Summary
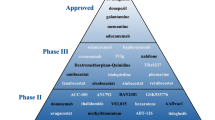
Alzheimer’s disease (AD) is a chronic neurodegenerative disease that mainly causes dementia. It is a serious threat to the health of the global elderly population. Considerable money and effort has been invested in the development of drug therapy for AD worldwide. Many drug therapies are currently under development or in clinical trials, based on two known mechanisms of AD, namely, Aβ toxicity and the abnormal Tau hyperphosphorylation. Numerous drugs are also being developed for other AD associated mechanisms such as neuroinflammation, neurotransmitter imbalance, oxidative damage and mitochondrial dysfunction, neuron loss and degeneration. Even so, the number of drugs that can successfully improve symptoms or delay the progression of the disease remains very limited. However, multi-drug combinations may provide a new avenue for drug therapy for AD. In addition, early diagnosis of AD and timely initiation of treatment may allow drugs that act on the early pathological processes of AD to help improve the symptoms and prevent the progression of the condition.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wenk GL. Neuropathologic changes in Alzheimer’s disease. J Clin Psychiatry, 2003,64(Suppl 9):7–10
Lane CA, Hardy J, Schott JM. Alzheimer’s disease. Eur J Neurol, 2018,25(1):59–70
Howard R, McShane R, Lindesay J, et al. Donepezil and memantine for moderate-to-severe Alzheimer’s disease. N Engl J Med, 2012,366(10): 893–903
Hyde C, Peters J, Bond M, et al. Evolution of the evidence on the effectiveness and cost-effectiveness of acetylcholinesterase inhibitors and memantine for Alzheimer’s disease: systematic review and economic model. Age Ageing, 2013,42(1): 14–20
Cummings J, Lee G, Ritter A, et al. Alzheimer’s disease drug development pipeline: 2020. Alzheimers Dement (N Y), 2020,6(1): e12050
Portelius E, Dean RA, Gustavsson MK, et al. A novel Aβ isoform pattern in CSF reflects γ-secretase inhibition in Alzheimer disease. Alzheimers Res Ther, 2010,2(2): 7
Younkin SG. The role of Aβ42 in Alzheimer’s disease. J Physiol Paris, 1998,92(3–4):289–292
Hardy JA, Higgins GA. Alzheimer’s disease: the amyloid cascade hypothesis. Science, 1992,256(5054):184–186
Yan R, Vassar R. Targeting the β secretase BACE1 for Alzheimer’s disease therapy. Lancet Neurol, 2014,13(3): 319–329
Huang LK, Chao SP, Hu CJ. Clinical trials of new drugs for Alzheimer disease. J Biomed Sci, 2020,27(1), DOI:https://doi.org/10.1186/s12929-019-0609-7
Neumann U, Ufer M, Jacobson LH et al. The BACE-1 inhibitor CNP 520 for prevention trials in Alzheimer’s disease. EMBO Mol Med, 2018,10(11): e9316
Shi X-P, Tugusheva K, Bruce JE, et al. β-Secretase cleavage at amino acid residue 34 in the amyloid β peptide is dependent upon γ-secretase activity. J Biol Chem, 2003,278(23): 21286–21294
Sun X, He G, Song W. BACE2, as a novel APP θ-secretase, is not responsible for the pathogenesis of Alzheimer’s disease in Down syndrome. FASEB J, 2006,20(9):1369–1376
Fluhrer R, Capell A, Westmeyer G, et al. A non-amyloidogenic function of BACE-2 in the secretory pathway. J Neurochem, 2002,81(5): 1011–1020
Abdul-Hay SO, Sahara T, McBride M, et al. Identification of BACE2 as an avid ß-amyloid-degrading protease. Mol. Neurodegene, 2012,7(1):1–12
Alic I, Goh PA, Murray A, et al. Patient-specific Alzheimer-like pathology in trisomy 21 cerebral organoids reveals BACE2 as a gene-dose-sensitive AD-suppressor in human brain. Mol Psychiatry, 2020, DOI:https://doi.org/10.1038/s41380-020-0806-5
Kumar D, Ganeshpurkar A, Kumar D, et al. Secretase inhibitors for the treatment of Alzheimer’s disease: Long road ahead. Eur J Med Chem, 2018,148:436–452
