Abstract
Objective
Disc calcification is strongly associated with disc degeneration; however, the underlying mechanisms driving its pathogenesis are poorly understood. This study aimed to provide a gene expression profile of nucleus pulposus cells (NPCs) from calcified discs, and clarify the potential mechanism in disc degeneration.
Methods
Primary NPCs were isolated from calcified and control discs (CAL-NPC and CON-NPC), respectively. The proliferation and extracellular matrix (ECM) metabolism capacities of the cells were evaluated using MTT and Western blotting, respectively. RNA sequencing was used to identify differentially expressed genes (DEGs) in the CAL-NPCs. The biological functions of the DEGs were analyzed using the Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) databases. The transcription factor database and Cytoscape software were used to construct the transcription factor-DEGs regulatory network. The role of the verified transcription factor in NPC proliferation and ECM metabolism was also investigated.
Results
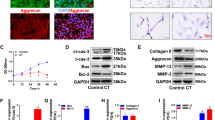
The CAL-NPCs exhibited a lower proliferation rate and higher ECM degradation capacity than the CON-NPCs. In total, 375 DEGs were identified in the CAL-NPCs. The GO and KEGG analyses showed that the DEGs were primarily involved in the regulation of ribonuclease activity and NF-kappa B and p53 signaling pathways. GATA-binding protein 3 (GATA3) with the highest verified levels was selected for further studies. Overexpression of GATA3 in the CON-NPCs significantly inhibited their proliferation and promoted their ECM degradation function, while the knockdown of GATA3 in the CAL-NPCs resulted in the opposite phenotypes.
Conclusion
This study provided a comprehensive gene expression profile of the NPCs from the calcified discs and supported that GATA3 could be a potential target for reversing calcification-associated disc degeneration.
Similar content being viewed by others
References
Urban JP, Roberts S. Degeneration of the intervertebral disc. Arthritis Res Ther, 2003,5(3):120–130
Cheung KM, Karppinen J, Chan D, et al. Prevalence and pattern of lumbar magnetic resonance imaging changes in a population study of one thousand forty-three individuals. Spine (Phila Pa 1976), 2009,34(9):934–940
Global Burden of Disease Study C. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet, 2015,386(9995):743–800
Gore M, Sadosky A, Stacey BR, et al. The burden of chronic low back pain: clinical comorbidities, treatment patterns, and health care costs in usual care settings. Spine (Phila Pa 1976), 2012,37(11):E668–677
Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet, 2012,380(9859):2163–2196
Risbud MV, Shapiro IM. Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat Rev Rheumatol, 2014,10(1):44–56
Wang J, Tian Y, Phillips KL, et al. Tumor necrosis factor alpha- and interleukin-1beta-dependent induction of CCL3 expression by nucleus pulposus cells promotes macrophage migration through CCR1. Arthritis Rheum, 2013,65(3):832–842
Sakai D, Nakamura Y, Nakai T, et al. Exhaustion of nucleus pulposus progenitor cells with ageing and degeneration of the intervertebral disc. Nat Commun, 2012,3:1264
Fardon DF, Williams AL, Dohring EJ, et al. Lumbar disc nomenclature: version 2.0: Recommendations of the combined task forces of the North American Spine Society, the American Society of Spine Radiology and the American Society of Neuroradiology. Spine J, 2014,14(11):2525–2545
Chen Y, Wang JX, Sun B, et al. Percutaneous Endoscopic Lumbar Discectomy in Treating Calcified Lumbar Intervertebral Disc Herniation. World Neurosurg, 2019,122:e1449–e1456
Dussault RG, Kaye JJ. Intervertebral disk calcification associated with spine fusion. Radiology, 1977,125(1):57–61
Krzyzanowska AK, Frawley RJ, Damle S, et al. Activation of nuclear factor-kappa B by TNF promotes nucleus pulposus mineralization through inhibition of ANKH and ENPP1. Sci Rep, 2021,11(1):8271
Rutges JP, Duit RA, Kummer JA, et al. Hypertrophic differentiation and calcification during intervertebral disc degeneration. Osteoarthritis Cartilage, 2010,18(11):1487–1495
Zhang Z, Huo Y, Zhou Z, et al. Role of lncRNA PART1 in intervertebral disc degeneration and associated underlying mechanism. Exp Ther Med, 2021,21(2):131
