Abstract
Objective
Cardiac remodeling is a common pathological change in various cardiovascular diseases and can ultimately result in heart failure. Thus, there is an urgent need for more effective strategies to aid in cardiac protection. Our previous work found that sphingosine-1-phosphate (S1P) could ameliorate cardiac hypertrophy. In this study, we aimed to investigate whether S1P could prevent cardiac fibrosis and the associated mechanisms in cardiac remodeling.
Methods
Eight-week-old male C57BL/6 mice were randomly divided into a sham, transverse aortic constriction (TAC) or a TAC+S1P treatment group.
Results
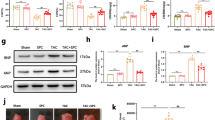
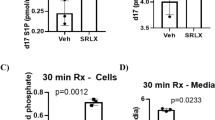
We found that S1P treatment improved cardiac function in TAC mice and that the cardiac fibrosis ratio in the TAC+S1P group was significantly lower and was accompanied by a decrease in α-smooth muscle actin (α-SMA) and collagen type I (COL I) expression compared with the TAC group. We also found that one of the key S1P enzymes, sphingosine kinase 2 (SphK2), which was mainly distributed in cytoblasts, was downregulated in the cardiac remodeling case and recovered after S1P treatment in vivo and in vitro. In addition, our in vitro results showed that S1P treatment activated extracellular regulated protein kinases (ERK) phosphorylation mainly through the S1P receptor 2 (S1PR2) and spurred p-ERK transposition from the cytoplasm to cytoblast in H9c2 cells exposed to phenylephrine.
Conclusion
These findings suggest that SphK2 and the S1PR2/ERK pathway may participate in the anti-remodeling effect of S1P on the heart. This work therefore uncovers a novel potential therapy for the prevention of cardiac remodeling.
Similar content being viewed by others
References
Yang MQ, Song YM, Gao HY, et al. Efficacy and Safety of Fuzi Formulae on the Treatment of Heart Failure as Complementary Therapy: A Systematic Review and Meta-Analysis of High-Quality Randomized Controlled Trials. Evid Based Complement Alternat Med, 2019, 2019:9728957
Su Q, Zhang P, Yu D, et al. Upregulation of miR-93 and inhibition of LIMK1 improve ventricular remodeling and alleviate cardiac dysfunction in rats with chronic heart failure by inhibiting RhoA/ROCK signaling pathway activation. Aging (Albany NY), 2019,11(18):7570–7586
Kong P, Christia P, Frangogiannis NG. The pathogenesis of cardiac fibrosis. Cell Mol Life Sci, 2014,71(4):549–574
Kyselovic J, Leddy JJ. Cardiac Fibrosis: The Beneficial Effects of Exercise in Cardiac Fibrosis. Adv Exp Med Biol, 2017,999:257–268
Cartier A, Hla T. Sphingosine 1-phosphate: Lipid signaling in pathology and therapy. Science, 2019,366(6463): eaar5551
Siow D, Wattenberg B. The compartmentalization and translocation of the sphingosine kinases: mechanisms and functions in cell signaling and sphingolipid metabolism. Crit Rev Biochem Mol Biol, 2011,46(5):365–375
Maceyka M, Spiegel S. Sphingolipid metabolites in inflammatory disease. Nature, 2014,510(7503):58–67
Kunkel GT, Maceyka M, Milstien S, et al. Targeting the sphingosine-1-phosphate axis in cancer, inflammation and beyond. Nat Rev Drug Discov, 2013,12(9):688–702
Bandhuvula P, Honbo N, Wang GY, et al. S1P lyase: a novel therapeutic target for ischemia-reperfusion injury of the heart. Am J Physiol Heart Circ Physiol, 2011,300(5):H1753–1761
Zhang J, Honbo N, Goetzl EJ, et al. Signals from type 1 sphingosine 1-phosphate receptors enhance adult mouse cardiac myocyte survival during hypoxia. Am J Physiol Heart Circ Physiol, 2007,293(5):H3150–3158
Means CK, Brown JH. Sphingosine-1-phosphate receptor signalling in the heart. Cardiovasc Res, 2009,82(2):193–200
Rosen H, Stevens RC, Hanson M, et al. Sphingosine-1-phosphate and its receptors: structure, signaling, and influence. Annu Rev Biochem, 2013,82:637–662
Yan H, Yi S, Zhuang H, et al. Sphingosine-1-phosphate ameliorates the cardiac hypertrophic response through inhibiting the activity of histone deacetylase-2. Int J Mol Med, 2018,41(3):1704–1714
National Research Council (U.S.). Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Institute for Laboratory Animal Research (U.S.), National Academies Press (U.S.), Guide for the care and use of laboratory animals. National Academies Press: Washington, D.C., 2011:p. xxv, 220 p.