Henley DB, May PC, Dean RA, et al. Development of semagacestat (LY450139), a functional γ-secretase inhibitor, for the treatment of Alzheimer’s disease. Expert Opin Pharmacother, 2009,10(10): 1657–1664
Doody RS, Raman R, Farlow M, et al. A phase 3 trial of semagacestat for treatment of Alzheimer’s disease. N Engl J Med, 2013,369(4): 341–350
Coric V, Salloway S, van Dyck CH, et al. Targeting prodromal Alzheimer disease with avagacestat: a randomized clinical trial. JAMA Neurol, 2015,72(11): 1324–1333
Postina R. A closer look at α-secretase. Curr Alzheimer Res, 2008,5(2):179–186
Marcade M, Bourdin J, Loiseau N, et al. Etazolate, a neuroprotective drug linking GABAA receptor pharmacology to amyloid precursor protein processing. J Neurochem, 2008,106(1): 392–404
Lahiri DK, Chen D, Maloney B, et al. The experimental Alzheimer’s disease drug posiphen [(+)-phenserine] lowers amyloid-β peptide levels in cell culture and mice. J Pharmacol Exp Ther, 2007,320(1): 386–396
Teich AF, Sharma E, Barnwell E, et al. Translational inhibition of APP by Posiphen: Efficacy, pharmacodynamics, and pharmacokinetics in the APP/PS1 mouse. Alzheimers Dement (N Y), 2018,4(1): 37–45
Cummings J, Lee G, Ritter A, et al. Alzheimer’s disease drug development pipeline: 2019. Alzheimers Dement (N Y), 2019,5:272–293
Wang J, Gu BJ, Masters CL, et al. A systemic view of Alzheimer disease—insights from amyloid-β metabolism beyond the brain. Nat Rev Neurol, 2017, 13(10):612
Mawuenyega KG, Sigurdson W, Ovod V, et al. Decreased clearance of CNS β-amyloid in Alzheimer’s disease. Science, 2010,330(6012): 1774–1774
Sevigny J, Chiao P, Bussière T, et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature, 2016,537(7618): 50–56
Selkoe DJ. Alzheimer disease and aducanumab: adjusting our approach. Nat Rev Neurol, 2019,15(7):365–366
Hung SY, Fu WM. Drug candidates in clinical trials for Alzheimer’s disease. J Biomed Sci, 2017,24(1):47
Vandenberghe R, Rinne JO, Boada M, et al. Bapineuzumab for mild to moderate Alzheimer’s disease in two global, randomized, phase 3 trials. Alzheimer’s Res Ther, 2016,8(1): 18
Panza F, Solfrizzi V, Imbimbo BP, et al. Amyloid-directed monoclonal antibodies for the treatment of Alzheimer’s disease: the point of no return? Expert Opin Biol Ther, 2014,14(10):1465–1476
Ostrowitzki S, Lasser RA, Dorflinger E, et al. A phase III randomized trial of gantenerumab in prodromal Alzheimer’s disease. Alzheimer’s Res Ther, 2017, 9(1):95
Tariot PN, Lopera F, Langbaum JB, et al. The Alzheimer’s Prevention Initiative Autosomal-Dominant Alzheimer’s Disease Trial: A study of crenezumab versus placebo in preclinical PSEN1 E280A mutation carriers to evaluate efficacy and safety in the treatment of autosomal-dominant Alzheimer’s disease, including a placebo-treated noncarrier cohort. Alzheimers Dement (N Y), 2018,4:150–160
Boada M, López O, Núñez L, et al. Plasma exchange for Alzheimer’s disease Management by Albumin Replacement (AMBAR) trial: Study design and progress. Alzheimers Dement (N Y), 2019,5:61–69
Wiessner C, Wiederhold KH, Tissot AC, et al. The second-generation active Aβ immunotherapy CAD106 reduces amyloid accumulation in APP transgenic mice while minimizing potential side effects. J Neurosci, 2011, 31(25):9323–9331
Wang CY, Wang PN, Chiu MJ, et al. UB-311, a novel UBITh® amyloid β peptide vaccine for mild Alzheimer’s disease. Alzheimers Dement (N Y), 2017,3(2): 262–272
Dodel R, Du Y, Depboylu C, et al. Intravenous immunoglobulins containing antibodies against β-amyloid for the treatment of Alzheimer’s disease. J Neurol Neurosurg Psychiatry, 2004,75(10): 1472–1474
Dodel R, Neff F, Noelker C, et al. Intravenous immunoglobulins as a treatment for Alzheimer’s disease. Drugs, 2010,70(5): 513–528
