver changes between before and after amiodarone administration.
Materials and methods
Twenty-five patients underwent non-enhanced CT including the liver before and after amiodarone administration. We set regions of interest (ROIs) at liver S8, spleen, paraspinal muscle, and calculated average CT density in these ROIs, then compared CT density between liver and other organs. Statistical differences between CT density of liver and various ratios before and after administration were determined, along with correlations between cumulative dose of amiodarone and liver density after administration, density change of liver, and various ratios after administration.
Results
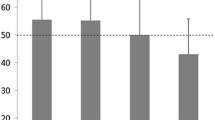
Liver density, liver-to-spleen ratio, and liver-to-paraspinal muscle ratio differed significantly between before and after amiodarone administration. No significant correlations were found between cumulative doses of amiodarone and any of liver density after administration, density change of liver, or various ratios after administration.
Conclusion
CT density of liver after amiodarone administration was significantly higher than that before administration. No correlations were identified between cumulative dose of amiodarone and either liver density after administration or density change of liver. Amiodarone usage should be checked when radiologists identify high density of the liver on CT.


Similar content being viewed by others
References
Nasir H, Anirban B, Suartcha P. Amiodarone-induced cirrhosis of liver: what predicts mortality? ISRN Cardiol. 2013. https://doi.org/10.1155/2013/617943.
Holt DW, Tucker GT, Jackson PR, Storey GC. Amiodarone pharmacokinetics. Am Heart J. 1983;106:840–7.
Goldschlager N, Epstein AE, Naccarelli G, Olshansky B, Singh B. Practical guidelines for clinicians who treat patients with amiodarone. Practice Guidelines Subcommittee, North American Society of Pacing and Electrophysiology. Arch Intern Med. 2000;160:1741–8.
Rotmensch HH, Belhassen B, Swanson BN, Shoshani D, Spielman SR, Greenspon AJ, et al. Steady-state serum amiodarone concentrations: relationships with antiarrhythmic efficacy and toxicity. Ann Intern Med. 1984;101:462–9.
Vorperian VR, Havighurst TC, Miller S, January CT. Adverse effects of low dose amiodarone: a meta-analysis. J Am Coll Cardiol. 1997;30:791–8.
Kojima S, Kojima S, Ueno H, Takeya M, Ogawa H. Increased density of the liver and amiodarone-associated phospholipidosis. Cardiol Res Pract. 2009. https://doi.org/10.4061/2009/598940.
Palakurthy PR, Iyer V, Meckler RJ. Unusual neurotoxicity associated with amiodarone therapy. Arch Intern Med. 1987;147:881–4.
Goldman IS, Winkler ML, Raper SE, Barker ME, Keung E, Goldberg HI, et al. Increased hepatic density and phospholipidosis due to amiodarone. AJR Am J Roentgenol. 1985;144:541–6.
Shenasa M, Vaisman U, Wojciechowski M, Denker S, Murthy V, Akhtar M. Abnormal abdominal computerized tomography with amiodarone therapy and clinical significance. Am Heart J. 1984;107:929–33.
Patrick D, White FE, Adams PC. Long-term amiodarone therapy: a cause of increased hepatic attenuation on CT. Br J Radiol. 1984;57:573–6.
Markos J, Veronese ME, Nicholson MR, McLean S, Shevland JE. Value of hepatic computerized tomographic scanning during amiodarone therapy. Am J Cardiol. 1985;56:89–92.
Hirakawa K, Abe K, Ayabe Y, Nishimura M. Analysis of increased hepatic density during chronic amiodarone therapy. Nihon Igaku Hoshasen Gakkai Zasshi. 2003;63:221–4.
Singh BN, Cho YW, Kuemmerle HP. Clinical pharmacology of antiarrhythmic drugs: a review and overview, part II. Int J Clin Pharmacol Ther Toxicol. 1981;19:185–99.
Rivinius R, Helmschrott M, Ruhparwar A, Darche FF, Thomas D, Bruckner T, et al. Comparison of posttransplant outcomes in patients with no, acute, or chronic amiodarone use before heart transplantation. Drug Des Dev Ther. 2017;11:1827–37.
Poucell S, Ireton J, Valencia-Mayoral P, Downar E, Larratt L, Patterson J, et al. Amiodarone-associated phospholipidosis and fibrosis of the liver. Light, immunohistochemical, and electron microscopic studies. Gastroenterology. 1984;86:926–36.
Morse RM, Valenzuela GA, Greenwald TP, Eulie PJ, Wesley RC, McCallum RW. Amiodarone-induced liver toxicity. Ann Intern Med. 1988;109:838–40.
Stravitz RT, Sanyal AJ. Drug-induced steatohepatitis. Clin Liver Dis. 2003;7:435–51.
Ishida S, Sugino M, Hosokawa T, Sato T, Furutama D, Fukuda A, et al. Amiodarone-induced liver cirrhosis and parkinsonism: a case report. Clin Neuropathol. 2010;29:84–8.
Oikawa H, Maesawa C, Sato R, Oikawa K, Yamada H, Oriso S, et al. Liver cirrhosis induced by long-term administration of a daily low dose of amiodarone: a case report. World J Gastroenterol. 2005;11:5394–7.
Puli SR, Fraley MA, Puli V, Kuperman AB, Alpert MA. Hepatic cirrhosis caused by low-dose oral amiodarone therapy. Am J Med Sci. 2005;330:257–61.
Rofsky NM, Fleishaker H. CT and MRI of diffuse liver disease. Semin Ultrasound CT MR. 1995;16:16–33.
Murakami T, Baron RL, Peterson MS. Liver necrosis and regeneration after fulminant hepatitis: pathologic correlation with CT and MR findings. Radiology. 1996;198:239–42.
Heye T, Nelson RC, Ho LM, Marin D, Boll DT. Dual-energy CT applications in the abdomen. AJR Am J Roentgenol. 2012;199:S64–70.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical statement
This study was approved by the clinical ethics committee of Akita University.
About this article
Cite this article
Matsuda, M., Otaka, A., Tozawa, T. et al. Analysis of computed tomography density of liver before and after amiodarone administration. Jpn J Radiol 36, 340–344 (2018). https://doi.org/10.1007/s11604-018-0733-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11604-018-0733-4