Abstract
Objective
Resuscitation with whole blood is known to be better than that with saline in attaining the return of spontaneous circulation (ROSC) and improving the short-term survival rate for hemorrhage-induced traumatic cardiac arrest (HiTCA). However, the resuscitation with whole blood alone fails to address the pathophysiological abnormalities, including hyperglycemia, hyperkalemia and coagulopathy, after HiTCA. The present study aimed to determine whether the modified glucose-insulin-potassium (GIK) therapy can ameliorate the above-mentioned pathophysiological abnormalities, enhance the ROSC, improve the function of key organs, and reduce the mortality after HiTCA.
Methods
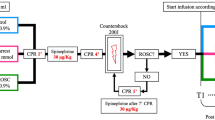
HiTCA was induced in rabbits (n=36) by controlled hemorrhage. Following arrest, the rabbits were randomly divided into three groups (n=12 each): group A (no resuscitation), group B (resuscitation with whole blood), and group C (resuscitation with whole blood plus GIK). The GIK therapy was administered based on the actual concentration of glucose and potassium. The ROSC rate and survival rate were obtained. Hemodynamical and biochemical changes were detected. Thromboelastography (TEG) was used to measure coagulation parameters, and enzyme-linked immunosorbent assay to detect parameters related to inflammation, coagulation and the function of brain.
Results
All animals in groups B and C attained ROSC. Two rabbits died 24–48 h after HiTCA in group B, while no rabbits died in group C. The GIK therapy significantly reduced the levels of blood glucose, potassium, and biological markers for inflammatory reaction, and improved the heart, kidney, liver and brain function in group C when compared to group B. Furthermore, the R values of TEG were significantly lower in group C than in group B, and the maximum amplitude of TEG was slightly lower in group B than in group C, with no significant difference found.
Conclusion
Resuscitation with whole blood and modified GIK therapy combined can ameliorate the pathophysiological disorders, including hyperglycemia, hyperkalemia and coagulopathy, and may improve the function of key organs after HiTCA.
Similar content being viewed by others
References
Xu J, Shen P, Gao Y, et al. The Effects of the Duration of Aortic Balloon Occlusion on Outcomes of Traumatic Cardiac Arrest in a Porcine Model. Shock, 2019,52(3):e12–e21
Barnard EBG, Hunt PAF, Lewis PEH, et al. The outcome of patients in traumatic cardiac arrest presenting to deployed military medical treatment facilities: data from the UK Joint Theatre Trauma Registry. J R Army Med Corps, 2018,164(3):150–154
Rall J, Cox JM, Maddry J. The Use of the AbdominalAortic and Junctional Tourniquet During Cardiopulmonary Resuscitation Following Traumatic Cardiac Arrest in Swine. Mil Med, 2017,182(9):e2001–e2005
Smith JE, Rickard A, Wise D. Traumatic cardiac arrest. J R Soc Med, 2015,108(1):11–16
Watts S, Smith JE, Gwyther R, et al. Closed chest compressions reduce survival in an animal model of haemorrhage-induced traumatic cardiac arrest. Resuscitation, 2019,140:37–42
Jeffcoach DR, Gallegos JJ, Jesty SA, et al. Use of CPR in hemorrhagic shock, a dog model. J Trauma Acute Care Surg, 2016,81(1):27–33
Lockey DJ, Lyon RM, Davies GE. Development of a simple algorithm to guide the effective management of traumatic cardiac arrest. Resuscitation, 2013,84(6):738–742
Hoops HE, Manning JE, Graham TL, et al. Selective Aortic Arch Perfusion with fresh whole blood or HBOC-201 reverses hemorrhage-induced traumatic cardiac arrest in a lethal model of non-compressible torso hemorrhage. J Trauma Acute Care Surg, 2019,87(2):263–273
McCowen KC, Malhotra A, Bistrian BR. Stress-induced hyperglycemia. Crit Care Clin, 2001,17(10):107–124
Kerby JD, Griffin RL, MacLennan P, et al. Stress-induced hyperglycemia, not diabetic hyperglycemia, is associated with higher mortality in trauma. Ann Surg, 2012,256(3):446–452
Bosarge PL, Shoultz TH, Griffin RL, et al. Stress-induced hyperglycemia is associated with higher mortality in severe traumatic brain injury. J Trauma Acute Care Surg, 2015,79(2):289–294
