Summary
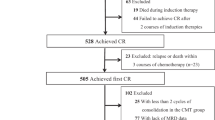
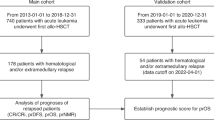
We performed a retrospective analysis to investigate dynamic peri-hematopoietic stem cell transplantation (HSCT) minimal/measurable residual disease (MRD) on outcomes in patients with T-cell acute lymphoblastic leukemia (T-ALL). A total of 271 patients were enrolled and classified into three groups: unchanged negative MRD pre- and post-HSCT group (group A), post-MRD non-increase group (group B), and post-MRD increase group (group C). The patients in group B and group C experienced a higher cumulative incidence of relapse (CIR) (42% vs. 71% vs. 16%, P<0.001) and lower leukemia-free survival (LFS) (46% vs. 21% vs. 70%, P<0.001) and overall survival (OS) (50% vs. 28% vs. 72%, P<0.001) than in group A, but there was no significant difference in non-relapse mortality (NRM) among three groups (14% vs. 12% vs. 8%, P=0.752). Multivariate analysis showed that dynamic peri-HSCT MRD was associated with CIR (HR=2.392, 95% CI, 1.816–3.151, P<0.001), LFS (HR=1.964, 95% CI, 1.546–2.496, P<0.001) and OS (HR=1.731, 95% CI, 1.348–2.222, P<0.001). We also established a risk scoring system based on dynamic peri-HSCT MRD combined with remission status pre-HSCT and onset of chronic graft-versus-host disease (GVHD). This risk scoring system could better distinguish CIR (c=0.730) than that for pre-HSCT MRD (c=0.562), post-HSCT MRD (c=0.616) and pre- and post-MRD dynamics (c=0.648). Our results confirm the outcome predictive value of dynamic peri-HSCT MRD either alone or in combination with other variables for patients with T-ALL.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wang Y, Liu QF, Xu LP, et al.Haploidentical versus Matched-Sibling Transplant in Adults with Philadelphia-Negative High-Risk Acute Lymphoblastic Leukemia: A Biologically Phase III Randomized Study. Clin Cancer Res, 2016,22(14):3467–3476
Zhao XS, Liu YR, Xu LP, et al.Minimal residual disease status determined by multiparametric flow cytometry pretransplantation predicts the outcome of patients with ALL receiving unmanipulated haploidentical allografts. Am J Hematol, 2019,94(4):512–521
Pui CH, Pei D, Coustan-Smith E, et al.Clinical utility of sequential minimal residual disease measurements in the context of risk-based therapy in childhood acute lymphoblastic leukaemia: a prospective study. Lancet Oncol, 2015,16(4):465–474
Sun YQ, Li SQ, Zhao XS,et al. Measurable residual disease of acute lymphoblastic leukemia in allograft settings: how to evaluate and intervene. Expert Rev Anticancer Ther, 2020,20(6):453–564
Chang YJ, Zhao XY, Huang XJ. Granulocyte Colony-Stimulating Factor-Primed Unmanipulated Haploidentical Blood and Marrow Transplantation. Front Immunol, 2019,10:2516
Hay KA, Gauthier J, Hirayama AV, et al.Factors associated with durable EFS in adult B-cell ALL patients achieving MRD-negative CR after CD19 CAR T-cell therapy. Blood, 2019,133(15):1652–1663
Modvig S, Madsen HO, Siitonen SM, et al.Minimal residual disease quantification by flow cytometry provides reliable risk stratification in T-cell acute lymphoblastic leukemia. Leukemia, 2019,33(6):1324–1336
Malard F, Mohty M. Acute lymphoblastic leukaemia. Lancet, 2020,395(10230):1146–1162
Burns MA, Place AE, Stevenson KE, et al.Identification of prognostic factors in childhood T-cell acute lymphoblastic leukemia: Results from DFCI ALL Consortium Protocols 05-001 and 11-001. Pediatr Blood Cancer, 2021,68(1):e28719
Xu M, Liu H, Liu Y, et al.Gene mutations and pretransplant minimal residual disease predict risk of relapse in adult patients after allogeneic hematopoietic stem-cell transplantation for T cell acute lymphoblastic leukemia. Leuk Lymphoma, 2019,60(11):2744–2753
