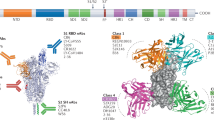
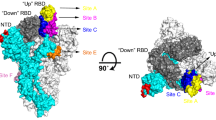
Abstract
Coronavirus disease 2019 is threatening thousands of millions of people around the world. In the absence of specific and highly effective medicines, the treatment of infected persons is still very challenging. As therapeutics, neutralizing antibodies (NAbs) have great potential. Many NAbs have been reported, and most target various regions on the receptor-binding domain of the spike (S) protein, or the N-terminal domain. Several NAbs and NAb cocktails have been authorized for emergency use, and more are in clinical trials or are under development. In this review, considering the angle of binding epitopes on the S protein, we summarize the functions and the underlying mechanisms of a set of well-recognized NAbs and provide guidance for vaccine design and the combinatorial use of these antibodies. In addition, we review the NAbs and NAb cocktails that have been approved for emergency use and discuss the effectiveness of these NAbs for combating severe acute respiratory syndrome coronavirus 2 mutants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Beigel JH, Tomashek KM, Dodd LE. Remdesivir for the Treatment of Covid-19-Preliminary Report. Reply. New Engl J Med, 2020,383(10):994
Group RC. Dexamethasone in hospitalized patients with Covid-19. New Engl J Med, 2021,384(8):693–704
Shen C, Wang Z, Zhao F, et al. Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma. JAMA, 2020,323(16):1582–1589
Simonovich VA, Burgos Pratx LD, Scibona P, et al. A Randomized Trial of Convalescent Plasma in Covid-19 Severe Pneumonia. N Engl J Med, 2021,384(7):619–629
Stanworth S. Convalescent plasma in the management of moderate covid-19 in adults in India: open label phase II multicentre randomized controlled trial. Transfus Med, 2020,371:m3939
Finkelstein MT, Mermelstein AG, Parker Miller E, et al. Structural Analysis of Neutralizing Epitopes of the SARS-CoV-2 Spike to Guide Therapy and Vaccine Design Strategies. Viruses, 2021,13(1):134
Huang Y, Sun H, Yu H, et al. Neutralizing antibodies against SARS-CoV-2: current understanding, challenge and perspective. Antib Ther, 2020,3(4):285–299
Corti D, Purcell LA, Snell G, et al. Tackling COVID-19 with neutralizing monoclonal antibodies. Cell, 2021,184(17):4593–4595
Zhou H, Chen Y, Zhang S, et al. Structural definition of a neutralization epitope on the N-terminal domain of MERS-CoV spike glycoprotein. Nat Commun, 2019,10(1):3068
Wrapp D, Wang N, Corbett KS, et al. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science, 2020,367(6483):1260–1263
Cai Y, Zhang J, Xiao T, et al. Distinct conformational states of SARS-CoV-2 spike protein. Science, 2020,369(6511):1586–1592
Walls AC, Tortorici MA, Snijder J, et al. Tectonic conformational changes of a coronavirus spike glycoprotein promote membrane fusion. Proc Natl Acad Sci USA, 2017,114(42):11 157–11 162
Zang R, Gomez Castro MF, McCune BT, et al. TMPRSS2 and TMPRSS4 promote SARS-CoV-2 infection of human small intestinal enterocytes. Sci Immunol, 2020, 5(47):eabc3582
Walls AC, Park YJ, Tortorici MA, et al. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell, 2020,181(2):281–292, e6
Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell, 2020,181(2):271–280, e8
Glowacka I, Bertram S, Muller MA, et al. Evidence that TMPRSS2 activates the severe acute respiratory syndrome coronavirus spike protein for membrane fusion and reduces viral control by the humoral immune response. J Virol, 2011,85(9):4122–4134