Matys V, Fricke E, Geffers R, et al. TRANSFAC: transcriptional regulation, from patterns to profiles. Nucleic Acids Res, 2003,31(1):374–378
Battie MC, Videman T, Levalahti E, et al. Genetic and environmental effects on disc degeneration by phenotype and spinal level: a multivariate twin study. Spine (Phila Pa 1976), 2008,33(25):2801–2808
Grant MP, Epure LM, Bokhari R, et al. Human cartilaginous endplate degeneration is induced by calcium and the extracellular calcium-sensing receptor in the intervertebral disc. Eur Cell Mater, 2016,32:137–151
Lv FJ, Peng Y, Lim FL, et al. Matrix metalloproteinase 12 is an indicator of intervertebral disc degeneration co-expressed with fibrotic markers. Osteoarthritis Cartilage, 2016,24(10):1826–1836
Fernandes LM, Khan NM, Trochez CM, et al. Single-cell RNA-seq identifies unique transcriptional landscapes of human nucleus pulposus and annulus fibrosus cells. Sci Rep, 2020,10(1):15263
Johnson ZI, Schoepflin ZR, Choi H, et al. Disc in flames: Roles of TNF-alpha and IL-1beta in intervertebral disc degeneration. Eur Cell Mater, 2015,30:104–116; discussion 116–107
Weber KT, Satoh S, Alipui DO, et al. Exploratory study for identifying systemic biomarkers that correlate with pain response in patients with intervertebral disc disorders. Immunol Res, 2015,63(1–3):170–180
Liu Y, Qu Y, Liu L, et al. PPAR-gamma agonist pioglitazone protects against IL-17 induced intervertebral disc inflammation and degeneration via suppression of NF-kappaB signaling pathway. Int Immunopharmacol, 2019,72:138–147
Yamaguchi Y, Ayaki T, Li F, et al. Phosphorylated NF-kappaB subunit p65 aggregates in granulovacuolar degeneration and neurites in neurodegenerative diseases with tauopathy. Neurosci Lett, 2019,704:229–235
Wang K, Chen T, Ying X, et al. Ligustilide alleviated IL-1beta induced apoptosis and extracellular matrix degradation of nucleus pulposus cells and attenuates intervertebral disc degeneration in vivo. Int Immunopharmacol, 2019,69:398–407
Zhang K, Zhang Y, Zhang C, et al. Upregulation of P53 promotes nucleus pulposus cell apoptosis in intervertebral disc degeneration through upregulating NDRG2. Cell Biol Int, 2021,45(9):1966–1975
Sanges D, Romo N, Simonte G, et al. Wnt/beta-catenin signaling triggers neuron reprogramming and regeneration in the mouse retina. Cell Rep, 2013,4(2):271–286
Smolders LA, Meij BP, Onis D, et al. Gene expression profiling of early intervertebral disc degeneration reveals a down-regulation of canonical Wnt signaling and caveolin-1 expression: implications for development of regenerative strategies. Arthritis Res Ther, 2013,15(1):R23
Holguin N, Silva MJ. In-Vivo Nucleus Pulposus-Specific Regulation of Adult Murine Intervertebral Disc Degeneration via Wnt/Beta-Catenin Signaling. Sci Rep, 2018,8(1):11191
Farhan M, Wang H, Gaur U, et al. FOXO Signaling Pathways as Therapeutic Targets in Cancer. Int J Biol Sci, 2017,13(7):815–827
Pardal J, Sundram U, Selim MA, et al. GATA3 and MYB Expression in Cutaneous Adnexal Neoplasms. Am J Dermatopathol, 2017,39(4):279–286
Yan Q, Xiao Q, Ge J, et al. Bioinformatics-Based Research on Key Genes and Pathways of Intervertebral Disc Degeneration. Cartilage, 2020,13(2_suppl):582S–591S
Pang L, Yu P, Liu X, et al. Fine particulate matter induces airway inflammation by disturbing the balance between Th1/Th2 and regulation of GATA3 and Runx3 expression in BALB/c mice. Mol Med Rep, 2021,23(5):378
Zhang GZ, Liu MQ, Chen HW, et al. NF-kappaB signalling pathways in nucleus pulposus cell function and intervertebral disc degeneration. Cell Prolif, 2021,54(7):e13057
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors report no conflicts of interest.
Additional information
This research was funded by the Youth Research Fund of the Peking Union Medical College Hospital (No. pumch201911708).
Supplementary data
Rights and permissions
About this article
Cite this article
Dong, Yl., Tang, N., Zhao, H. et al. Nucleus Pulposus Cells from Calcified Discs Promote the Degradation of the Extracellular Matrix through Upregulation of the GATA3 Expression. CURR MED SCI 43, 146–155 (2023). https://doi.org/10.1007/s11596-022-2686-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-022-2686-1