Abstract
Objective
To identify factors affecting the efficacy of steroid-eluting sinus stents implanted after endoscopic sinus surgery (ESS) in patients with chronic rhinosinusitis with nasal polyps (CRSwNP).
Methods
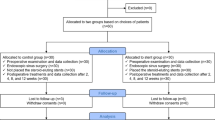
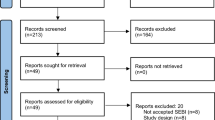
We performed a post-hoc analysis of a randomized self-controlled clinical trial on post-operative implantation of bioabsorbable steroid-eluting stents in patients with CRSwNP. Univariate logistic regression analysis was conducted to identify which of the following factors affect the response to post-operative stent implantation: sex, serum eosinophil levels, history of prior surgery, endoscopic scores, and comorbid conditions (asthma and allergic rhinitis). The primary outcome was the rate of post-operative intervention on day 30, and the secondary outcome was the rate of polypoid tissue formation (grades 2–3) on days 14, 30, and 90.
Results
A total of 151 patients with CRSwNP were included in the post-hoc analysis. Asthma was identified as the only risk factor for a poor response to steroid-eluting sinus stents on post-operative day 30, with an odds ratio of 23.71 (95% CI, 2.81, 200.16; P=0.004) for the need for post-operative intervention and 19 (95% CI, 2.20, 164.16; P=0.003) for moderate-to-severe polypoid tissue formation. In addition, the asthmatic group showed higher rates of post-operative intervention and polypoid tissue formation than the non-asthmatic group on post-operative day 30. Blood eosinophil levels were not identified as a risk factor for poor outcomes after stent implantation.
Conclusion
Comorbid asthma, but not blood eosinophil level, impairs the efficacy of steroid-eluting sinus stents in the short term after ESS in patients with CRSwNP.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fokkens WJ, Lund VJ, Hopkins C, et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhinology, 2020,58(Suppl S29):1–464
Bachert C, Han JK, Desrosiers M, et al. Efficacy and safety of dupilumab in patients with severe chronic rhinosinusitis with nasal polyps (LIBERTY NP SINUS-24 and LIBERTY NP SINUS-52): results from two multicentre, randomised, double-blind, placebo-controlled, parallel-group phase 3 trials. Lancet, 2019,394(10209):1638–1650
Han JK, Bachert C, Fokkens W, et al. Mepolizumab for chronic rhinosinusitis with nasal polyps (SYNAPSE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Respir Med, 2021,9(10):1141–1153
Gevaert P, Omachi TA, Corren J, et al. Efficacy and safety of omalizumab in nasal polyposis: 2 randomized phase 3 trials. J Allergy Clin Immunol, 2020,146(3):595–605
Wu AC, Fuhlbrigge AL, Robayo MA, et al. Cost-Effectiveness of Biologics for Allergic Diseases. J Allergy Clin Immunol Pract, 2021,9(3):1107–1117.e1102
Liu Z, Chen J, Cheng L, et al. Chinese Society of Allergy and Chinese Society of Otorhinolaryngology-Head and Neck Surgery Guideline for Chronic Rhinosinusitis. Allergy Asthma Immunol Res, 2020,12(2):176–237
Wormald PJ, Boustred RN, Le T, et al. A prospective single-blind randomized controlled study of use of hyaluronic acid nasal packs in patients after endoscopic sinus surgery. Am J Rhinol, 2006,20(1):7–10
Kennedy DW, Wright ED, Goldberg AN. Objective and subjective outcomes in surgery for chronic sinusitis. Laryngoscope, 2000,110(3 Pt 3):29–31
Lipworth BJ, Jackson CM. Safety of inhaled and intranasal corticosteroids: lessons for the new millennium. Drug Saf, 2000,23(1):11–33
Waljee AK, Rogers MA, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ, 2017,357:j1415
Murr AH, Smith TL, Hwang PH, et al. Safety and efficacy of a novel bioabsorbable, steroid-eluting sinus stent. Int Forum Allergy Rhinol, 2011,1(1):23–32
Luong A, Ow RA, Singh A, et al. Safety and Effectiveness of a Bioabsorbable Steroid-Releasing Implant for the Paranasal Sinus Ostia: A Randomized Clinical Trial. JAMA Otolaryngol Head Neck Surg, 2018,144(1):28–35
Kennedy DW. The PROPEL™ steroid-releasing bioabsorbable implant to improve outcomes of sinus surgery. Expert Rev Respir Med, 2012,6(5):493–498
Huang Z, Zhou B, Wang D, et al. Comparison of Bioabsorbable Steroid-Eluting Sinus Stents Versus Nasopore After Endoscopic Sinus Surgery:A Multicenter, Randomized, Controlled, Single-Blinded Clinical Trial. Ear Nose Throat J, 2020,145561320947632
Forwith KD, Chandra RK, Yun PT, et al. ADVANCE: a multisite trial of bioabsorbable steroid-eluting sinus implants. Laryngoscope, 2011,121(11):2473–2480
Goshtasbi K, Abouzari M, Abiri A, et al. Efficacy of steroid-eluting stents in management of chronic rhinosinusitis after endoscopic sinus surgery: updated meta-analysis. Int Forum Allergy Rhinol, 2019,9(12):1443–1450