Shulla A, Heald-Sargent T, Subramanya G, et al. A transmembrane serine protease is linked to the severe acute respiratory syndrome coronavirus receptor and activates virus entry. J Virol, 2011,85(2):873–882
Hsieh CL, Goldsmith JA, Schaub JM, et al. Structure-based design of prefusion-stabilized SARS-CoV-2 spikes. Science, 2020,369(6510):1501–1505
White JM, Delos SE, Brecher M, et al. Structures and mechanisms of viral membrane fusion proteins: multiple variations on a common theme. Crit Rev Biochem Mol Biol, 2008,43(3):189–219
Barnes CO, West AP, Huey-Tubman KE, et al. Structures of Human Antibodies Bound to SARS-CoV-2 Spike Reveal Common Epitopes and Recurrent Features of Antibodies. Cell, 2020,182(4):828–842, e16
Kreye J, Reincke SM, Kornau HC, et al. A Therapeutic Non-self-reactive SARS-CoV-2 Antibody Protects from Lung Pathology in a COVID-19 Hamster Model. Cell, 2020,183(4):1058–1069, e19
Shi R, Shan C, Duan X, et al. A human neutralizing antibody targets the receptor-binding site of SARS-CoV-2. Nature, 2020,584(7819):120–124
Wu Y, Wang F, Shen C, et al. A noncompeting pair of human neutralizing antibodies block COVID-19 virus binding to its receptor ACE2. Science, 2020, 368(6496):1274–1278
Meulen JT, van den Brink EN, Poon LLM, et al. Human Monoclonal Antibody Combination against SARS Coronavirus: Synergy and Coverage of Escape Mutants. PLoS Med, 2006,3(7):e237
Huo J, Zhao Y, Ren J, et al. Neutralization of SARS-CoV-2 by destruction of the prefusion Spike. Cell Host Microbe, 2020,28(3):445–454, e6
Zhou D, Duyvesteyn HME, Chen CP, et al. Structural basis for the neutralization of SARS-CoV-2 by an antibody from a convalescent patient. Nat Struct Mol Biol, 2020,27(10):950–958
Wrapp D, DeVlieger D, Corbett KS, et al. Structural Basis for Potent Neutralization of Betacoronaviruses by Single-Domain Camelid Antibodies. Cell, 2020,181(5):1004–1015, e15
Lv Z, Deng YQ, Ye Q, et al. Structural basis for neutralization of SARS-CoV-2 and SARS-CoV by a potent therapeutic antibody. Science, 2020,369(6510):1505–1509
Cao Y, Su B, Guo X, et al. Potent Neutralizing Antibodies against SARS-CoV-2 Identified by High-Throughput Single-Cell Sequencing of Convalescent Patients’ B Cells. Cell, 2020,182(1):73–84
Tortorici MA, M Beltramello, FA Lempp, et al. Ultrapotent human antibodies protect against SARS-CoV-2 challenge via multiple mechanisms. Science, 2020,370(6519):950–957
Liu L, Wang P, Nair MS, et al. Potent neutralizing antibodies against multiple epitopes on SARS-CoV-2 spike. Nature, 2020,584(7821):450–456
Ju B, Zhang Q, Ge J, et al. Human neutralizing antibodies elicited by SARS-CoV-2 infection. Nature, 2020,584(7819):115–119
Custodio TF, Das H, Sheward DJ, et al. Selection, biophysical and structural analysis of synthetic nanobodies that effectively neutralize SARS-CoV-2. Nat Commun, 2020,11(1):5588
Pinto D, Park YJ, Beltramello M, et al. Structural and functional analysis of a potent sarbecovirus neutralizing antibody. bioRxiv, 2020,04.07.023903
Chi X, Yan R, Zhang J, et al. A neutralizing human antibody binds to the N-terminal domain of the Spike protein of SARS-CoV-2. Science, 2020,369(6504):650–655
Wang S, Qiu ZY, Hou YN, et al. AXL is a candidate receptor for SARS-CoV-2 that promotes infection of pulmonary and bronchial epithelial cells. Cell Research, 2021,31(2):126–140
Zhang L, Cao L, Gao X, et al. A proof of concept for neutralizing antibody-guided vaccine design against SARS-CoV-2. bioRxiv, 2020.