Tsai YW, Wu SC, Huang CY, et al. Impact of stress-induced hyperglycemia on the outcome of children with trauma: A cross-sectional analysis based on propensity score-matched population. Sci Rep, 2019,9(1):16311
Nehme Z, Nair R, Andrew E, et al. Effect of diabetes and pre-hospital blood glucose level on survival and recovery after out-of-hospital cardiac arrest. Crit Care Resusc, 2016,18(2):69–77
Mentzelopoulos SD, Malachias S, Chamos C, et al. Vasopressin, steroids, and epinephrine and neurologically favorable survival after in-hospital cardiac arrest: a randomized clinical trial. JAMA, 2013,310(3):270–279
Russo JJ, James TE, Hibbert B, et al. CAPITAL Investigators. Hyperglycaemia in comatose survivors of out-of-hospital cardiac arrest. Eur Heart J Acute Cardiovasc Care, 2018,7(5):442–449
Kim SH, Choi SP, Park KN, et al. Association of blood glucose at admission with outcomes in patients treated with therapeutic hypothermia after cardiac arrest. Am J Emerg Med, 2014,32(8):900–904
Zhao K, Zhang Y, Li J, et al. Modified Glucose-Insulin-Potassium Regimen Provides Cardioprotection With Improved Tissue Perfusion in Patients Undergoing Cardiopulmonary Bypass Surgery. J Am Heart Assoc, 2020,9(6):e012376
Festing MF. On determining sample size in experiments involving laboratory animals. Lab Anim, 2018,52(4):341–350
Duan K, Yu W, Lin Z, et al. A time course study of acute traumatic coagulopathy prior to resuscitation: from hypercoagulation to hypocoagulation caused by hypoperfusion? Transfus Apher Sci, 2014,50(3):399–406
Jin PY, Zhang HS, Guo XY, et al. Glucose-insulin-potassium therapy in patients with acute coronary syndrome: a meta-analysis of randomized controlled trials. BMC Cardiovasc Disord, 2014,14:169
Molnar M, Bergquist M, Larsson A, et al. Hyperglycaemia increases S100beta after short experimental cardiac arrest. Acta Anaesthesiol Scand, 2014,58(1):106–113
Kreutziger J, Schmid S, Umlauf N, et al. Association between Blood Glucose and cardiac Rhythms during pre-hospital care of Trauma Patients — a retrospective Analysis. Scand J Trauma Resusc Emerg Med, 2018,26(1):58
Diebel LN, Diebel ME, Martin JV, et al. Acute hyperglycemia exacerbates trauma-induced endothelial and glycocalyx injury: An in vitro model. J Trauma Acute Care Surg, 2018, 85(5):960–967
Zhao XD, Qin YH, Ma JX, et al. Influence of intensive insulin therapy on vascular endothelial growth factor in patients with severe trauma. J Huazhong Univ Sci Technolog Med Sci, 2013,33(1):107–110
Kahn NN. Platelet-stimulated thrombin and PDGF are normalized by insulin and Ca2+ channel blockers. Am J Physiol, 1999,276(5):E856–E862
Chen S, Hu M, Shen M, et al. IGF-1 facilitates thrombopoiesis primarily through Akt activation. Blood, 2018,13(2):210–222
Moore EE, Moore HB, Kornblith LZ, et al. Trauma-induced coagulopathy. Nat Rev Dis Primers, 2021,7(1):30
Koami H, Sakamoto Y, Sakurai R, et al. Thromboelastometric analysis of the risk factors for return of spontaneous circulation in adult patients with out-of-hospital cardiac arrest. PLoS One, 2017,12(40):e0175257
Cicero L, Fazzotta S, Palumbo VD, et al. Anesthesia protocols in laboratory animals used for scientific purposes. Acta Biomed, 2018,89(3):337–342
Kiefer D, Müller-Wirtz LM, Maurer F, et al. Intravenous propofol, ketamine (ketofol) and rocuronium after sevoflurane induction provides long lasting anesthesia in ventilated rats. Exp Anim, 2022,71(2):231–239
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no competing interests.
Additional information
This work was supported by the Key Clinical Innovation Project of Army Medical University and Xinqiao Hospital (CX2019JS107/2018JSLC0023).
Rights and permissions
About this article
Cite this article
Zhang, L., Du, Wq., Zong, Zw. et al. Modified Glucose-insulin-potassium Therapy for Hemorrhage-induced Traumatic Cardiac Arrest in Rabbits. CURR MED SCI 43, 1238–1246 (2023). https://doi.org/10.1007/s11596-023-2796-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-023-2796-4