Abstract
Objective
This study aimed to develop a nomogram to predict the overall survival (OS) of patients with acinar-predominant adenocarcinoma (APA).
Methods
Data from patients with APA obtained from the Surveillance, Epidemiology, and End Results (SEER) database between 2008 and 2016 were used. Significant prognostic factors were incorporated to construct a nomogram for predicting the 1-, 3-, and 5-year OS in these patients. The discrimination and calibration abilities of the nomogram were assessed using a C-index and calibration curves, respectively.
Results
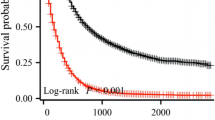
A total of 2242 patients with APA were randomly divided into a training cohort (n=1576) and validation cohort (n=666). The independent prognostic factors for OS incorporated into the nomogram included marital status, age, gender, differentiation grade, T stage, N stage, and M stage. The nomogram showed good prediction capability, as indicated by the C-index [0.713, 95% confidence interval (CI): 0.705–0.721 in the training cohort, and 0.662, 95% CI: 0.649–0.775 in the validation cohort]. The calibration curves demonstrated that the 1-, 3-, and 5-year OS probabilities were consistent between the observed and predicted outcome frequencies. Patients were divided into the high-risk and low-risk groups with the former showing significantly worse survival than the latter (P<0.001).
Conclusion
Using the SEER database, a nomogram was established to predict the 1-, 3-, and 5-year OS of patients with APA and was superior to the tumor size, lymph node, and metastasis staging system in terms of evaluating long-term prognosis.
Similar content being viewed by others
References
Travis WD, Brambilla E, Nicholson AG, et al. The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification. J Thorac Oncol, 2015,10(9):1243–1260
Lantuejoul S, Rouquette I, Brambilla E, et al. New WHO classification of lung adenocarcinoma and preneoplasia. Ann Pathol,2016,36(1):5–14
Tsao MS, Marguet S, Le Teuff G, et al. Subtype Classification of Lung Adenocarcinoma Predicts Benefit From Adjuvant Chemotherapy in Patients Undergoing Complete Resection. J Clin Oncol, 2015,33(30):3439–3346
Ito M, Miyata Y, Tsutani Y, et al. Positive EGFR mutation status is a risk of recurrence in pN0-1 lung adenocarcinoma when combined with pathological stage and histological subtype: A retrospective multi-center analysis. Lung Cancer, 2020,141:107–113
Mansuet-Lupo A, Bobbio A, Blons H, et al. The new histologic classification of lung primary adenocarcinoma subtypes is a reliable prognostic marker and identifies tumors with different mutation status: the experience of a French cohort. Chest, 2014,146(3):633–643
Yanagawa N, Shiono S, Abiko M, et al. New IASLC/ATS/ERS classification and invasive tumor size are predictive of disease recurrence in stage I lung adenocarcinoma. J Thorac Oncol, 2013,8(5):612–618
Li C, Shen Y, Hu F, et al. Micropapillary pattern is associated with the development of brain metastases and the reduction of survival time in EGFR-mutation lung adenocarcinoma patients with surgery. Lung Cancer, 2020,141:72–77
Ma M, She Y, Ren Y, et al. Micropapillary or solid pattern predicts recurrence free survival benefit from adjuvant chemotherapy in patients with stage IB lung adenocarcinoma. J Thorac Dis, 2018,10(9):5384–5393
Yaldız D, Acar A, Örs Kaya Ş, et al. Papillary predominant histological subtype predicts poor survival in lung adenocarcinoma. Turk Gogus Kalp Damar Cerrahisi Derg, 2019,27(3):360–366
Yoshizawa A, Sumiyoshi S, Sonobe M, et al. Validation of the IASLC/ATS/ERS lung adenocarcinoma classification for prognosis and association with EGFR and KRAS gene mutations: analysis of 440 Japanese patients. J Thorac Oncol, 2013,8(1):52–61