Bader P, Kreyenberg H, von Stackelberg A, et al.Monitoring of minimal residual disease after allogeneic stem-cell transplantation in relapsed childhood acute lymphoblastic leukemia allows for the identification of impending relapse: results of the ALL-BFM-SCT 2003 trial. J Clin Oncol, 2015,33(11):1275–1284
Bader P, Salzmann-Manrique E, Balduzzi A, et al.More precisely defining risk peri-HCT in pediatric ALL: pre- vs post-MRD measures, serial positivity, and risk modeling. Blood Adv, 2019,3(21):3393–3405
Eckert C, Hagedorn N, Sramkova L, et al.Monitoring minimal residual disease in children with high-risk relapses of acute lymphoblastic leukemia: prognostic relevance of early and late assessment. Leukemia, 2015,29(8):1648–1655
Zhao X, Hong Y, Qin Y, et al.The clinical significance of monitoring the expression of the SIL-TAL1 fusion gene in T-cell acute lymphoblastic leukemia after allogeneic hematopoietic stem cell transplantation. Int J Lab Hematol, 2017,39(6):613–619
Li SQ, Fan QZ, Xu LP, et al.Different Effects of Pre-transplantation Measurable Residual Disease on Outcomes According to Transplant Modality in Patients With Philadelphia Chromosome Positive ALL. Front Oncol, 2020,10:320
Wang XY, Fan QZ, Xu LP, et al.The Quantification of Minimal Residual Disease Pre- and Post-Unmanipulated Haploidentical Allograft by Multiparameter Flow Cytometry in Pediatric Acute Lymphoblastic Leukemia. Cytometry B Clin Cytom, 2020,98(1):75–87
Chang YJ, Wang Y, Xu LP, et al.Haploidentical donor is preferred over matched sibling donor for pretransplantation MRD positive ALL: a phase 3 genetically randomized study. J Hematol Oncol, 2020,13(1):27
Teachey DT, Pui CH. Comparative features and outcomes between paediatric T-cell and B-cell acute lymphoblastic leukaemia. Lancet Oncol, 2019,20(3):e142–e54
Cao LQ, Zhou Y, Liu YR, et al.A risk score system for stratifying the risk of relapse in B cell acute lymphocytic leukemia patients after allogenic stem cell transplantation. Chin Med J (Engl), 2021,134(10):1199–1208
Lovisa F, Zecca M, Rossi B, et al.Pre- and post-transplant minimal residual disease predicts relapse occurrence in children with acute lymphoblastic leukaemia. Br J Haematol, 2018,180(5):680–693
Zhao XS, Liu YR, Zhu HH, et al.Monitoring MRD with flow cytometry: an effective method to predict relapse for ALL patients after allogeneic hematopoietic stem cell transplantation. Ann Hematol, 2012,91(2):183–192
Quist-Paulsen P, Toft N, Heyman M, et al.T-cell acute lymphoblastic leukemia in patients 1–45 years treated with the pediatric NOPHO ALL2008 protocol. Leukemia, 2020,34(2):347–357
Tembhare PR, Narula G, Khanka T, et al.Post-induction Measurable Residual Disease Using Multicolor Flow Cytometry Is Strongly Predictive of Inferior Clinical Outcome in the Real-Life Management of Childhood T-Cell Acute Lymphoblastic Leukemia: A Study of 256 Patients. Front Oncol, 2020,10:577
Brammer JE, Saliba RM, Jorgensen JL, et al.Multicenter analysis of the effect of T-cell acute lymphoblastic leukemia subtype and minimal residual disease on allogeneic stem cell transplantation outcomes. Bone Marrow Transplant, 2017,52(1):20–27
Sutton R, Shaw PJ, Venn NC, et al.Persistent MRD before and after allogeneic BMT predicts relapse in children with acute lymphoblastic leukaemia. Br J Haematol, 2015,168(3):395–404
Yeshurun M, Weisdorf D, Rowe JM, et al.The impact of the graft-versus-leukemia effect on survival in acute lymphoblastic leukemia. Blood Adv, 2019,3(4):670–680
