Abstract
Objective
Metabolic disorders are regarded as hallmarks of multiple myeloma (MM) and are responsible for rapid cancer cell proliferation and tumor growth. However, the exact biological roles of metabolites in MM cells have not been fully explored. This study aimed to explore the feasibility and clinical significance of lactate for MM and investigate the molecular mechanism of lactic acid (Lac) in the proliferation of myeloma cells and cell sensitivity to bortezomib (BTZ).
Methods
Metabolomic analysis of the serum was carried out to obtain metabolites expression and clinical characteristics in MM patients. The CCK8 assay and flow cytometry were used to detect cell proliferation, apoptosis, and cell cycle changes. Western blotting was used to detect the potential mechanism and apoptosis- and cycle-related protein changes.
Results
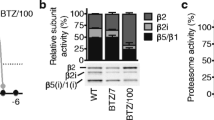
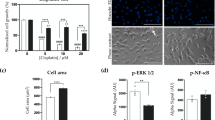
Lactate was highly expressed in both the peripheral blood and bone marrow of MM patients. It was significantly correlated with Durie-Salmon Staging (DS Staging) and the International Staging System (ISS Staging) and the serum and urinary involved/uninvolved free light chain ratios. Patients with relatively high lactate levels had a poor treatment response. Moreover, in vitro experiments showed that Lac could promote the proliferation of tumor cells and decrease the proportion of G0/G1-phase cells, which was accompanied by an increased proportion of S-phase cells. In addition, Lac could decrease tumor sensitivity to BTZ by disrupting the expression of nuclear factor kappa B subunit 2 (NFkB2) and RelB.
Conclusion
Metabolic changes are important in MM cell proliferation and treatment response; lactate could be used as a biomarker in MM and as a therapeutic target to overcome cell resistance to BTZ.
Similar content being viewed by others
References
Röllig C, Knop S, Bornhäuser M. Multiple myeloma. Lancet, 2015,385(9983):2197–2208
Cowan AJ, Green DJ, Kwok M, et al. Diagnosis and Management of Multiple Myeloma. JAMA, 2022,327(5):464–477
Durie BGM, Salmon SE. A clinical staging system for multiple myeloma correlation of measured myeloma cell mass with presenting clinical features, response to treatment, and survival. Cancer, 1975,36(3):842–854
Greipp PR, Miguel JS, Durie BGM, et al. International Staging System for Multiple Myeloma. J Clin Oncol, 2005,23(15):3412–3420
Warburg O, Wind F, Negelein E. The metabolism of tumors in the body. J Gen Physiol, 1927,8(6):519–530
Moreno-Felici J, Hyroššová P, Aragó M, et al. Phosphoenolpyruvate from Glycolysis and PEPCK Regulate Cancer Cell Fate by Altering Cytosolic Ca2+. Cells, 2020,9(1):18
Roudier E, Bachelet C, Perrin A. Pyruvate reduces DNA damage during hypoxia and after reoxygenation in hepatocellular carcinoma cells. FEBS J, 2007,274(19):5188–5198
Hirschhaeuser F, Sattler UGA, Mueller-Klieser W. Lactate: A Metabolic Key Player in Cancer. Cancer Res, 2011,71(22):6921–6925
Ngwa VM, Edwards DN, Philip M, et al. Microenvironmental Metabolism Regulates Antitumor Immunity. Cancer Res, 2019,79(16):4003–4008
Dong Q, Zhou C, Ren H, et al. Lactate-induced MRP1 expression contributes to metabolism-based etoposide resistance in non-small cell lung cancer cells. Cell Commun Signal, 2020,18(1):167
Govoni M, Rossi V, Di Stefano G, et al. Lactate Upregulates the Expression of DNA Repair Genes, Causing Intrinsic Resistance of Cancer Cells to Cisplatin. Pathol Oncol Res, 2021,27:1609951
Qu J, Sun Z, Peng C, et al. C. tropicalis promotes chemotherapy resistance in colon cancer through increasing lactate production to regulate the mismatch repair system. Int J Biol Sci, 2021,17(11):2756–2769
Rossi V, Govoni M, Farabegoli F, et al. Lactate is a potential promoter of tamoxifen resistance in MCF7 cells. Biochim Biophys Acta Gen Subj, 2022,1866(9):130185