Ito H, Nakayama H, Murakami, et al. Does the histologic predominance of pathological stage IA lung adenocarcinoma influence the extent of resection? Gen Thorac Cardiovasc Surg, 2017,65(9):512–518
Zombori T, Furák J, Nyári T, et al. Evaluation of grading systems in stage I lung adenocarcinomas: a retrospective cohort study. J Clin Pathol, 2018,71(2):135–140
Moreira AL, Ocampo PSS, Xia Y, et al. A Grading System for Invasive Pulmonary Adenocarcinoma: A Proposal From the International Association for the Study of Lung Cancer Pathology Committee. J Thorac Oncol, 2020,15(10):1599–1610
Ito M, Miyata Y, Yoshiya T, et al. Second predominant subtype predicts outcomes of intermediate-malignant invasive lung adenocarcinoma. Eur J Cardiothorac Surg, 2017,51(2):218–222
Li H, Wang Z, Yang F, et al. Development and validation of a nomogram for predicting cancer-specific survival of surgical resected stage I–II adenosquamous carcinoma of the lung. J Surg Oncol, 2020,121(6):1027–1035
Zeng Y, Mayne N, Yang CJ, et al. A Nomogram for Predicting Cancer-Specific Survival of TNM 8th Edition Stage I Non-small-cell Lung Cancer. Ann Surg Oncol, 2019,26(7):2053–2062
Iasonos A, Schrag D, Raj GV, et al. How to build and interpret a nomogram for cancer prognosis. J Clin Oncol, 2008,26(8):1364–1370
Shariat SF, Karakiewicz PI, Suardi N, et al. Comparison of nomograms with other methods for predicting outcomes in prostate cancer: a critical analysis of the literature. Clin Cancer Res, 2008,14(14):4400–4407
Heagerty PJ, Lumley T, Pepe MS. Time-dependent ROC curves for censored survival data and a diagnostic marker. Biometrics, 2000,56(2):337–344
Pencina MJ, D’Agostino RB, Sr., D’Agostino RB, Jr., et al. Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat Med, 2008,27(2):157–172
Vickers AJ, Elkin EB. Decision curve analysis: a novel method for evaluating prediction models. Med Decis Making, 2006,26(6):565–574
Mackinnon AC, Jr., Luevano A, de Araujo LC, et al. Cribriform adenocarcinoma of the lung: clinicopathologic, immunohistochemical, and molecular analysis of 15 cases of a distinctive morphologic subtype of lung adenocarcinoma. Mod Pathol, 2014,27(8):1063–1072
Kim M, Chung YS, Kim KA, et al. Prognostic factors of acinar- or papillary-predominant adenocarcinoma of the lung. Lung Cancer, 2019,137:129–135
Lu D, Yang J, Liu X, et al. Clinicopathological features, survival outcomes, and appropriate surgical approaches for stage I acinar and papillary predominant lung adenocarcinoma. Cancer Med, 2020,9(10):3455–3462
Suzuki K, Saji H, Aokage K, et al. Comparison of pulmonary segmentectomy and lobectomy: Safety results of a randomized trial. J Thorac Cardiovasc Surg, 2019,158(3):895–907
Wen Z, Zhao Y, Fu F, et al. Comparison of outcomes following segmentectomy or lobectomy for patients with clinical N0 invasive lung adenocarcinoma of 2 cm or less in diameter. J Cancer Res Clin Oncol, 2020,146(6):1603–1613
Cao S, Teng J, Xu J, et al. Value of adjuvant chemotherapy in patients with resected stage IB solid predominant and solid non-predominant lung adenocarcinoma. Thorac Cancer, 2019,10(2):249–255
Wang C, Yang J, Lu M. Micropapillary Predominant Lung Adenocarcinoma in Stage IA Benefits from Adjuvant Chemotherapy. Ann Surg Oncol, 2020,27(6):2051–2060
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no competing interests.
Additional information
This study was supported in part by the National Key Research and Development Program of China (No. 2016YFC1303201).
Rights and permissions
About this article
Cite this article
Zuo, Zc., Wang, Ld., Peng, K. et al. Development and Validation of a Nomogram for Predicting the 1-, 3-, and 5-year Survival in Patients with Acinar-predominant Lung Adenocarcinoma. CURR MED SCI 42, 1178–1185 (2022). https://doi.org/10.1007/s11596-022-2672-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-022-2672-7