Terwey TH, Le Duc TM, Hemmati PG, et al.NIH-defined graft-versus-host disease and evidence for a potent graft-versus-leukemia effect in patients with acute lymphoblastic leukemia. Ann Oncol, 2013,24(5):1363–1370
van Rood JJ, Oudshoorn M. When selecting an HLA mismatched stem cell donor consider donor immune status. Curr Opin Immunol, 2009,21(5):538–543
Ballen KK, Koreth J, Chen YB, et al.Selection of optimal alternative graft source: mismatched unrelated donor, umbilical cord blood, or haploidentical transplant. Blood, 2012,119(9):1972–1980
Huang XJ, Liu DH, Liu KY, et al.Treatment of acute leukemia with unmanipulated HLA-mismatched/haploidentical blood and bone marrow transplantation. Biol Blood Marrow Transplant, 2009,15(2):257–265
Chang YJ, Xu LP, Wang Y, et al.Controlled, Randomized, Open-Label Trial of Risk-Stratified Corticosteroid Prevention of Acute Graft-Versus-Host Disease After Haploidentical Transplantation. J Clin Oncol, 2016,34(16):1855–1863
Chang YJ, Wu DP, Lai YR, et al.Antithymocyte Globulin for Matched Sibling Donor Transplantation in Patients With Hematologic Malignancies: A Multicenter, Open-Label, Randomized Controlled Study. J Clin Oncol, 2020,38(29):3367–3376
Shulman HM, Cardona DM, Greenson JK, et al.NIH Consensus development project on criteria for clinical trials in chronic graft-versus-host disease: II. The 2014 Pathology Working Group Report. Biol Blood Marrow Transplant, 2015,21(4):589–603
International CLLIPIwg. An international prognostic index for patients with chronic lymphocytic leukaemia (CLL-IPI): a meta-analysis of individual patient data. Lancet Oncol, 2016,17(6):779–790
Guo H, Chang YJ, Hong Y, et al.Dynamic immune profiling identifies the stronger graft-versus-leukemia (GVL) effects with haploidentical allografts compared to HLA-matched stem cell transplantation. Cell Mol Immunol, 2021,18(5):1172–1185
Walter RB, Appelbaum FR. Next-generation sequencing for measuring minimal residual disease in AML. Nat Rev Clin Oncol, 2018,15(8):473–474
Liu J, Ma R, Liu YR, et al.The significance of peri-transplantation minimal residual disease assessed by multiparameter flow cytometry on outcomes for adult AML patients receiving haploidentical allografts. Bone Marrow Transplant, 2019,54(4):567–577
Zhou Y, Othus M, Araki D, et al.Pre- and post-transplant quantification of measurable (‘minimal’) residual disease via multiparameter flow cytometry in adult acute myeloid leukemia. Leukemia, 2016,30(7):1456–1464
Enshaei A, O’Connor D, Bartram J, et al.A validated novel continuous prognostic index to deliver stratified medicine in pediatric acute lymphoblastic leukemia. Blood, 2020,135(17):1438–1446
Gabelli M, Veys P, Chiesa R. Current status of umbilical cord blood transplantation in children. Br J Haematol, 2020,190(5):650–683
Acknowledgement
We would like to thank Miss Xu Wu for her assistance in minimal residual disease detection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Additional information
This work was supported (in part) by grants from the Beijing Municipal Science and Technology Commission (No. Z181100009618032), and the National Natural Science Foundation of China (Nos.: 81870141, 82070185, 81670186).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Zd., Wang, Yw., Xu, Lp. et al. Predictive Value of Dynamic Peri-Transplantation MRD Assessed By MFC Either Alone or in Combination with Other Variables for Outcomes of Patients with T-Cell Acute Lymphoblastic Leukemia. CURR MED SCI 41, 443–453 (2021). https://doi.org/10.1007/s11596-021-2390-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-021-2390-6