Takimoto E, Champion HC, Li M, et al. Chronic inhibition of cyclic GMP phosphodiesterase 5A prevents and reverses cardiac hypertrophy. Nat Med, 2005,11(2):214–222
Ma H, Gong H, Chen Z, et al. Association of Stat3 with HSF1 plays a critical role in G-CSF-induced cardioprotection against ischemia/reperfusion injury. J Mol Cell Cardiol, 2012,52(6):1282–1290
Yan H, Meng J, Zhang S, et al. Pretreatment of rAAV-Mediated Expression of Myostatin Propeptide Lowers Type 2 Diabetes Incidence in C57BL/6 Mice on a High-Fat Diet. Hum Gene Ther, 2019,30(5):661–671
Karliner JS. Sphingosine kinase and sphingosine 1-phosphate in the heart: a decade of progress. Biochim Biophys Acta, 2013,1831(1):203–212
Kennedy S, Kane KA, Pyne NJ, et al. Targeting sphingosine-1-phosphate signalling for cardioprotection. Curr Opin Pharmacol, 2009,9(2):194–201
Gellings Lowe N, Swaney JS, Moreno KM, et al. Sphingosine-1-phosphate and sphingosine kinase are critical for transforming growth factor-beta-stimulated collagen production by cardiac fibroblasts. Cardiovasc Res, 2009,82(2):303–312
Pchejetski D, Foussal C, Alfarano C, et al. Apelin prevents cardiac fibroblast activation and collagen production through inhibition of sphingosine kinase 1. Eur Heart J, 2012,33(18):2360–2369
Huang LS, Berdyshev E, Mathew B, et al. Targeting sphingosine kinase 1 attenuates bleomycin-induced pulmonary fibrosis. FASEB J, 2013,27(4):1749–1760
Schwalm S, Timcheva TM, Filipenko I, et al. Sphingosine kinase 2 deficiency increases proliferation and migration of renal mouse mesangial cells and fibroblasts. Biol Chem, 2015,396(6–7):813–825
Pang M, Zhuang S. Histone deacetylase: a potential therapeutic target for fibrotic disorders. J Pharmacol Exp Ther, 2010,335(2):266–272
Renaud L, Harris LG, Mani SK, et al. HDACs Regulate miR-133a Expression in Pressure Overload-Induced Cardiac Fibrosis. Circ Heart Fail, 2015,8(6):1094–1104
Hait NC, Allegood J, Maceyka M, et al. Regulation of histone acetylation in the nucleus by sphingosine-1-phosphate. Science, 2009,325(5945):1254–1257
Ahmed N, Linardi D, Muhammad N, et al. Sphingosine 1-Phosphate Receptor Modulator Fingolimod (FTY720) Attenuates Myocardial Fibrosis in Post-heterotopic Heart Transplantation. Front Pharmacol, 2017,8:645
Ohkura SI, Usui S, Takashima SI, et al. Augmented sphingosine 1 phosphate receptor-1 signaling in cardiac fibroblasts induces cardiac hypertrophy and fibrosis through angiotensin II and interleukin-6. PLoS One, 2017,12(8):e0182329
Ding BS, Liu CH, Sun Y, et al. HDL activation of endothelial sphingosine-1-phosphate receptor-1 (S1P1) promotes regeneration and suppresses fibrosis in the liver. JCI Insight, 2016,1(21):e87058
Takuwa N, Ohkura S, Takashima S, et al. S1P3-mediated cardiac fibrosis in sphingosine kinase 1 transgenic mice involves reactive oxygen species. Cardiovasc Res, 2010,85(3):484–493
Yung BS, Brand CS, Xiang SY, et al. Selective coupling of the S1P3 receptor subtype to S1P-mediated RhoA activation and cardioprotection. J Mol Cell Cardiol, 2017,103:1–10
Ikeda H, Watanabe N, Ishii I, et al. Sphingosine 1-phosphate regulates regeneration and fibrosis after liver injury via sphingosine 1-phosphate receptor 2. J Lipid Res, 2009,50(3):556–564
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
There is no conflict of interest.
Additional information
This work was supported by the National Natural Science Foundation of China (No. 81873505).
Rights and permissions
About this article
Cite this article
Yan, H., Zhao, H., Yi, Sw. et al. Sphingosine-1-Phosphate Protects Against the Development of Cardiac Remodeling via Sphingosine Kinase 2 and the S1PR2/ERK Pathway. CURR MED SCI 42, 702–710 (2022). https://doi.org/10.1007/s11596-022-2600-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-022-2600-x