Dodel R, Rominger A, Blennow K, et al. P4–411: A randomized, double-blind, placebo-controlled dose-finding trial of intravenous immunoglobulin (IVIG; Octagam® 10%, Octapharma AG) in patients with mild to moderate Alzheimer’s disease (GAM10-04). Alzheimers Dement, 2011,7:e55–e56
Kadavath H, Hofele RV, Biernat J, et al. Tau stabilizes microtubules by binding at the interface between tubulin heterodimers. Proc Natl Acad Sci, 2015,112(24): 7501–7506
Wang JZ, Tian Q. Mechanisms of Tau hyperphosphorylation and its role in neuronal degeneration in Alzheimer’s disease (In Chinese). Adv Biochem Biophysics, 2012,39(8):771–777
Alonso AdC, Grundke-Iqbal I, Iqbal K. Alzheimer’s disease hyperphosphorylated tau sequesters normal tau into tangles of filaments and disassembles microtubules. Nat Med, 1996,2(7):783–787
Feinstein SC, Wilson L. Inability of tau to properly regulate neuronal microtubule dynamics: a loss-of-function mechanism by which tau might mediate neuronal cell death. Biochim Biophys Acta Mol Basis Dis, 2005,1739(2–3):268–279
Wang JZ, Xia YY, Grundke-Iqbal I, et al. Abnormal hyperphosphorylation of tau: sites, regulation, and molecular mechanism of neurofibrillary degeneration. J Alzheimers Dis, 2013,33(Suppl 1):S123–S139
Sereno L, Coma M, Rodriguez M, et al. A novel GSK-3β inhibitor reduces Alzheimer’s pathology and rescues neuronal loss in vivo. Neurobiol Dis, 2009,35(3):359–367
Zhang X, Heng X, Li T, et al. Long-term treatment with lithium alleviates memory deficits and reduces amyloid-β production in an aged Alzheimer’s disease transgenic mouse model. J Alzheimers Dis, 2011,24(4): 739–749
Morris G, Berk M. The putative use of lithium in Alzheimer’s disease. Curr Alzheimer Res, 2016,13(8): 853–861
van Eersel J, Ke YD, Liu X, et al. Sodium selenate mitigates tau pathology, neurodegeneration, and functional deficits in Alzheimer’s disease models. Proc Natl Acad Sci USA, 2010,107(31): 13888–13893
Malpas CB, Vivash L, Genc S, et al. A phase IIa randomized control trial of VEL015 (Sodium Selenate) in mild-moderate Alzheimer’s disease. J Alzheimers Dis, 2016,54(1): 223–232
Chohan MO, Khatoon S, Iqbal IG, et al. Involvement of I2PP2A in the abnormal hyperphosphorylation of tau and its reversal by memantine. FEBS Lett, 2006,580(16): 3973–3979
Iqbal K, Zaidi T, Wen G, et al. Defective brain microtubule assembly in Alzheimer’s disease. Lancet, 1986,328(8504): 421–426
Defensor E, Farmer G, Gan L, et al. Effects of TPI 287, a novel taxoid, on a transgenic mouse model of Alzheimer’s disease. In: Neuroscience Meeting Planner, Washington, DC: Society for Neuroscience, Online, 2014
Tsai RM, Miller Z, Koestler M, et al. Reactions to multiple ascending doses of the microtubule stabilizer TPI-287 in patients with Alzheimer disease, progressive supranuclear palsy, and corticobasal syndrome: a randomized clinical trial. JAMA Neurol, 2020,77(2): 215–224
Paban V, Manrique C, Filali M, et al. Therapeutic and preventive effects of methylene blue on Alzheimer’s disease pathology in a transgenic mouse model. Neuropharmacology, 2014,76:68–79
Pedersen JT, Sigurdsson EM. Tau immunotherapy for Alzheimer’s disease. Trends Mol Med, 2015,21(6):394–402
Theunis C, Crespo-Biel N, Gafner V, et al. Efficacy and safety of a liposome-based vaccine against protein Tau, assessed in tau. P301L mice that model tauopathy. PLoS One, 2013,8(8):e72301
Yiannopoulou KG, Papageorgiou SG. Current and Future Treatments in Alzheimer Disease: An Update. J Cent Nerv Syst Dis, 2020,12:1179573520907397
Rogers J, Webster S, Lue LF, et al. Inflammation and Alzheimer’s disease pathogenesis. Neurobiol Aging, 1996,17(5): 681–686
Heppner FL, Ransohoff RM, Becher B. Immune attack: the role of inflammation in Alzheimer disease. Nat Rev Neurosci, 2015,16(6):358–372