Ustun C, Fall P, Szerlip HM, et al. Multiple Myeloma Associated with Lactic Acidosis. Leuk Lymphoma, 2009,43(12):2395–2397
Abdullah SY, Ali MK, Sabha MM. Type-B lactic acidosis associated with progressive multiple myeloma. Saudi Med J, 2015,36(2):239–242
Rajkumar SV, Dimopoulos MA, Palumbo A, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol, 2014,15(12):e538–e548
Garcés J, Cedena M, Puig N, et al. Circulating Tumor Cells for the Staging of Patients With Newly Diagnosed Transplant-Eligible Multiple Myeloma. J Clin Oncol, 2022,40(27):3151–3161
Bertamini L, Oliva S, Rota-Scalabrini D, et al. High Levels of Circulating Tumor Plasma Cells as a Key Hallmark of Aggressive Disease in Transplant-Eligible Patients With Newly Diagnosed Multiple Myeloma. J Clin Oncol, 2022,40(27):3120–3131
Okorie ON, Dellinger P. Lactate: Biomarker and Potential Therapeutic Target. Crit Care Clin, 2011,27(2):299–326
Hayes C, Donohoe CL, Davern M, et al. The oncogenic and clinical implications of lactate induced immunosuppression in the tumour microenvironment. Cancer Lett, 2021,500:75–86
Mariappan R, Venkatraghavan L, Vertanian A, et al. Serum lactate as a potential biomarker of malignancy in primary adult brain tumours. J Clin Neurosci, 2015,22(1):144–148
Branco M, Linhares P, Carvalho B, et al. Serum lactate levels are associated with glioma malignancy grade. Clin Neurol Neurosurg, 2019,186:105546
Kumar SK, Dispenzieri A, Lacy MQ, et al. Continued improvement in survival in multiple myeloma: changes in early mortality and outcomes in older patients. Leukemia, 2014,28(5):1122–1128
Joseph NS, Kaufman JL, Dhodapkar MV, et al. Long-Term Follow-Up Results of Lenalidomide, Bortezomib, and Dexamethasone Induction Therapy and Risk-Adapted Maintenance Approach in Newly Diagnosed Multiple Myeloma. J Clin Oncol, 2020,38(17):1928–1937
Franke NE, Niewerth D, Assaraf YG, et al. Impaired bortezomib binding to mutant β5 subunit of the proteasome is the underlying basis for bortezomib resistance in leukemia cells. Leukemia, 2012,26(4):757–768
Dytfeld D, Luczak M, Wrobel T, et al. Comparative proteomic profiling of refractory/relapsed multiple myeloma reveals biomarkers involved in resistance to bortezomib-based therapy. Oncotarget, 2016–7(35):56726–56736
Qiang Y, Ye S, Chen Y, et al. MAF protein mediates innate resistance to proteasome inhibition therapy in multiple myeloma. Blood, 2016,128(25):2919–2930
Zhang L, Rastgoo N, Wu J, et al. MARCKS inhibition cooperates with autophagy antagonists to potentiate the effect of standard therapy against drug-resistant multiple myeloma. Cancer Lett, 2020,480:29–38
Annunziata CM, Davis RE, Demchenko Y, et al. Frequent Engagement of the Classical and Alternative NF-κB Pathways by Diverse Genetic Abnormalities in Multiple Myeloma. Cancer Cell, 2007,12(2):115–130
Keats JJ, Fonseca R, Chesi M, et al. Promiscuous Mutations Activate the Noncanonical NF-κB Pathway in Multiple Myeloma. Cancer Cell, 2007,12(2):131–144
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflict of interests.
Additional information
This work was supported by grants from the National Natural Science Foundation of China (No. 82070208), the Military Clinical Medical Innovation Project of Xinqiao Hospital (No. 2021JSLC0003), the National Natural Science Foundation of Chongqing (No. cstc2020jcyj-msxmX0433), the Translational Research Grant of NCRCH (Nos. 2020ZKZC02, 2021WWB05), and the Chongqing Science and Health Joint Medical Research Project (Nos. 2021MSXM226, 2023QNXM047).
Supplementary data
Rights and permissions
About this article
Cite this article
Ma, Ny., Li, Q., Li, Xl. et al. Lactate Decreases Bortezomib Sensitivity and Predicts Poor Clinical Outcomes of Multiple Myeloma. CURR MED SCI 43, 679–688 (2023). https://doi.org/10.1007/s11596-023-2747-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-023-2747-0