Hong H, Wang D, Tan KS, et al. Sinus computed tomography predicts clinical response to corticosteroids in chronic rhinosinusitis with nasal polyps. Clin Transl Allergy, 2018,8:24
Epperson MV, Phillips KM, Caradonna DS, et al. Predictors of efficacy for combination oral and topical corticosteroids to treat patients with chronic rhinosinusitis with nasal polyps. Int Forum Allergy Rhinol, 2019,9(12):1436–1442
Reddel HK, Bateman ED, Becker A, et al. A summary of the new GINA strategy: a roadmap to asthma control. Eur Respir J, 2015,46(3):622–639
Hu Y, Cao PP, Liang GT, et al. Diagnostic significance of blood eosinophil count in eosinophilic chronic rhinosinusitis with nasal polyps in Chinese adults. Laryngoscope, 2012,122(3):498–503
Tajudeen BA, Ganti A, Kuhar HN, et al. The presence of eosinophil aggregates correlates with increased postoperative prednisone requirement. Laryngoscope, 2019,129(4):794–799
Xu G, Jiang H, Li H, et al. Stages of nasal mucosal transitional course after functional endoscopic sinus surgery and their clinical indications. ORL J Otorhinolaryngol Relat Spec, 2008,70(2):118–123
Gurrola J2nd, Borish L. Chronic rhinosinusitis: Endotypes, biomarkers, and treatment response. J Allergy Clin Immunol, 2017,140(6):1499–1508
Sella GCP, Tamashiro E, Sella JA, et al. Asthma Is the Dominant Factor for Recurrence in Chronic Rhinosinusitis. J Allergy Clin Immunol Pract, 2020,8 (1):302–309
Bai J, Huang JH, Price CPE, et al. Prognostic factors for polyp recurrence in chronic rhinosinusitis with nasal polyps. J Allergy Clin Immunol, 2022,150(2):352–361.e357
Liao B, Liu JX, Li ZY, et al. Multidimensional endotypes of chronic rhinosinusitis and their association with treatment outcomes. Allergy, 2018,73(7):1459–1469
Tomassen P, Vandeplas G, Van Zele T, et al. Inflammatory endotypes of chronic rhinosinusitis based on cluster analysis of biomarkers. J Allergy Clin Immunol, 2016,137(5):1449–1456.e1444
Laidlaw TM, Mullol J, Woessner KM, et al. Chronic Rhinosinusitis with Nasal Polyps and Asthma. J Allergy Clin Immunol Pract, 2021,9(3):1133–1141
de Borja Callejas F, Martínez-Antón A, Picado C, et al. Corticosteroid treatment regulates mucosal remodeling in chronic rhinosinusitis with nasal polyps. Laryngoscope, 2015,125(5):E158–E167
Jin M, Watkins S, Larriba Y, et al. Real-time imaging of asthmatic epithelial cells identifies migratory deficiencies under type-2 conditions. J Allergy Clin Immunol, 2022,149(2):579–588
Zeng M, Wang H, Liao B, et al. Clinical and Biological Markers Predict the Efficacy of Glucocorticoid- and Macrolide-Based Postoperative Therapy in Patients With Chronic Rhinosinusitis. Am J Rhinol Allergy, 2021,35(5):596–606
Vento SI, Blomgren K, Hytönen M, et al. Prevention of relapses of nasal polyposis with intranasal triamcinolone acetonide after polyp surgery: a prospective double-blind, placebo-controlled, randomised study with a 9-month follow-up. Clin Otolaryngol, 2012,37(2):117–123
De Corso E, Settimi S, Tricarico L, et al. Predictors of Disease Control After Endoscopic Sinus Surgery Plus Long-Term Local Corticosteroids in CRSwNP. Am J Rhinol Allergy, 2021,35(1):77–85
Mukherjee M, Svenningsen S, Nair P. Glucocortiosteroid subsensitivity and asthma severity. Curr Opin Pulm Med, 2017,23(1):78–88
Fahy JV. Type 2 inflammation in asthma—present in most, absent in many. Nat Rev Immunol, 2015,15(1):57–65
Fan Y, Chen S, Qu X, et al. A lower prevalence of asthma among patients with chronic rhinosinusitis in southern China. J Allergy Clin Immunol, 2011,127(2):520–522.e521–e525
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All authors declare no conflict of interest regarding the publication of this article.
Author Zheng LIU is a member of the Editorial Board for Current Medical Science. The paper was handled by other editors and has undergone rigorous peer review process. Author Zheng LIU was not involved in the journal’s review of, or decisions related to, this manuscript.
Additional information
This study was supported by grants from the National Natural Science Foundation of China (No. 81873694), the Key Research and Development Program of Hubei Province (No. 2021BCA119 and No. 2022BCA005), and Knowledge Innovation Program of Wuhan-Basic Research (No. 2022020801010446).
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, A., Li, T., Li, Ms. et al. Association of Comorbid Asthma and the Efficacy of Bioabsorbable Steroid-eluting Sinus Stents Implanted After Endoscopic Sinus Surgery in Patients with Chronic Rhinosinusitis with Nasal Polyps. CURR MED SCI 43, 1005–1012 (2023). https://doi.org/10.1007/s11596-023-2786-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-023-2786-6