Holmes C, Cunningham C, Zotova E, et al. Systemic inflammation and disease progression in Alzheimer disease. Neurology, 2009,73(10): 768–774
Zhang C, Griciuc A, Hudry E, et al. Cromolyn reduces levels of the Alzheimer’s disease-associated amyloid β-protein by promoting microglial phagocytosis. Sci Rep, 2018,8(1): 1–9
Dominy SS, Lynch C, Ermini F, et al. Porphyromonas gingivalis in Alzheimer’s disease brains: Evidence for disease causation and treatment with small-molecule inhibitors. Sci Adv, 2019,5(1): eaau3333
Detke M, Lynch C, Holsinger L, et al. COR388 for the Treatment of Alzheimer’s Disease (4098). In: AAN Enterprises, 2020
Wang X, Sun G, Feng T, et al. Sodium oligomannate therapeutically remodels gut microbiota and suppresses gut bacterial amino acids-shaped neuroinflammation to inhibit Alzheimer’s disease progression. Cell Res, 2019,29(10): 787–803
Hampel H, Mesulam MM, Cuello AC, et al. The cholinergic system in the pathophysiology and treatment of Alzheimer’s disease. Brain, 2018,141(7): 1917–1933
Wang R, Reddy PH. Role of glutamate and NMDA receptors in Alzheimer’s disease. J Alzheimers Dis, 2017,57(4):1041–1048
Zádori D, Veres G, Szalárdy L, et al. Alzheimer’s disease: recent concepts on the relation of mitochondrial disturbances, excitotoxicity, neuroinflammation, and kynurenines. J Alzheimers Dis, 2018,62(2): 523–547
Hardy J, Adolfsson R, Alafuzoff I, et al. Transmitter deficits in Alzheimer’s disease. Neurochem Int, 1985, 7(4):545–563
Matsunaga S, Kishi T, Iwata N. Memantine monotherapy for Alzheimer’s disease: a systematic review and meta-analysis. PloS One, 2015,10(4):e0123289
Wang X, Wang W, Li L, et al. Oxidative stress and mitochondrial dysfunction in Alzheimer’s disease. Biochim Biophys Acta Mol Basis Dis, 2014,1842(8): 1240–1247
Oken BS, Storzbach DM, Kaye JA. The efficacy of Ginkgo biloba on cognitive function in Alzheimer disease. Arch Neurol, 1998,55(11):1409–1415
Vellas B, Coley N, Ousset PJ, et al. Long-term use of standardised Ginkgo biloba extract for the prevention of Alzheimer’s disease (GuidAge): a randomised placebo-controlled trial. Lancet Neurol, 2012,11(10): 851–859
Kimura T, Hayashida H, Murata M, et al. Effect of ferulic acid and Angelica archangelica extract on behavioral and psychological symptoms of dementia in frontotemporal lobar degeneration and dementia with Lewy bodies. Geriatr Gerontol Int, 2011,11(3): 309–314
Kudoh C, Hori T, Yasaki S, et al. Effects of Ferulic Acid and Angelica archangelica Extract (Feru-guard®) on Mild Cognitive Impairment: A Multicenter, Randomized, Double-Blind, Placebo-Controlled Prospective Trial. J Alzheimers Dis Rep, 2020(Preprint):1–6
Matsuyama K, Yamamoto Y, Sora I. Effect of Feruguard 100M on amyloid-beta deposition in individuals with mild cognitive impairment. Psychogeriatrics, 2020,20(5):726–736
Kabir M, Uddin M, Mamun AA, et al. Combination drug therapy for the management of Alzheimer’s disease. Int J Mol Med Sci, 2020,21(9): 3272
Author information
Authors and Affiliations
Corresponding authors
Additional information
This study was supported by the Ministry of Science and Technology of China (No. 2016YFC1305800), the National Natural Science Foundation of China (No. 31771114 and No. 31929002), the Innovative Research Groups of the National Natural Science Foundation of China (No. 81721005), and the Academic Frontier Youth Team Project to Xiao-chuan WANG from Huazhong University of Science and Technology.
Conflict of Interest Statement
The authors declare that they have no competing interests.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhu, Cc., Fu, Sy., Chen, Yx. et al. Advances in Drug Therapy for Alzheimer’s Disease. CURR MED SCI 40, 999–1008 (2020). https://doi.org/10.1007/s11596-020-2281-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-020-2281-2