Plante JA, Liu Y, Liu J, et al. Spike mutation D614G alters SARS-CoV-2 fitness. Nature, 2021,592(7852):116–121
Korber B, Fischer WM, Gnanakaran S, et al. Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID-19 Virus. Cell, 2020,182(4):812–827, e19
Kidd M, Richter A, Best A, et al. S-variant SARS-CoV-2 lineage B1. 1.7 is associated with significantly higher viral loads in samples tested by ThermoFisher TaqPath RT-qPCR. J Infect Dis, 2021,223(10):1666–1670
Mwenda M, Saasa N, Sinyange N, et al. Detection of B.1.351 SARS-CoV-2 Variant Strain — Zambia, December 2020. Morb Mortal Weekly Rep, 2021,70(8):280–282
da Silva Francisco Jr R, Benites LF, Lamarca AP, et al. Pervasive transmission of E484K and emergence of VUI-NP13L with evidence of SARS-CoV-2 co-infection events by two different lineages in Rio Grande do Sul, Brazil. Virus Res, 2021,296:198–345
Hoffmann M, Arora P, Gross R, et al. SARS-CoV-2 variants B.1.351 and P.1 escape from neutralizing antibodies. Cell, 2021,184(9):2384–2393, e12
Cherian S, Potdar V, Jadhav S, et al. Convergent evolution of SARS-CoV-2 spike mutations, L452R, E484Q and P681R, in the second wave of COVID-19 in Maharashtra, India. bioRxiv, 2021.9(7):1542
Yadav PD, Sapkal GN, Abraham P, et al. Neutralization of variant under investigation B.1.617 with sera of BBV152 vaccinees. Clin Infect Dis, 2021:ciab411
Singh J, Rahman SA, Ehtesham NZ, et al. SARS-CoV-2 variants of concern are emerging in India. Nat Med, 2021,27(7):1131–1133
Pronker ES, Weenen TC, Commandeur H, et al. Risk in vaccine research and development quantified. PLoS One, 2013,8(3):e57755
Le TT, Cramer JP, Chen R, et al. Evolution of the COVID-19 vaccine development landscape. Nat Rev Drug Discov, 2020,19(10):667–668
Qi H, Ma M, Hu C, et al. Antibody binding epitope Mapping (AbMap) of hundred antibodies in a single run. Mol Cell Proteomics, 2021:100 059
Li Y, Lai DY, Zhang NH, et al. Linear epitopes of SARS-CoV-2 spike protein elicit neutralizing antibodies in COVID-19 patients. Cell Mol Immunol, 2020,17(10):1095–1097
Shi R, Shan C, Duan X, et al. A human neutralizing antibody targets the receptor-binding site of SARS-CoV-2. Nature, 2020,584(7819):120–124
Hurlburt NK, Seydoux E, Wan YH, et al. Structural basis for potent neutralization of SARS-CoV-2 and role of antibody affinity maturation. Nat Commun, 2020,11(1):5413
Rogers TF, Zhao R, Huang D, et al. Isolation of potent SARS-CoV-2 neutralizing antibodies and protection from disease in a small animal model. Science, 2020,369(6506):956–963
Du S, Cao Y, Zhu Q, et al. Structurally Resolved SARS-CoV-2 Antibody Shows High Efficacy in Severely Infected Hamsters and Provides a Potent Cocktail Pairing Strategy. Cell, 2020,183(4):1013–1023, e13
Wu NC, Yuan M, Liu H, et al. An alternative binding mode of IGHV3-53 antibodies to the SARS-CoV-2 receptor binding domain. bioRxiv, 2020:2020.07.26.222232
Hansen J, Baum A, Pascal KE, et al. Studies in humanized mice and convalescent humans yield a SARS-CoV-2 antibody cocktail. Science, 2020,369(6506):1010–1014
Piccoli L, Park YJ, Tortorici MA, et al. Mapping Neutralizing and Immunodominant Sites on the SARS-CoV-2 Spike Receptor-Binding Domain by Structure-Guided High-Resolution Serology. Cell, 2020,183(4):1024–1042, e21
Huo J, Zhao Y, Ren J, et al. Neutralization of SARS-CoV-2 by destruction of the prefusion spike. Cell Host Microbe, 2020,28(3):445–454, e6
Pinto D, Park YJ, Beltramello M, et al. Cross-neutralization of SARS-CoV-2 by a human monoclonal SARS-CoV antibody. Nature, 2020,583(7815):290–295
Huo J, Le Bas A, Ruza RR, et al. Neutralizing nanobodies bind SARS-CoV-2 spike RBD and block interaction with ACE2. Nat Struct Mol Biol, 2020,27(9):846–854
Hanke L, Vidakovics Perez L, Sheward DJ, et al. An alpaca nanobody neutralizes SARS-CoV-2 by blocking receptor interaction. Nat Commun, 2020,11(1):4420
Barnes CO, Jette CA, Abernathy MF, et al. SARS-CoV-2 neutralizing antibody structures inform therapeutic strategies. Nature, 2020,588(7839):682–687
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest Statement
The authors declare no competing interests.
This work was supported by the National Natural Science Foundation of China (Nos. 31970130, 31600672, 31670831, and 31370813).
Rights and permissions
About this article
Cite this article
Xue, Jb., Tao, Sc. Epitope Analysis of Anti-SARS-CoV-2 Neutralizing Antibodies. CURR MED SCI 41, 1065–1074 (2021). https://doi.org/10.1007/s11596-021-2453-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-021-2453-8